Abstract
Background
GRP78 plays an essential role in embryonic development and in the therapeutic treatment and progression of cancer. However, little is known about the role of GRP78 in hepatocellular carcinoma (HCC).
Methods
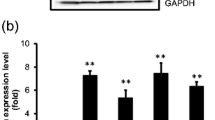
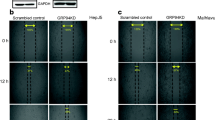
In this study, we characterized five different HCC cell lines to examine GRP78 expression patterns and found that only HepJ5 cells ectopically overexpress GRP78. We knocked down GRP78 expression in HepJ5 cells using a small interfering RNA (siRNA), and the proliferation assay and migration assay were performed.
Results
Using siRNA technique, we could successfully reduce GRP78 expression levels in HepJ5 cells. In a cell growth study, we found that GRP78-siRNA caused no significant changes in cellular proliferation in 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), colony formation, and cell cycle distribution. In a cell migration study, we found that GRP78-siRNA HepJ5 cells had dramatically increased migration ability in Transwell assay.
Conclusions
We conclude that ectopically expressed GRP78 does not contribute to the increased proliferation of HepJ5 cells, but does correlate with the migration of HCC cells under normoxic conditions.





Similar content being viewed by others
References
Parkin DM. Global cancer statistics in the year 2000. Lancet Oncol. 2001;2(9):533–43.
El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132(7):2557–76.
McGlynn KA, Tsao L, Hsing AW, et al. International trends and patterns of primary liver cancer. Int J Cancer. 2001;94(2):290–6.
El-Serag HB, Mason AC. Rising incidence of hepatocellular carcinoma in the United States. N Engl J Med. 1999;340(10):745–50.
Mulder CJ, Rondas AA, Wiltink EH, Tytgat GN. Topical corticosteroids in inflammatory bowel disease. Neth J Med. 1989;35(Suppl 1):S27–34.
Aravalli RN, Steer CJ, Cressman EN. Molecular mechanisms of hepatocellular carcinoma. Hepatology. 2008;48(6):2047–63.
Wang M, Wey S, Zhang Y, et al. Role of the unfolded protein response regulator GRP78/BiP in development, cancer and neurological disorders. Antioxid Redox Signal. 2009;11(9):2307–16.
Li J, Lee B, Lee AS. Endoplasmic reticulum stress-induced apoptosis: multiple pathways and activation of p53-up-regulated modulator of apoptosis (PUMA) and NOXA by p53. J Biol Chem. 2006;281(11):7260–70.
Dong D, Dubeau L, Bading J, et al. Spontaneous and controllable activation of suicide gene expression driven by the stress-inducible grp78 promoter resulting in eradication of sizable human tumors. Hum Gene Ther. 2004;15(6):553–61.
Lee AS. GRP78 induction in cancer: therapeutic and prognostic implications. Cancer Res. 2007;67(8):3496–9.
Luo S, Mao C, Lee B, Lee AS. GRP78/BiP is required for cell proliferation and protecting the inner cell mass from apoptosis during early mouse embryonic development. Mol Cell Biol. 2006;26(15):5688–97.
Jamora C, Dennert G, Lee AS. Inhibition of tumor progression by suppression of stress protein GRP78/BiP induction in fibrosarcoma B/C10ME. Proc Natl Acad Sci USA. 1996;93(15):7690–4.
Xing X, Lai M, Wang Y, et al. Overexpression of glucose-regulated protein 78 in colon cancer. Clin Chim Acta. 2006;364(1–2):308–15.
Wang Q, He Z, Zhang J, et al. Overexpression of endoplasmic reticulum molecular chaperone GRP94 and GRP78 in human lung cancer tissues and its significance. Cancer Detect Prev. 2005;29(6):544–51.
Song MS, Park YK, Lee JH, Park K. Induction of glucose-regulated protein 78 by chronic hypoxia in human gastric tumor cells through a protein kinase C-epsilon/ERK/AP-1 signaling cascade. Cancer Res. 2001;61(22):8322–30.
Park HR, Ryoo IJ, Choo SJ, et al. Glucose-deprived HT-29 human colon carcinoma cells are sensitive to verrucosidin as a GRP78 down-regulator. Toxicology. 2007;229(3):253–61.
Fu Y, Li J, Lee AS. GRP78/BiP inhibits endoplasmic reticulum BIK and protects human breast cancer cells against estrogen starvation-induced apoptosis. Cancer Res. 2007;67(8):3734–40.
Chiou JF, Tai CJ, Huang MT, et al. Glucose-regulated protein 78 is a novel contributor to acquisition of resistance to sorafenib in hepatocellular carcinoma. Ann Surg Oncol. 2009 Oct 15 [Epub ahead of print] PMID: 19830497.
Zhang J, Jiang Y, Jia Z, et al. Association of elevated GRP78 expression with increased lymph node metastasis and poor prognosis in patients with gastric cancer. Clin Exp Metastasis. 2006;23(7–8):401–10.
Misra UK, Deedwania R, Pizzo SV. Activation and cross-talk between Akt, NF-kappaB, and unfolded protein response signaling in 1-LN prostate cancer cells consequent to ligation of cell surface-associated GRP78. J Biol Chem. 2006;281(19):13694–707.
Dai RY, Chen Y, Fu J, et al. p28GANK inhibits endoplasmic reticulum stress-induced cell death via enhancement of the endoplasmic reticulum adaptive capacity. Cell Res. 2009;19(11):1243–57.
Luk JM, Lam CT, Siu AF, et al. Proteomic profiling of hepatocellular carcinoma in Chinese cohort reveals heat-shock proteins (Hsp27, Hsp70, GRP78) up-regulation and their associated prognostic values. Proteomics. 2006;6(3):1049–57.
