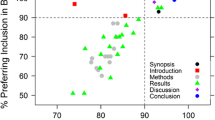
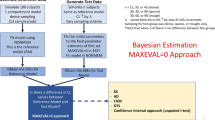
Abstract
Population pharmacokinetic analyses are frequently part of regulatory submissions and are mainly used to provide information on special populations (effects of age, renal impairment, etc) and drug-drug interactions. A varying standard of population analysis reports has been received at the Medical Products Agency in Sweden, some very good and some unassessable. In the latter case, it may be that it is a report of an inadequate analysis or may be a report of a perfectly acceptable analysis, but too little detail has been provided in the report for the conclusions reached to be properly assessed. A sufficient level of detail must be present in these reports in order for them to be assessable and to allow the conclusions reached to be incorporated into the summary of product characteristics. The report should specify the goal(s) of the analysis, describe in detail the origin and nature of the data, clearly describe the model-building process, include a range of goodness of fit (GOF) plots to support decisions made during the model-building process, and demonstrate that the final model is a good description of the data. The use of color in GOF plots is encouraged so that key features are easily visible. Covariate effects in the final model should be clearly presented and their clinical relevance discussed. In the case of many covariates in the final model, it may be useful to perform some simulations to illustrate the effect of various covariate combinations for a series of different “typical” subjects.
Similar content being viewed by others
References
Beal S, Sheiner L, eds. NONMEM Users Guides.NONMEM Project Group, San Francisco: University of California at San Francisco; 1998.
Wählby U, Jonsson EN, Karlsson MO. Assessment of actual significance levels for covariate effects in NONMEM.J Pharmacokinet Pharmacodyn. 2001;28:231–252.
Ribbing J, Jonsson EN. Power, selection bias and predictive performance of the population pharmacokinetic covariate model.J Pharmacokinet Pharmacodyn. 2004;31:109–134.
CPMP/EWP/560/95. Note for Guidance on the Investigation of Drug Interactions (CPMP adopted Dec. 97).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published: October 5, 2005
Rights and permissions
About this article
Cite this article
Wade, J.R., Edholm, M. & Salmonson, T. A guide for reporting the results of population pharmacokinetic analyses: A Swedish perspective. AAPS J 7, 45 (2005). https://doi.org/10.1208/aapsj070245
Received:
Accepted:
DOI: https://doi.org/10.1208/aapsj070245