Abstract
Background: Serum levels of CA19-9 have been shown to correlate with both recurrence and survival in patients with pancreatic cancer. However, little is known about the prognosis for patients with undetectable levels of serum CA19-9.
Methods :One hundred twenty-nine patients with pancreatic cancer who underwent preoperative assessment of serum CA19-9 followed by resection with curative intent between 1990 and 2002 were retrospectively analyzed. Data collected included preoperative serum CA19-9 level (U/mL), age, pathologic staging, and survival. Data were analyzed with the SAS system according to four distinct preoperative serum CA19-9 levels: undetectable, normal (<37), 38–200, and ⦔200 U/mL.
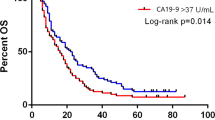
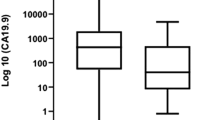
Results: Serum CA19-9 levels ranged from undetectable to 16,300 U/mL. Stage III/IV disease accounted for 86%, 67%, 59%, and 53% of patients in the four CA19-9 groups. The overall median and 5-year survivals were 19 months and 11%, respectively. Survival was similar between nonsecretors and those with normal CA 19-9 levels. However, both groups had statistically significant prolonged survival compared with the two groups with elevated CA 19-9 levels (P = .003). The only factors that were significant on univariate and multivariate analysis for overall survival were lymph node positivity (P = .015 and .002) and CA 19-9 grouping (P = .003 and P < .0001). Although this group of patients presented with predominately advanced-stage disease, their overall survival was superior.
Conclusions: These findings suggest that patients who present with undetectable preoperative CA19-9 levels and potentially resectable pancreatic cancer, regardless of advanced stage, should be considered candidates for aggressive therapy.
Similar content being viewed by others
REFERENCES
Koprowski H, Steplewski Z, Mitchell K, et al. Colorectal carcinoma antigens detected by hybridoma antibodies. Somatic Cell Genetics 1979;5:957–71.
Glenn J, Steinberg WM, Kurtzman SH, et al. Evaluation of the utility of a radioimmunoassay for serum CA 19-9 levels in patients before and after treatment of carcinoma of the pancreas. J Clin Oncol 1988;6:462–8.
Sperti C, Pasquali C, Catalini S, et al. CA 19-9 as a prognostic index after resection for pancreatic cancer. J Surg Oncol 1993;52:137–41.
Montgomery RC, Hoffman JP, Riley LB, et al. Prediction of recurrence and survival by post-resection CA 19-9 values in patients with adenocarcinoma of the pancreas. Ann Surg Oncol 1997;4:551–6.
Safi F, Schlosser W, Falkenreck S, Beger HG. Prognostic values of CA 19-9 serum course in pancreatic cancer. Hepatogastroenterology 1998;45:253–9.
Halm U, Schumann T, Schiefke I, et al. Decrease of CA 19-9 during chemotherapy with gemcitabine predicts survival time in patients with advanced pancreatic cancer. Br J Cancer 2000;82:1013–6.
Magnani J, Nilsson B, Brockhaus M, et al. A monoclonal antibody-defined antigen associated with gastrointestinal cancer is a ganglioside containing sialylated lacto-N-fucopenteose II. J Biological Chem 1982;257:14365–9.
Ritts RE, Pitt HA. CA 19-9 in pancreatic cancer. Surg Oncol Clin North Am 1998;7:93–100.
Lamerz R. Role of tumour markers, cytogenetics. Ann Oncol1990;10(Supp. 4):145–9.
Brockhaus M, Wysocka M, Magnani JL, Steplewski Z, Koprowski H, Ginsburg V. Normal salivary mucin contains the gastrointestinal cancer associated antigen detected by monoclonal antibody CA 19-9 in the serum mucin of patients. Vox Sang 1985;48:34–8.
Kalthoff H, Kreiker C, Schmiegel WH, Greten H, Thiele HG. Characterization of CA 19-9 bearing mucins as physiological exocrine pancreatic secretion products. Cancer Res 1986;46:3605–7.
Safi F, Beger HG, Bittner R, Buchler M, Krautzberger W. CA 19-9 and pancreatic adenocarcinoma. Cancer 1986;57:779–83.
Tian F, Appert HE, Myles J, Howard JM. Prognostic value of serum CA 19-9 levels in pancreatic adenocarcinoma. Ann Surg 1992;215:350–5.
Heinemann V, Schermuly MM, Stieber P, Schulz L, Jungst D, Wilkowski R, Schalhorn A. CA 19-9: A predictor of response in pancreatic cancer treated with gemcitabine and cisplatin. Anticancer Res 1999;19:2433–6.
Magnani JL, Steplewski Z, Koprowski H, Ginsburg V. Identification of the gastrointestinal and pancreatic cancer-associated antigen detected by monoclonal antibody 19-9 in the sera of patients as a mucin. Cancer Res 1983;43:5489–92.
Koprowski H, Brockhaus M, Blaszczyk M, Magnani J, Steplewski Z, Ginsburg V. Lewis blood type may affect the incidence of gastrointestinal cancer. Lancet 1982;1:1332–3.
Kawa S, Oguchi H, Kobayashi T, et al. Elevated serum levels of Dupan-2 in pancreatic cancer patients negative for Lewis blood group phenotype. Br J Cancer 1991;64:899–902.
Yazawa S, Nishihara S, Iwasaki H, et al. Genetic and enzymatic evidence for Lewis enzyme expression in Lewis-negative cancer patients. Cancer Res 1995;55:1473–8.
Hirano K, Kawa S, Oguchi H, et al. Loss of Lewis antigen expression on erythrocytes in some cancer patients with high serum CA 19-9 levels. JNCI 1987;79:1261–8.
Narimatsu H, Iwasaki H, Nakayama F, et al. Lewis and secretor gene dosages affect CA 19-9 and DU-PAN-2 serum levels in normal individuals and colorectal cancer patients. Cancer Res 1998;58:512–8.
Orntoft TF, Vestergaard EM, Holmes E, et al. Influence of Lewis α1–3/4-L-fucosyltransferase (FUT3) gene mutations on enzyme activity, erythrocyte phenotyping, and circulating tumor marker sialyl-Lewis a levels. J Biol Chem 1996;271:32260–8.
Lim JE, Chien MW, Earle CC. Prognostic factors following curative resection for pancreatic adenocarcinoma: a population-based, linked database analysis of 396 patients. Ann Surg 2003;237:74–85.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Berger, A.C., Meszoely, I.M., Ross, E.A. et al. Undetectable Preoperative Levels of Serum CA 19-9 Correlate with Improved Survival for Patients with Resectable Pancreatic Adenocarcinoma. Ann Surg Oncol 11, 644–649 (2004). https://doi.org/10.1245/ASO.2004.11.025
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1245/ASO.2004.11.025