Summary
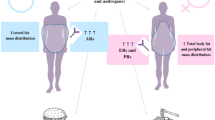
Drug disposition for many drugs has now been studied in obese individuals and some general conclusions can be drawn. Absorption of drugs evaluated to date is unchanged due to obesity. Apparent volume of distribution is greatly increased for some drugs including most benzodiazepines, thiopentone, phenytoin, verapamil and lignocaine (lidocaine). Modest increases in volume of distribution have been noted for methylxanthines, aminoglycosides, vancomycin, ibuprofen, prednisolone and heparin. Distribution of digoxin, cimetidine and procainamide is unchanged in obesity. The mechanism for the increased distribution of some drugs and unchanged distribution of others in obesity is unclear at present. It may be in part due to the lipophilic character of the drug molecule; however, other complex and as yet poorly understood factors contribute to the variability in drug distribution in obese patients.
Protein binding of drugs bound to albumin is not dramatically changed in obesity. In contrast, some studies report that drugs bound to α1-acid glycoprotein (AAG) may have increased binding that is related to increased serum AAG concentration; however, this is not a consistent finding.
Oxidative drug biotransformation is minimally changed in obesity with the exceptions of ibuprofen and prednisolone, for which clearance increases as a highly correlated function of total bodyweight. Drug conjugation uniformly increases as a function of bodyweight in obesity, with paracetamol (acetaminophen), lorazepam and oxazepam having been studied. Drug acetylation may be unchanged in obesity, with only procainamide evaluated at this time. High clearance drugs, including lignocaine, verapamil and midazolam, have no change in clearance in obese individuals compared to normal bodyweight controls. Renal clearance of drugs is little changed for some drugs evaluated (digoxin, cimetidine), and increased for others (aminoglycosides, unmetabolised procainamide).
Characterisation of appropriate animal models of obesity is underway to clarify the mechanisms for these in vivo pharmacokinetic observations in obese man. Two models, the Zucker obese and the obese cafeteria-fed male Sprague-Dawley rat, have provided preliminary physiological pharmacokinetic data with evaluations of theophylline, phenobarbitone and verapamil. The Zucker genetically obese rat may be somewhat limited as a model due to impairment in renal function and impairment in capacity for regulation of cytochrome P-450 activity in obese animals that differs from heterozygous lean Zucker rats. Limitations of the cafeteria-fed Sprague-Dawley rat model of obesity identified to date are that only male rats will eat to a significant extent of obesity, the alteration in animal diet required to achieve obesity is difficult to properly control in pharmacokinetic studies in which dietary alteration in man and animals is well known to change oxidative drug clearance, and that at least 3 months of feeding the specialised diet are required to achieve bodyweight at least 40% in excess of control animals. With further identification and understanding of the limitations of these models, a mechanistic understanding of pharmacokinetic alterations associated with human obesity may be forthcoming.
Similar content being viewed by others
References
Abernethy DR, Divoll M, Greenblatt DJ, Ameer B. Obesity, sex, and acetaminophen disposition. Clinical Pharmacology and Therapeutics 31: 783–790, 1982
Abernethy DR, Greenblatt DJ. Pharmacokinetics of drugs in obesity. Clinical Pharmacokinetics 7: 108–124, 1982
Abernethy DR, Greenblatt DJ. Lidocaine disposition in obesity. American Journal of Cardiology 53: 1183–1186, 1984
Abernethy DR, Greenblatt DJ. Ibuprofen disposition in obesity. Arthritis and Rheumatism 28: 1117–1121, 1985b
