Abstract
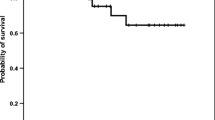
In order to study the relation to the metastatic profile and survival, we evaluated the association between pretreatment serum levels of IL-6, TNF-alpha, and erythropoietin (EPO) and metastatic distribution and survival in 16 patients with metastatic malignant melanoma. IL-6 and TNF-alpha immunoassay (R&D Systems, Inc. Minneapolis, USA) employs the quantitative sandwich enzyme immunoassay technique. The Quantikine IVD EPO ELISA (R&D Systems, Inc.) is based on the double-antibody sandwich method. The baseline serum IL-6 and EPO levels were significantly higher in patients with metastatic malignant melanoma than in the control group (p=0.009 and p=0.033, respectively). Serum IL-6 levels were higher in patients with weight loss (p=0.02), who were anemic (p=0.026), with elevated serum LDH levels (p=0.028), and who were chemotherapy nonresponding (p=0.016). The relationship of serum IL-6 concentration to the tumor burden was not determined. Only serum EPO level was influenced by hemoglobin status (p=0.001). No difference was shown among these three parameters. Elevated serum IL-6 concentration (p=0.002) was found to be an adverse prognostic factor in patients with poor performance status, weight loss, low serum hemoglobin, elevated serum LDH, and unresponsive to chemotherapy. On the other hand, both serum TNF-alpha and EPO levels were of no prognostic value. Serum levels of IL-6 were found to be prognostic factors as valuable as serum LDH levels in patients with metastatic malignant melanoma. Their prognostic value should be further evaluated in a larger patient population.
Similar content being viewed by others
References
Balch CM, et al. Cutaneous melanoma. In: DeVita VT Jr, Hellman S, Rosenberg SA (eds). Cancer: Principles and Practice of Oncology, 5th ed. Lippincot Raven, Philadelphia: 1997.
Manola J, Atkins M, Ibrahim J, Kirkwood J. Prognostic factors in metastatic melanoma: a pooled analysis of Eastern Cooperative Oncology Group Trials. J Clin Oncol 2000; 18: 3782–3793.
Presant CA, Bartolucci AA. Prognostic factors in metastatic malignant melanoma: The Southeastern Cancer Study Group experience. Cancer 1982; 49: 2192–2196.
Ryan L, Kramar A, Borden E. Prognostic factors in metastatic melanoma. Cancer 1993; 71: 2995–3005.
Sirott MN, et al. Prognostic factors in patients with metastatic malignant melanoma: a multivariate analysis. Cancer 1993; 72: 3091–3098.
Barth A, Wanek LA, Morton DL. Prognostic factors in 1,521 melanoma patients with distant metastases. J Am Coll Surg 1995; 181: 193–201.
Brand CU, et al. Prolonged survival of 2 years or longer for patients with disseminated melanoma: an analysis of related prognostic factors. Cancer 1997; 79: 2345–2353.
Falkson CI, Falkson HC. Prognostic factors in metastatic malignant melanoma. Oncology 1998; 55: 59–64.
Eton O, et al. Prognostic factors for survival of patients treated systemically for disseminated melanoma. J Clin Oncol 1998; 16: 1103–1111.
Keilholz U, Martus P, Punt CJ. Prognostic factors for survival and factors associated with long-term remission in patients with advanced melanoma receiving cytokine-based treatments: second analysis of a randomised EORTC Melanoma Group trial comparing interferon-alpha2a (IFNalpha) and interleukin 2 (IL-2) with or without cisplatin. Eur J Cancer 2002; 38: 1501–1511.
Balch CM, et al. A multifactorial analysis of melanoma: IV. Prognostic factors in 200 melanoma patients with distant metastases (stage III). J Clin Oncol 1983; 1: 126–134.
Moretti S, et al. Serum imbalance of cytokines in melanoma patients. Melanoma Res 2001; 11: 395–399.
Deichmann M, et al. Interleukin-6 and its surrogate C-reactive protein are useful serum markers for monitoring metastasized malignant melanoma. J Exp Clin Cancer Res 2000; 19: 301–307.
Vuoristo MS, et al. The value of serum S-100beta and interleukins as tumour markers in advanced melanoma. Melanoma Res 2000; 10: 237–241.
Mauawad R, et al. Is there any relationship between interleukin-6/interleukin-6 receptor modulation and endogenous interleukin-6 release in metastatic malignant melanoma patients treated by biochemotherapy? Melanoma Res 1999; 9: 181–188.
Mouawad R, et al. Endogenous interleukin 6 levels in patients with metastatic malignant melanoma: correlation with tumor burden. Clin Cancer Res 1996; 2: 1405–1409.
Mouawad R, Antoine EC, Khayat D, Soubrane C. Effect of endogenous interleukin-6 on Fas (APO-1/CD95) receptor expression in advanced melanoma patients. Cytokines Cell Mol Ther 2000; 6: 135–140.
Mouawad R, Rixe O, Meric JB, Khayat D, Soubrane C. Serum interleukin-6 concentrations as predictive factor of time to progression in metastatic malignant melanoma patients treated by biochemotherapy: a retrospective study. Cytokines Cell Mol Ther 2002; 7: 151–156.
Szlosarek PW, Balkwill FR. Tumour necrosis factor alpha: a potential target for the therapy of solid tumours. Lancet Oncol 2003; 4: 565, 573.
Ocvirk J, Stabuc B, Rudolf Z, Galvani V, Curin-Serbec V. Serum values of tumour necrosis factor-alpha and of soluble tumour necrosis factor-R55 in melanoma patients. Melanoma Res 2000; 10: 253–258.
Cubillos S, Scallon B, Feldmann M, Taylor P. Effect of blocking TNF on IL-6 levels and metastasis in a B16-BL6 melanoma/mouse model. Anticancer Res. 1997; 17: 2207–2211.
Kettelhack C, Schoter D, Matthias D, Schlag PM. Serum erythropoietin levels in patients with solid tumours. Eur J Cancer 1994; 30A: 1289–1291.
Yasuda Y, et al. Erythropoietin regulates tumour growth of human malignancies. Carcinogenesis 2003; 24: 1021–1029.
Selzer E, et al. Erythropoietin receptor expression in human melanoma cells. Melanoma Res 2000; 10: 421–426.
Brouckaert P, et al. Tumor necrosis factor-alpha augmented tumor response in B16BL6 melanoma-bearing mice treated with stealth liposomal doxorubicin (Doxil ®) correlates with altered Doxil ® pharmacokinetics. Int J Cancer 2004; 109: 442–448.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tas, F., Oguz, H., Argon, A. et al. The value of serum levels of IL-6, TNF-alpha, and erythropoietin in metastatic malignant melanoma. Med Oncol 22, 241–246 (2005). https://doi.org/10.1385/MO:22:3:241
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/MO:22:3:241