- © 2005 Canadian Medical Association or its licensors
Abstract
Background: There is uncertainty about whether physician specialty influences the outcomes of outpatients with congestive heart failure after adjustment for differences in case mix. Our objective was to determine the impact of physician specialty on outcomes in outpatients with new-onset congestive heart failure.
Methods: The study was a population-based retrospective cohort study involving patients with new-onset congestive heart failure discharged from 128 acute care hospitals in Alberta between Apr. 1, 1998, and July 1, 2000. Outcomes were resource utilization (clinic visits, emergency department visits and hospital admissions) and survival at 30 days and 1 year.
Results: A total of 3136 patients were discharged from hospital with a new diagnosis of congestive heart failure (median age 76 years, 50% men). Of these, 1062 (34%) received no follow-up visits for cardiovascular care, 738 (24%) were seen by a family physician (FP) alone, 29 (1%) by a specialist (cardiologist or general internist) alone and 1307 (42%) by both a specialist and an FP. Compared with patients who received no follow-up cardiovascular care, patients who received regular cardiovascular follow-up visits with a physician had fewer visits to the emergency department (38% v. 80%), fewer were admitted to hospital (13% v. 94%), and the adjusted 1-year mortality was lower (22% v. 37%) (all p < 0.001). Compared with patients who received combined specialist and FP care, patients cared for exclusively by FPs had fewer outpatient visits (median 9 v. 17 in the first year), fewer of these patients presented to the emergency department (24% v. 45% in the first year), and fewer were readmitted for cardiovascular care (7% v. 16%) (all p < 0.001). However, the adjusted mortality at 1 year was lower among patients treated with combined care (17% v. 28%, p < 0.001) despite a higher burden of comorbidities. In a multivariate model adjusting for comorbidities (with no cardiovascular follow-up visits as the reference category), the mortality was lower among patients followed on an outpatient basis by an FP alone (odds ratio [OR] 0.66, 95% confidence interval [CI] 0.53–0.82) or by an FP and a specialist (OR 0.34, 95% CI 0.28–0.42). In a proportional hazards model with time-dependent covariates (with adjustment for frequency of follow-up visits), the risk of all-cause mortality was reduced significantly (hazard ratio 0.98, 95% CI 0.97– 0.99) with each specialist visit compared with FP care alone.
Interpretation: Patients with congestive heart failure followed by both specialists and FPs had significantly better survival than those followed by FPs alone (or those who received no specific cardiovascular follow-up care). Methods to improve timely and appropriate access to specialists and to improve collaborative care structures are needed.
Congestive heart failure (CHF) afflicts up to 2% of North American adults and, despite many advances in diagnosis and therapy, still portends a poor prognosis, with 1-year mortality of 30%–50%.1,2,3,4,5 Although the prognosis of patients with CHF is poor even with optimal management, suboptimal diagnosis, investigation and treatment of heart failure and comorbidities (e.g., coronary artery disease) in community-dwelling patients contributes to poor survival.6,7,8,9
In previous studies hospital inpatients with CHF who were cared for by specialists received more evidence-based therapies and had better outcomes than those cared for by nonspecialists.8,10,11,12,13 However, none of these studies examined the care delivered after discharge from hospital. Although management in specialized multidisciplinary clinics is associated with better outcomes,14 it is unclear whether similar benefits can be expected when patients are cared for by specialist physicians operating outside the setting of a multidisciplinary clinic. Two recent studies from the United States that reported better outcomes with specialist care7,15 were flawed, in that neither study adjusted for frequency of outpatient visits or the possibility of time-dependent bias16 (whereby some variables, including the number of visits, will change over time).
To address this important public health issue, we sought to determine whether there is a relation between ambulatory care follow-up and outcomes in patients with new-onset CHF.
Methods
We linked 4 databases maintained by Alberta Health and Wellness, which record individual patient interactions with the health care system in Alberta: the Discharge Abstract Database, which records information (including dates, diagnoses and procedures) on all admissions to any of 128 acute care facilities; the Ambulatory Care Database, which tracks all visits to hospital-based physicians' offices and about 100 emergency departments; the Physician Claims Database, which tracks all claims for service (by diagnostic code) made by physicians for the outpatient care of patients in about 5000 physicians' offices; and the Alberta Health Care Insurance Registry, which tracks the vital status of all 3.1 million Albertans.17,18 Outpatient cardiovascular visits were identified as those with International Classification of Diseases, 9th revision, clinical modification (ICD-9-CM) codes 390–459 as the most responsible diagnosis.