Gospodarowicz MK, Miller D, Groome PA, et al. The process for continuous improvement of the TNM classification. Cancer. 2004;100(1):1–5.
Scudamore CH, Ragaz J, Kluftinger AM, Owen DA. Hepatocellular carcinoma. A comparison of Oriental and Caucasian patients. Am J Surg. 1988;155(5):659–62.
Primary liver cancer in Japan. Clinicopathologic features and results of surgical treatment. Liver Cancer Study Group of Japan. Ann Surg. 1990;211(3):277–87.
Yamanaka N, Okamoto E, Toyosaka A, et al. Prognostic factors after hepatectomy for hepatocellular carcinomas. A univariate and multivariate analysis. Cancer. 1990;65(5):1104–10.
Dong D, Ko B, Baumeister P, et al. Vascular targeting and antiangiogenesis agents induce drug resistance effector GRP78 within the tumor microenvironment. Cancer Res. 2005;65(13):5785–91.
Wang SK, Liang PH, Astronomo RD, et al. Targeting the carbohydrates on HIV-1: Interaction of oligomannose dendrons with human monoclonal antibody 2G12 and DC-SIGN. Proc Natl Acad Sci USA. 2008;105(10):3690–5.
Sowinski S, Jolly C, Berninghausen O, et al. Membrane nanotubes physically connect T cells over long distances presenting a novel route for HIV-1 transmission. Nat Cell Biol. 2008;10(2):211–9.
Chiu CC, Lin CY, Lee LY, et al. Glucose-regulated protein 78 regulates multiple malignant phenotypes in head and neck cancer and may serve as a molecular target of therapeutic intervention. Mol Cancer Ther. 2008;7(9):2788–97.
A new prognostic system for hepatocellular carcinoma: a retrospective study of 435 patients: the Cancer of the Liver Italian Program (CLIP) investigators. Hepatology. 1998;28(3):751–5.
Hsu C, Shen YC, Cheng CC, et al. Geographic difference in survival outcome for advanced hepatocellular carcinoma: Implications on future clinical trial design. Contemp Clin Trials. 2009 Sep 6 [Epub ahead of print].
Shah SA, Wei AC, Cleary SP, et al. Prognosis and results after resection of very large (>or=10 cm) hepatocellular carcinoma. J Gastrointest Surg. 2007;11(5):589–95.
Pandey D, Lee KH, Wai CT, et al. Long term outcome and prognostic factors for large hepatocellular carcinoma (10 cm or more) after surgical resection. Ann Surg Oncol. 2007;14(10):2817–23.
Sugioka A, Tsuzuki T, Kanai T. Postresection prognosis of patients with hepatocellular carcinoma. Surgery. 1993;113(6):612–8.
Nagorney DM, van Heerden JA, Ilstrup DM, Adson MA. Primary hepatic malignancy: surgical management and determinants of survival. Surgery. 1989;106(4):740–8 (discussion 748–9).
Paquet KJ, Koussouris P, Mercado MA, et al. Limited hepatic resection for selected cirrhotic patients with hepatocellular or cholangiocellular carcinoma: a prospective study. Br J Surg. 1991;78(4):459–62.
Yan F, Wang XM, Pan C, Ma QM. Down-regulation of extracellular signal-regulated kinase 1/2 activity in P-glycoprotein-mediated multidrug resistant hepatocellular carcinoma cells. World J Gastroenterol. 2009;15(12):1443–51.
Silva FP, Hamamoto R, Nakamura Y, Furukawa Y. WDRPUH, a novel WD-repeat-containing protein, is highly expressed in human hepatocellular carcinoma and involved in cell proliferation. Neoplasia. 2005;7(4):348–55.
Watanabe G, Behrns KE, Kim JS, Kim RD. Heat shock protein 90 inhibition abrogates hepatocellular cancer growth through cdc2-mediated G2/M cell cycle arrest and apoptosis. Cancer Chemother Pharmacol. 2009;64(3):433–43.
Seitz SJ, Schleithoff ES, Koch A, et al. Chemotherapy-induced apoptosis in hepatocellular carcinoma involves the p53 family and is mediated via the extrinsic and the intrinsic pathway. Int J Cancer. 2009 Aug 26 [Epub ahead of print].
Chambers AF, Groom AC, MacDonald IC. Dissemination and growth of cancer cells in metastatic sites. Nat Rev Cancer. 2002;2(8):563–72.
Fidler IJ. The pathogenesis of cancer metastasis: the ‘seed and soil’ hypothesis revisited. Nat Rev Cancer. 2003;3(6):453–8.
Lee HK, Xiang C, Cazacu S, et al. GRP78 is overexpressed in glioblastomas and regulates glioma cell growth and apoptosis. Neuro Oncol. 2008;10(3):236–43.
Dong D, Ni M, Li J, et al. Critical role of the stress chaperone GRP78/BiP in tumor proliferation, survival, and tumor angiogenesis in transgene-induced mammary tumor development. Cancer Res. 2008;68(2):498–505.
Acknowledgment
This work was supported by a grant from Taipei Medical University/Chi-Mei Medical Grant (96CM-TMU-06).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Yu-Jia Chang and Chong-Chi Chiu contributed equally to this study.
Rights and permissions
About this article
Cite this article
Chang, YJ., Chiu, CC., Wu, CH. et al. Glucose-Regulated Protein 78 (GRP78) Silencing Enhances Cell Migration but Does Not Influence Cell Proliferation in Hepatocellular Carcinoma. Ann Surg Oncol 17, 1703–1709 (2010). https://doi.org/10.1245/s10434-010-0912-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-010-0912-8