Abstract
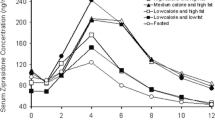
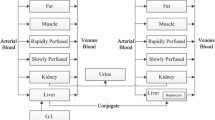
Models have been developed to explain double-peaked plasma concentration-time profiles using mechanisms such as variable absorption and enterohepatic recirculation. Interruption of gastric emptying has also been shown to produce double-peaks, and this work proposes models for analysis of such data. In the presence of levodopa, gastric emptying is interrupted at times associated with double-peaks in pharmacokinetic profiles. Data from a simultaneous scintigraphy and paracetamol absorption study with levodopa was obtained, and models with compartments for stomach, intestine, central and peripheral tissue were developed to describe levodopa and paracetamol pharmacokinetics, including the double-peak phenomenon. The empirical model uses two gastric emptying parameter rates which are applied over separate time periods to describe the varying gastric emptying rate. The semi-mechanistic model uses a feedback mechanism acting via an effect compartment to link the plasma concentration of levodopa to the rate of gastric emptying, allowing levodopa pharmacokinetics to vary the rate of gastric emptying and give rise to a multiple-peaked plasma pharmacokinetic profile. The models were applied to plasma levodopa and paracetamol pharmacokinetic data with and without simultaneous analysis of scintigraphy data, in both cases giving a good fit and in the absence of scintigraphy data adequately predicting the stomach profile. For the semi-mechanistic model, the first-order constant governing gastric emptying was shown to switch between fast and slow values at a critical levodopa effect compartment concentration. New models have thus been proposed for analysis of plasma concentration profiles that exhibit double-peak phenomenon and applied successfully to levodopa data.







Similar content being viewed by others
References
Davies NM, Takemoto JK, Brocks DR, Yanez JA. Multiple peaking phenomena in pharmacokinetic disposition. Clin Pharmacokinet. 2010;49(6):351–77.
Yin OQ, Tomlinson B, Chow AH, Chow MS. A modified two-portion absorption model to describe double-peak absorption profiles of ranitidine. Clin Pharmacokinet. 2003;42(2):179–92.
Keranen T, Gordin A, Harjola VP, Karlsson M, Korpela K, Pentikainen PJ, et al. The effect of catechol-O-methyl transferase inhibition by entacapone on the pharmacokinetics and metabolism of levodopa in healthy-volunteers. Clin Neuropharmacol. 1993;16(2):145–56.
Yamamoto M, Yokochi M, Kuno S, Hattori Y, Tsukamoto Y, Narabayashi H, et al. Effects of tolcapone, a catechol-O-methyltransferase inhibitor, on motor symptoms and pharmacokinetics of levodopa in patients with Parkinson's disease. J Neural Transm. 1997;104(2):229–36.
Jorga K, Banken L, Fotteler B, Snell P, Steimer JL. Population pharmacokinetics of levodopa in patients with Parkinson's disease treated with tolcapone. Clin Pharmacol Ther. 2000;67(6):610–20.
Jorga K, Fotteler B, Sedek G, Nielsen T, Aitken J. The effect of tolcapone on levodopa pharmacokinetics is independent of levodopa/carbidopa formulation. J Neurol. 1998;245(4):223–30.
Mearrick PT, Wade DN, Birkett DJ, Morris J. Metoclopramide, gastric emptying and L-dopa absorption. Aust N Z J Med. 1974;4(2):144–8.
Goetze O, Nikodem AB, Wiezcorek J, Banasch M, Przuntek H, Mueller T, et al. Predictors of gastric emptying in Parkinson's disease. Neurogastroenterol Motil. 2006;18(5):369–75.
Robertson DRC, Renwick AG, Wood ND, Cross N, Macklin BS, Fleming JS, et al. The influence of levodopa on gastric-emptying in man. Br J Clin Pharmacol. 1990;29(1):47–53.
Robertson DR, Wood ND, Everest H, Monks K, Waller DG, Renwick AG, et al. The effect of age on the pharmacokinetics of levodopa administered alone and in the presence of carbidopa. Br J Clin Pharmacol. 1989;28(1):61–9.
Levein NG, Thörn SE, Wattwil M. Dopamine delays gastric emptying and prolongs orocaecal transit time in volunteers. Eur J Anaesthesiol. 1999;16(4):246–50.
Soykan I, Sarosiek I, Shifflett J, Wooten GF, McCallum RW. Effect of chronic oral domperidone therapy on gastrointestinal symptoms and gastric emptying in patients with Parkinson's disease. Mov Disord. 1997;12(6):952–7.