We used the Discharge Abstract Database to identify all Alberta residents who had been admitted to hospital at least once between Apr. 1, 1998, and July 1, 2000, with a most responsible diagnosis of CHF (ICD-9-CM code 428.0) and established a cohort of incident cases by excluding patients who had been admitted because of CHF in the preceding 5 years. The accuracy of our data acquisition and the use of discharge coding to identify cases and comorbidities have been described elsewhere.17 Patient comorbidities were also determined from the Discharge Abstract Database, which records up to 16 premorbid or complicating conditions. We characterized hospitals as “urban” if they were located in health regions servicing the populations of Edmonton or Calgary (n = 20); all others were characterized as “rural” (n = 108). Charlson Index comorbidity scores were calculated by the method of Deyo and associates,19 and income was determined by the neighbourhood postal code (Forward Sortation Area) system available from Statistics Canada.
We followed all patients from the date of discharge from the index hospital admission until they died or until Mar. 31, 2001 (whichever came first). Follow-up was 100%. We restricted our analyses to all-cause mortality or readmission for cardiovascular reasons only (any of ICD-9-CM codes 390–459 as the most responsible diagnosis). The primary outcome measure was 1-year mortality; we hypothesized that the 1-year mortality would be lowest (after adjustment for important covariates) among patients followed by a specialist.
The Ambulatory Care and Physician Claims databases record the specialty of each physician who submits claims. Of the 236 specialists in Alberta who submitted claims for cardiovascular diagnoses during the study period, 202 worked in urban regions and 34 in rural areas. Patients with no ambulatory care claims for a cardiovascular diagnosis after their discharge from an acute care facility were classified as having no cardiovascular claims, those seen solely by a family practitioner (FP) were classified as having received FP care, those seen solely by an internist or a cardiologist were classified as having received specialist care, and those seen by both an FP and a specialist were classified as having received combined care if there was at least 1 claim from a physician in each group. Patients who received FP, specialist or combined care constituted the cohort with “any cardiovascular claims.”
We summarized baseline descriptive data in terms of percentages for categorical variables and medians with interquartile ranges for continuous variables. The χ2 test and the Kruskal– Wallis test were used to compare characteristics across groups. We used Kaplan– Meier analysis to examine unadjusted survival patterns across patient groups.
To adjust for differences in baseline characteristics and important covariates between groups, we performed multivariate logistic regression using a backward, stepwise method for 30-day and 1-year outcomes, entering all variables with p < 0.25 on bivariate analysis into the model (including all variables in Table 1). Multiple logistic regression analyses were run to determine which factors were associated with patient death. Statistical significance was accepted at p < 0.05, and all first-order interactions were tested.
Table 1.
A time-dependent analysis is essential when examining the effect of physician follow-up because patients' outcomes can determine their exposure (e.g., patients may die before having the opportunity to attend a follow-up visit). We therefore performed a Cox proportional hazard regression with time-dependent covariates to determine whether specialist visits after discharge from hospital were associated with a lower risk of all-cause mortality. Fixed covariates included in the model were those significantly (p < 0.1) associated with time to death in a backward-stepping Cox model that included only fixed covariates. Postdischarge interactions with the health care system were expressed as time-dependent variables, including cumulative number of cardiovascular visits, cumulative number of cardiovascular visits with a specialist and cumulative number of days in hospital. These time-dependent variables were added to a Cox model containing the significant fixed covariates. All cardiovascular visits (i.e., the number of visits) were included in the model as a measure of overall illness (i.e., sicker patients tend to see physicians more frequently). Proportionality assumptions for the Cox model were tested with both the goodness-of-fit test and log-minus-log test and were met for all analyses.
In a sensitivity analysis, we used patients without any outpatient visits for any diagnosis after discharge as the reference group for the logistic and Cox models.
This study was approved by the Health Research Ethics Board of the University of Alberta.
Results
We identified 3466 patients with an index admission for new-onset CHF between 1998 and 2000; the 3136 (90%) who survived the index hospital admission represented the cohort for this study. Compared with patients who were discharged alive, patients who died during the index hospital stay had higher rates of ischemic heart disease (46% v. 34%), chronic renal disease (22% v. 6%), anemia (25% v. 16%), malignant disease (11% v. 6%) and cerebrovascular disease (9% v. 4%). The median length of stay was 7 days (interquartile range 4–11), and 58% (2015) of the 3466 patients with new-onset CHF were treated in the 10 Alberta hospitals that had more than 100 CHF-related admissions in the study period. During the index admission, 48% of the patients (n = 1490) were treated by FPs, 21% (n = 653) by general internists, 8% (n = 257) by cardiologists and 23% (n = 736) by other physicians.
In the year after the index admission, 34% of the patients (n = 1062) did not see any physician who reported a cardiovascular diagnosis as the reason for the visit (“no cardiovascular claims”). Twenty-four percent (n = 738) had a cardiovascular diagnosis reported by an FP alone, 1% (29) had a cardiovascular diagnosis reported by a specialist alone, and 42% (n = 1307) were seen by both a specialist and an FP for a cardiovascular diagnosis (“combined care”).
