Abstract
Our objective was to compare the blood pressure of 20-y-old very low birth weight (VLBW; <1.5 kg) individuals with that of normal birth weight (NBW) control individuals. The population included 195 VLBW (92 female and 103 male) and 208 NBW (107 female and 101 male) individuals who were born between 1977 and 1979. Independent effects of birth weight status (VLBW versus NBW) and within the VLBW cohort of intrauterine growth (birth weight z score) were examined via multiple regression analyses. VLBW individuals had a higher mean systolic blood pressure (SBP) than NBW control individuals (114 ± 11 versus 112 ± 13 mm Hg). SBP for VLBW female infants was 110 ± 9 versus NBW 107 ± 12 and for VLBW male individuals was 118 ± 11 versus NBW 117 ± 11 mm Hg. After adjustment for gender, race, and maternal education, the difference in SBP between VLBW and NBW individuals was 1.9 mm Hg but was 3.5 mm after also adjustment for later size (20-y weight and height z scores), which reflects catch-up growth. For female individuals, the difference in SBP between VLBW and NBW individuals was significant both unadjusted and adjusted for later size, whereas for male individuals, the difference was significant only after adjustment for later size. Intrauterine growth did not have a significant effect on SBP within the VLBW group, even after adjustment for later size. VLBW individuals, specifically female individuals, have a higher SBP than NBW control individuals. This is not explained by intrauterine growth failure.
Similar content being viewed by others
Main
Reports of the outcomes of very low birth weight (VLBW) infants have until recently pertained mainly to neurodevelopmental sequelae (1–3). However, as the survivors of neonatal intensive care reach adulthood, there is increasing interest in sequelae that may present later in life (4,5). This interest is heightened by growing evidence that intrauterine and/or early childhood experiences may have long-term implications for adult health. Biologic markers that are considered to be predictors of long-term adult health outcomes include catch-up growth (6), blood pressure (BP) (7–9), and metabolic abnormalities (10).
As part of a longitudinal study of the outcomes of VLBW (<1.5 kg) individuals, we recently reported on young adult health and educational outcomes, behavior, and growth attainment compared with normal birth weight (NBW) control individuals (4,6,11). The objective of the present study was to examine gender-specific BP and to identify perinatal, childhood, and young adult correlates of BP at age 20 y. We hypothesized that the VLBW group would have higher BP at age 20 y compared with the NBW control individuals. Because of our previous finding of greater catch-up growth among VLBW female individuals than among VLBW male individuals (6) and the reported association between catch-up growth and adult cardiovascular risk (12,13), we hypothesized that the difference in BP between the VLBW and NBW control individuals would be greater in the female individuals. We further hypothesized that within the VLBW group, BP at age 20 y would be associated with intrauterine growth failure.
METHODS
VLBW population.
A total of 490 VLBW infants were admitted to Rainbow Babies and Children's Hospital in Cleveland, Ohio, between 1977 and 1979, 312 (64%) of whom survived. A total of 242 (78%) individuals were followed to 20 y of age, 47 of whom were excluded: 25 had neurosensory impairments, one had Liddles syndrome, 12 women were pregnant, and nine had missing BP measurements. The population thus included 195 VLBW individuals who had BP and growth measured at age 20 y. BP and height were also measured in 159 (82%) of their mothers. The VLBW cohort constituted 68% (195 of 287) of the birth cohort who did not have neurosensory impairments. They did not differ significantly from the birth cohort of nonparticipant survivors in maternal sociodemographic status or in infant birth or neonatal descriptors with the exception that there were fewer female individuals (47 versus 61%; p < 0.05) as a result of the exclusion of those who were pregnant at the time of study. Fewer of the mothers who had BP measured compared with those who did not had less than a high school education (17 versus 36%; p < 0.01), but they did not differ in marital status, race, or their children's birth data. Preeclampsia was present in 10% of mothers who had BP measured versus 16% of those who did not (p = 0.384).
Control population.