Abernethy DR, Greenblatt DJ, Divoll M, Harmatz JS, Shader RI. Alterations in drug distribution and clearance due to obesity. J. Pharmacol. Exp. Therap. 217: 681–685, 1981a
Abernethy DR, Greenblatt DJ, Divoll M, Shader RI. Prolongation of drug half-life due to obesity: studies of desmethyldiazepam (clorazepate). Journal of Pharmaceutical Sciences 71: 942–944, 1982b
Abernethy DR, Greenblatt DJ, Divoll M, Shader RI. Enhanced glucuronide conjugation of drugs in obesity: studies of lorazepam, oxazepam, and acetaminophen. Journal of Laboratory and Clinical Medicine 101: 873–880, 1983
Abernethy DR, Greenblatt DJ, Divoll M, Smith RB, Shader RT. The influence of obesity on the pharmacokinetics of oral alprazolam and triazolam. Clin. Pharmacok. 9: 177–183, 1984a
Abernethy DR, Greenblatt DJ, Matlis R, Gugler R. Cimetidine disposition in obesity. American Journal of Gastroenterology 79: 91–94, 1984b
Abernethy DR, Greenblatt DJ, Mitchell JR. In vitro indices of drug distribution in obesity. Clinical Pharmacology and Therapeutics 35: 224, 1984c
Abernethy DR, Greenblatt DJ, Smith TW. Digoxin disposition in obesity: clinical pharmacokinetic investigation. American Heart Journal 102: 740–744, 1981b
Abernethy DR, Schwartz JB, Todd EL, Mitchell JR. Verapamil dynamics and disposition in obese hypertensives. Federation Proceedings 44: 1128, 1985a
Abernethy DR, Todd EL, Schwartz JB. Caffeine disposition in obesity. Brit. J. Clin. Pharmacol. 20: 61–66, 1985b
Adler M, Schaffner F. Fatty liver hepatitis and cirrhosis in obese patients. American Journal of Medicine 67: 811–816, 1979
Baecke JAH, Burema T, Deurenberg P. Body fatness, relative weight, and frame size in young adults. British Journal of Nutrition 84: 1–6, 1982
Bauer LA, Blouin RA, Griffen WO, Record KE, Bell RM. Amikacin pharmacokinetics in morbidly obese patients. American Journal of Hospital Pharmacy 37: 519–522, 1980
Bauer LA, Edwards WAD, DeHuger EP, Simonowitz DA. Influence of weight on aminoglycoside pharmacokinetics in normal weight and markedly obese patients. European Journal of Clinical Pharmacology 24: 643–647, 1983
Bauer LA, Wareing-Tran C, Edwards WAD, Raisys V, Ferrer L, et al. Cimetidine clearance in the obese. Clinical Pharmacology and Therapeutics 37: 425–430, 1985
Benedeck TH, Blouin RAS, McNamara PJ. Serum protein binding and the role of increased alpha-1-acid glycoprotein in moderately obese male subjects. British Journal of Clinical Pharmacology 18: 941–946, 1984
Benedek IH, Fiske WD, Griffen WO, Bell RM, Blouin RA, et al. Serum alpha-1-acid glycoprotein and the binding of drugs in obesity. Brit. J. Clin. Pharmacol. 16: 751–754, 1983
Blouin RA, Bauer LA, Miller DD, Record KE, Griffen WO. Vancomycin pharmacokinetics in normal and morbidly obese subjects. Antimicrob. Agents Chemoth. 21: 575–580, 1982
Blouin RA, Mann HJ, Griffen WO, Bauer LA, Record KE. Tobramycin pharmacokinetics in morbidly obese patients. Clinical Pharmacology and Therapeutics 26: 508–512, 1979
Blouin RA, Rowse-Brouwer KL, Record KE, Griffen WO, Plezia PM, et al. Amikacin pharmacokinetics in morbidly obese patients undergoing gastric bypass surgery. Clinical Pharmacy 4: 70–72, 1985
Bray GA, York DA. Genetically transmitted obesity in rodents. Physiological Reviews 51: 598–646, 1971
Brouwer KLR, Kostenbauder HB, McNamara PJ, Blouin RA. Phenobarbital in the genetically obese Zucker rat. I. Pharmacokinetics after acute and chronic administration. J. Pharmacol. Exp. Therap. 231: 649–653, 1984a
Brouwer KLR, Kostenbauder HB, McNamara PJ, Blouin RA. Phenobarbital in the genetically obese Zucker rat. II. In vivo and in vitro assessments of microsomal enzyme induction. J. Pharmacol. Exp. Therap. 231: 654–659, 1984b
Browne TR. Status epileptieus, In Miller RR & Greenblatt DJ (Eds). Drug therapy reviews, Vol. 2, pp. 453–455, New York, Elsevier, North Holland, 1979