Compared with patients who received combined care, those seen exclusively by FPs were older, more likely to be female, more likely to live in a rural setting and less likely to have ischemic heart disease, hypertension, diabetes mellitus or chronic obstructive pulmonary disease (Table 1). Charlson Index comorbidity scores were higher among patients who received combined care than among those seen solely by FPs (p = 0.006); patients with no cardiovascular claims had higher Charlson Index comorbidity scores than patients who had at least 1 cardiovascular outpatient visit (p = 0.002).
Median household income was significantly different across all 3 groups (p < 0.02); patients who received combined care had the highest income (median $45 337, interquartile range $41 818–$54 896) (Table 1).
Within the first year, more of the patients who received no cardiovascular follow-up visits after discharge than of the patients who saw a physician for an outpatient cardiovascular visit had visits to the emergency department (80% v. 38%, p < 0.001) and more were admitted to hospital for cardiovascular reasons (94% v. 13%, p < 0.001); unadjusted mortality rates were also higher among those with no cardiovascular follow-up (7% v. 2% at 30 days, 33% v. 20% at 1 year) (Table 2). Patients without cardiovascular follow-up visits also presented to the emergency department or were admitted to hospital sooner than patients with cardiovascular follow-up visits. Of patients who had a physician visit for a cardiovascular diagnosis, those cared for by FPs had fewer outpatient visits than those who received combined care (median 9 v. 17 in the first year), fewer of these patients had visits to the emergency department (24% v. 45%), and fewer were readmitted for cardiovascular reasons (7% v. 16%) (all p < 0.001). However, FP-treated patients who presented to the emergency department did so earlier than patients who received combined care (median 63 days v. 95 days, p < 0.001). Among the patients who received combined care, the median number of ambulatory visits to a specialist was 3 (interquartile range 1–7), and 16% of these patients received over half of their postdischarge care from a specialist.
Table 2.
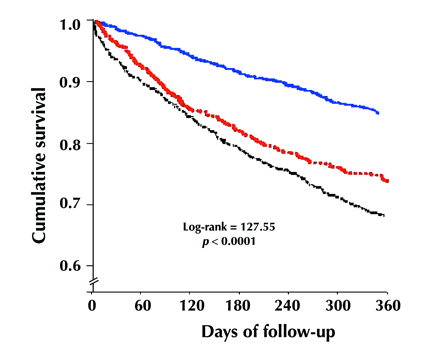
The 30-day and 1-year mortality rates differed significantly between patients who received treatment exclusively from FPs and those who received combined care from an FP and a specialist (Table 2 and Fig. 1, unadjusted survival log-rank = 61, p < 0.001). For the 1-year unadjusted survival analysis, the group with no cardiovascular claims had a higher mortality than the FP-treated group (log-rank = 6.5, p = 0.01) and the group treated by combined care (log-rank = 131.4, p < 0.001). Similarly, in the unadjusted survival analysis using all claims rather than cardiovascular claims, the group who did not receive any follow-up care had a lower survival rate than the FP-treated group (log-rank = 57.0, p < 0.001) and the group treated by combined care (log-rank = 110.3, p < 0.001).
Fig. 1: Kaplan–Meier survival curves for care received, by ambulatory specialty. Blue line = combined care (both specialist and family physician), red line = care by family physician only, black line = no cardiovascular claims (i.e., no physician visits for a cardiovascular cause).
The results for the multivariate logistic regression model for 1-year mortality are outlined in Table 3. Patients who were followed as outpatients by an FP or by both a specialist and an FP for cardiovascular diagnoses had better survival than patients for whom no cardiovascular claims were made. The adjusted odds ratios were virtually identical in the sensitivity analyses when patients with no outpatient claims were used as the reference group.
Table 3.
When a Cox proportional hazards time-dependent covariate model was used to adjust for differing follow-up periods and frequency of follow-up visits, higher age, diabetes mellitus, ischemic heart disease, cerebrovascular disease, peripheral vascular disease, renal failure, malignant disease and cumulative days spent in hospital within 1 year after discharge from the index admission were all associated with a higher mortality rate. Female sex and seeing a specialist as an outpatient (hazards ratio 0.98 per visit to specialist, 95% confidence interval 0.97–0.99]) were associated with lower mortality.
Interpretation
Patients discharged after their first hospital stay for heart failure had better outcomes (fewer emergency department visits, fewer readmissions to hospital and lower mortality rates) if they received regular cardiovascular follow-up visits with a physician; those who saw both an FP and a specialist had the most comorbidities but the lowest mortality rates. Perhaps as a reflection of the higher disease burden in these patients, we found that resource use (hospital admissions, emergency department visits) was greatest for patients followed by both a specialist and an FP.