Hellström PM, Grybäck P, Jacobsson H. The physiology of gastric emptying. Best Practice & Research Clinical Anaesthesiology. 2006;20(3):397–407.
Verbeke K. Will the 13C-octanoic acid breath test ever replace scintigraphy as the gold standard to assess gastric emptying? Neurogastroenterol Motil. 2009;21(10):1013–6.
Clements JA, Heading RC, Nimmo WS, Prescott LF. Kinetics of acetaminophen absorption and gastric-emptying in man. Clin Pharmacol Ther. 1978;24(4):420–31.
Clements JA, Nimmo WS, Heading RC, Prescott LF. Physiologically-Based Pharmacokinetic Model for Absorption of Oral Paracetamol in Man. J Pharm Pharmacol. 1978;30 Supple S:60.
Heading RC. Gastric emptying: a clinical perspective. Clin Sci. 1982;63(3):231–5.
Holt S, Reid J, Taylor TV, Tothill P, Heading RC. Gastric emptying of solids in man. Gut. 1982;23(4):292–6.
Mirfazaelian A, Mahmoudian M. A simple pharmacokinetics subroutine for modeling double peak phenomenon. Biopharm Drug Dispos. 2006;27(3):119–24.
Wang Y, Roy A, Sun L, Lau CE. A double-peak phenomenon in the pharmacokinetics of alprazolam after oral administration. Drug Metab Dispos. 1999;27(8):855–9.
Plusquellec Y, Efthymiopoulos C, Duthil P, Houin G. A pharmacokinetic model for multiple sites discontinuous gastrointestinal absorption. Med Eng Phys. 1999;21(8):525–32.
Plusquellec Y, Campistron G, Staveris S, Barre J, Jung L, Tillement JP, et al. A double-peak phenomenon in the pharmacokinetics of veralipride after oral administration: a double-site model for drug absorption. J Pharmacokinet Pharmacodyn. 1987;15(3):225–39.
Godfrey KR, Arundel PA, Dong Z, Bryant R. Modelling the double peak phenomenon in pharmacokinetics. Comput Methods Programs Biomed. 2011;104(2):62–9.
Funaki T. Enterohepatic circulation model for population pharmacokinetic analysis. J Pharm Pharmacol. 1999;51(10):1143–8.
Ezzet F, Krishna G, Wexler DB, Statkevich P, Kosoglou T, Batra VK. A population pharmacokinetic model that describes multiple peaks due to enterohepatic recirculation of ezetimibe. Clin Ther. 2001;23(6):871–85.
Plusquellec Y, Houin G. Drug recirculation model with multiple cycles occurring at unequal time intervals. J Biomed Eng. 1992;14(6):521–6.
Sherwin CMT, Fukuda T, Brunner HI, Goebel J, Vinks AA. The evolution of population pharmacokinetic models to describe the enterohepatic recycling of mycophenolic acid in solid organ transplantation and autoimmune disease. Clin Pharmacokinet. 2011;50(1):1–24.
Marathe PH, Sandefer EP, Kollia GE, Greene DS, Barbhaiya RH, Lipper RA, et al. In vivo evaluation of the absorption and gastrointestinal transit of avitriptan in fed and fasted subjects using gamma scintigraphy. J Pharmacokinet Biopharm. 1998;26(1):1–20.
Berkowitz DM, McCallum RW. Interaction of levodopa and metoclopramide on gastric-emptying. Clin Pharmacol Ther. 1980;27(3):414–20.
GetData Graph Digitizer, http://getdata-graph-digitizer.com/. 2013.
MATLAB 8.1.0.604. Natick, Massachusetts: The MathWorks Inc. R2013a.
Landaw EM, DiStefano 3rd JJ. Multiexponential, multicompartmental, and noncompartmental modeling II data analysis and statistical considerations. Am J Physiol. 1984;246(5 Pt 2):R665–77.
Jamei M, Turner D, Yang J, Neuhoff S, Polak S, Rostami-Hodjegan A, et al. Population-based mechanistic prediction of oral drug absorption. AAPS J. 2009;11(2):225–37.
Acknowledgment
The authors would like to thank Dr Lakshmi Vasist, Dr George Dukes and Dr Malcolm Young, GlaxoSmithKline, Research Triangle Park, NC, USA for helpful discussions during the course of this work.
Conflict of Interest
The authors declare that they have no conflicts of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ogungbenro, K., Pertinez, H. & Aarons, L. Empirical and Semi-Mechanistic Modelling of Double-Peaked Pharmacokinetic Profile Phenomenon Due to Gastric Emptying. AAPS J 17, 227–236 (2015). https://doi.org/10.1208/s12248-014-9693-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-014-9693-5