Bruce A, Andersson M, Arvidsson B, Isaksson B. Body composition: prediction of normal body potassium, body water and body fat in adults on the basis of body height, body weight and age. Scandinavian Journal of Clinical and Laboratory Investigation 40: 461–473, 1980
Christoff PB, Conti DR, Naylor C, Jusko WJ. Procainamide disposition in obesity. Drug Intelligence and Clinical Pharmacy 17: 516–522, 1983
Colliver JA, Frank S, Frank A. Similarity of obesity indices in clinical studies of obese adults: a factor in analytic study. American Journal of Clinical Nutrition 38: 640–647, 1983
de Divitiis O, Fazio S, Petitto M, Maddalena G, Contalde F, et al. Obesity and cardiac function. Circulation 64: 477–482, 1981
Dionne RE, Bauer LA, Gibson GA, Griffen WO, Blouin RA. Estimating creatinine clearance in morbidly obese patients. American Journal of Hospital Pharmacy 38: 841–844, 1981
Doherty JE, Perkins WH, Flanigan WJ. The distribution and concentration of tritiated digoxin in human tissues. Annals of Internal Medicine 66: 116–124, 1967
Dorsey JL, Smith JT. Comparison of hepatic cytochrome P-450 levels of obese mice and nonobese mice. Nutrition Reports International 24: 777–783, 1981
Dundee JW. The influence of body weight, sex and age on the dosage of thiopentone. Brit. J. Anaes. 26: 164–173, 1954
Ewy GA, Groves BM, Ball MF, Nimmo L, Jackson B, et al. Digoxin metabolism in obesity. Circulation 44: 810–814, 1971
Forbes GB, Welle SL. Lean body mass in obesity. International Journal of Obesity 7: 99–107, 1983
Friedman H, Ochs HR, Greenblatt DJ, Shader RI. Tissue distribution of diazepam and its metabolite desmethyldiazepam: a human autopsy study. J. Clin. Pharmacol. 25: 613–615, 1985
Gal P, Jusko WJ, Yurchak AM, Franklin BA. Theophylline disposition in obesity. Clinical Pharmacology and Therapeutics 23: 438–444, 1978
Greenblatt DJ, Abernethy DR, Locniskar A, Harmatz JS, Limjuco RA, et al. Effect of age, gender, and obesity on midazolam kinetics. Anesthesiology 61: 27–35, 1984
Greenblatt DJ, Alen MD, Harmatz JS, Shader RI. Diazepam disposition determinants. Clinical Pharmacology and Therapeutics 27: 301–312, 1980
Greenblatt DJ, Arendt RM, Abernethy DR, Giles HG, Sellers EM et al. In vitro quantitation of benzodiazepine lipophilicity: relation to in vivo distribution. British Journal of Anaesthesia 55: 985–989, 1983
Hubert HR, Feinleib M, McNamara PM, Castelli WP. Obesity as an independent risk factor for cardiovascular disease: a 26-year follow-up of participants in the Framingham heart study. Circulation 67: 968–977, 1983
Ingalls AM, Dickie MM, Snell GD. Obese, new mutation in the mouse. Journal of Heredity 41: 317–318, 1950
Jung D, Mayersohn M, Perrier D, Calkins J, Saunders R. Thiopental disposition in lean and obese patients undergoing surgery. Anesthesiology 56: 265–274, 1982
Kappas A, Anderson KE, Conney AH, Alvares AP. Influence of dietary protein and carbohydrate on antipyrine and theophylline metabolism in man. Clinical Pharmacology and Therapeutics 20: 643–653, 1976
Keys A, Fidanza F, Karvonen MJ, Kimura N, Taylor HL. Indices of relative weight and obesity. Journal of Chronic Disease 25: 329–343, 1972
Knapp TR. A methodological critique of the ‘ideal weight’ concept. J. Am. Med. Assoc. 250: 506–510, 1983
Korsager S. Administration of gentamicin to obese patients. International Journal of Clinical Pharmacology, Therapy and Toxicology 18: 549–553, 1980
Litterst CL. In vitro hepatic drug metabolism and microsomal enzyme induction in genetically obese rats. Biochemical Pharmacology 29: 289–296, 1980
Martin E, Tozer TN, Sheiner LB, Riegelman S. The clinical pharmacokinetics of phenytoin. Journal of Pharmacokinetics and Biopharmaceutics 5: 579–596, 1977
Masoro EJ. Rats as models for the study of obesity. Experimental Aging Research 6: 261–270, 1980