Why did outcomes differ by care provider? Patients cared for by both a specialist and an FP have more contacts with the medical community; thus, there are more opportunities for deterioration in status to be detected, for medications to be added or titrated, or for the nonpharmacologic therapies that are important in CHF to be reinforced. Other studies of myocardial infarction and CHF have shown greater prescribing of proven efficacious therapies by specialists than by generalist physicians.11,12,17,20,21 Certainly, a wealth of practice audit data have confirmed that angiotensin-converting-enzyme inhibitors, β-blockers and spironolactone are underused and underdosed in CHF patients who are cared for in the community.6,9,17 Indeed, differences in outcomes have been seen for many diseases when care is provided by specialists or generalist physicians.22,23,24 Although specialty heart failure clinics have been shown to improve outcomes and are ubiquitous in most academic centres in North America,25,26 only a minority of heart failure patients are followed in these multidisciplinary clinics.14 Thus, our finding that patients who see both a specialist and their FP have better outcomes is particularly encouraging.
There are limitations to any study that uses data from an administrative database. The key limitation of this study was the lack of information on functional status, severity of CHF, revascularization and medication use. However, we restricted our analysis to patients who had been admitted to hospital with a primary diagnosis of CHF, virtually all of whom would have had class III or IV symptoms.27 We relied upon discharge coding to identify both the most responsible diagnosis and the comorbidities. Previous studies have confirmed the accuracy of our data source for cardiovascular conditions, particularly CHF.28,29,30 Furthermore, although the use of ICD-9-CM code 428 likely underestimates the prevalence of heart failure, it is a highly specific marker for CHF, and we are confident that the patients we identified did have heart failure.29 Most patients have at least 1 hospital admission within 2 years of identification of their heart failure; we extended our exclusion period to 5 years, thus ensuring that our cohort consisted of incident cases of new-onset CHF.
Finally, it is impossible in an observational study to exclude the possibility that unmeasured confounders not captured in the administrative databases may have accounted for the differences in outcomes between the groups. However, we did collect substantial data on comorbidities and we believe that by restricting our analysis to incident cases of CHF and adjusting for length of initial hospital stay along with other comorbidities (including readmissions and frequency of clinic visits), we have minimized the possibility that there were important clinical differences between the groups we examined. Notably, we were not able to adjust for revascularization rates directly, but we did adjust for hospital readmissions in the multivariate logistic regression and Cox proportional hazards models (because coronary artery bypass grafting, stenting and angioplasty in a patient with CHF would be done in the hospital setting). A trial in which patients were randomly assigned to different types of physicians would be the only means to conclusively determine how best to follow CHF patients, but such a trial would be impossible to conduct, because it would be unethical to assign a sicker patient to see an FP alone if they needed combined care and access to the diagnostic resources of a specialist, such as coronary angiography.
Patients with CHF living in the community are at high risk for early and late death, and the risk is greatest among those who do not receive regularly scheduled medical care after discharge. Outpatient care involving both an FP and a specialist is associated with a 66% reduction in the risk of 1-year mortality, even after adjustment for differences in comorbidities, visit frequency and length of follow-up. Our data suggest that methods to improve timely and appropriate access to specialty care are needed, and patients with new-onset CHF are at particular risk for early adverse outcomes if appropriate care is not delivered.
𝛃 See related article page 207
Footnotes
-
This article has been peer reviewed.
Contributors: Justin Ezekowitz, Finlay McAlister and Paul Armstrong contributed to the conception and design of the study. All of the authors contributed to analysis and interpretation of the data. Carl van Walraven and Padma Kaul provided statistical expertise. Justin Ezekowitz, Finlay McAlister, Paul Armstrong and Padma Kaul drafted the article, and all of the authors provided critical revision for important intellectual content. All of the authors gave their final approval of the article submitted.
Acknowledgements: We thank Cynthia Yau for her assistance with data management and statistical support. Justin Ezekowitz is a Canadian Institutes of Health (CIHR) Strategic Training Fellow in TORCH (Tomorrow's Research Cardiovascular Health Professionals). Finlay McAlister is a Population Health Investigator of the Alberta Heritage Foundation for Medical Research and a New Investigator of the CIHR. Carl van Walraven is a Career Scientist of the Ontario Ministry of Health and Long-Term Care. None of the funding agencies had input into the design, conduct, analysis or reporting of this study.
Competing interests: None declared.
Correspondence to: Dr. Paul W. Armstrong, 2-51 Medical Sciences Building, University of Alberta, Edmonton AB T6G 2H7; fax 780 492-9486; paul.armstrongualberta.ca