Messerli FH. Obesity in hypertension: how innocent a bystander? American Journal of Medicine 77: 1077–1082, 1984
Messerli FH, Sundgaard-Riise K, Reisin ED, Dreslinski GR, Ventura HO, et al. Dimorphic cardiac adaptations to obesity and arterial hypertension. Ann. Int. Med. 99: 757–761, 1983
Metropolitan Life Insurance Tables. Weights of insured persons in the United States associated with lowest mortality. Statistical Bulletin of the Metropolitan Life Insurance Company [USA] 40: Nov–Dec 1959
Milsap RL, Plaisance KI, Jusko WJ. Prednisolone disposition in obese man. Clin. Pharmacol. Therap. 36: 824–831, 1984
Mujais SK, Tarazi RC, Dustan HP, Fouad FM, Bravo EL. Hypertension in obese patients: hemodynamic and volume studies. Hypertension 4: 84–92, 1982
Mulder GJ, Weitering JG, Scholtens E, Dawson JR, Pang KS. Extrahepatic sulfation and glucuronidation in the rat in vivo: determination of the hepatic extraction ratio of harmol and the extrahepatic contribution to harmol conjugation. Biochemical Pharmacology 33: 3081–3087, 1984
National Institutes of Health Consensus Development Panel on the Health Implications of Obesity. Health implications of obesity: National Institutes of Health consensus development conference statement. Ann. Int. Med. 103: 147–151, 1985
Rohrbaugh TM, Danish M, Ragni MC, Yaffe SJ. The effect of obesity on apparent volume of distribution of theophylline. Pediatric Pharmacology 2: 75–83, 1982
Rolls BJ, Rowe EA. Dietary obesity: Permanent changes in body weight. Journal of Physiology 272: 2P, 1977
Schentag JJ, Cerra FB, Calleri GM, Leising ME, French MA, et al. Age, disease, and cimetidine disposition in healthy subjects and chronically ill patients. Clinical Pharmacology and therapeutics 29: 737–743, 1981
Schwartz SN, Prazin GJ, Lyon JA, Ho M, Pascalle AW. A controlled investigation of the pharmacokinetics of gentamicin and tobramycin in obese subjects. Journal of Infectious Disease 138: 499–505, 1978
Shum L, Jusko WJ. Theophylline disposition in obese rats. J. Pharmacol. Exp. Therap. 228: 380–386, 1984
Simopoulos AP, van Itallie TB. Body weight, health, and longevity. Annals of Internal Medicine 100: 285–295, 1984
Sims EAH. Mechanisms of hypertension in the overweight. Hypertension 4 (Suppl. III): III43–III49, 1982
Sketris I, Lesar T, Zaske DE, Cipolle RJ. Effect of obesity on gentamicin pharmacokinetics. Journal of Clinical Pharmacology 21: 288–293, 1982
Stokholm KH, Brochner-Mortensen J, Holland-Carlsen PF. Increased glomerular filtration rate and adrenocortical function in obese women. Int. J. Obesity 4: 57–63, 1980
Todd EL, Abernethy DR, Mitchell JR. Tissue distribution of ± verapamil in obese and lean Zucker rats. Abstract. Pharmacologist 27(3): 245, 1985
Verbeeck RK, Blackburn JL, Loewen GR. Clinical pharmacokinetics of nonsteroidal anti-inflammatory drugs. Clinical Pharmacokinetics 8: 297–331, 1983
Vesell ES, Penno MB. Assessment of methods to identify sources of interindividual pharmacokinetic variations. Clinical Pharmacokinetics 8: 378–409, 1983
Wallis W, Kutt H, McDowell F. Intravenous diphenylhydantoin in treatment of acute repetitive seizures. Neurology 18: 513–535, 1968
Watson PE, Watson ID, Batt RD. Total body water volumes for adult males and females estimated from simple anthropometric measurements. Am. J. Clin. Nutrit. 33: 27–39, 1980
Wilkinson GR, Shand DG. A physiological approach to hepatic drug clearance. Clin. Pharmacol. Therap. 18: 377–390, 1975
Zucker LM, Zucker TF. Fatty, a new mutation in the rat. Journal of Heredity 52: 275–278, 1961
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Abernethy, D.R., Greenblatt, D.J. Drug Disposition in Obese Humans. Clin-Pharmacokinet 11, 199–213 (1986). https://doi.org/10.2165/00003088-198611030-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-198611030-00002