The control group was selected at age 8 y by means of a population sampling procedure. The original plan was to randomly select 8-y-old NBW children from 50 randomly selected schools that were assigned to six strata on the basis of racial composition and median family income of the schools' catchment area. However, because of busing of school children in the city of Cleveland, the stratification scheme was used only for children who were enrolled in suburban and Catholic schools. For children who attended public school in the city of Cleveland, a random sample was selected. A total of 643 children were selected, 124 (19%) of whom refused to participate and 156 (24%) of whom were either born outside the area or preterm. The original control population thus included 363 NBW individuals who were born in 1977, 1978, and 1979 (3). A total of 233 (64%) were followed to 20 y, 25 of whom were excluded: one had a neurosensory impairment, nine women were pregnant, and 15 had missing BP measures. The control population thus included 208 participants. They constituted 57% of eligible individuals who were recruited at 8 y of age and did not have a neurosensory impairment. BP and height were also measured in 180 (87%) of their mothers.
The 208 control individuals did not differ from the nonparticipating control individuals in birth weight or gender but differed significantly in maternal sociodemographic descriptors: fewer of their mothers had less than a high school education (10 versus 26%), fewer were black (53 versus 75%), and more were married (64 versus 42%; p < 0.001 for all comparisons). The mothers who had BP and height measured did not differ in race, education, or marital status from those who were not measured.
Follow-up protocol.
The methods of neonatal care have been previously described (14,15). The children were followed prospectively, and growth was measured at birth and then longitudinally at the expected term date of delivery (40 wk after the last menstrual period) and then at 8 mo, 20 mo, and 8 y of age corrected for preterm birth and at 20 y postnatal age.
At 20 y of age, BP was measured according to standard procedure recommended by the American Heart Association (16). The research assistants were trained and certified in BP measurement techniques. The individual was seated in a comfortable position with the right arm fully exposed and resting on a supportive surface at heart level. A standard mercury gravity sphygmomanometer was used and an appropriately sized cuff was selected. After inflation of the cuff, the first Korotkoff sound in phase I, which is heard on deflation of the cuff, corresponded to systolic BP (SBP), and the cessation of the fifth sound corresponded to diastolic BP (DBP). Two measurements were made 1–2 min apart and averaged. Standing height was measured with a Harpenden Stadiometer after removal of shoes and stockings (17). The individuals were weighed lightly clothed on an electronic portable scale. To correct for the weight of clothing, similar to others, we subtracted 0.5 kg from the measured weight of female individuals and 1.0 kg from that of male individuals (18,19). A similar proportion of VLBW and NBW study subjects were seen by each of the three research assistants who participated in the study.
Weight and length/height z scores (i.e. SD) were computed from the intrauterine growth standards of Usher and McLean (20) at birth and at the corrected term date and from the revised Centers for Disease Control and Prevention growth data thereafter (21). Body mass index (BMI) was calculated as the relation of body weight to height squared (wt/ht2, kg/m2), and z scores were computed at 8 and 20 y (21). The study was approved by the Institutional Review Board of University Hospitals of Cleveland, and informed consent was obtained from all participants.
Documentation of correlates of BP.
Variables that were examined for their relationship to young adult BP included maternal marital status, level of education, race, a history of preeclampsia, and current maternal BP and height. We used the mother's educational status at the time the child was 8 y old as a proxy for social class because it was considered to span childhood and be more relevant to child growth and development than 20-y status. Infant data that were considered included birth weight, gestational age, the birth weight z score used as a measure of intrauterine growth, small for gestational age (SGA) status [birth weight <−2 SD for gestational age (20)], multiple birth, type of delivery, and the duration of the neonatal hospital stay. We used the duration of neonatal hospital stay as the measure of severity of neonatal illness because it represents an index of immaturity and severity of respiratory distress syndrome such as oxygen and ventilator dependence (22,23). Indices of postnatal growth included distance growth between the time periods of study and weight, height, and BMI z scores at each time period. Additional variables that were considered included age at menarche and chronic illness at 20 y.
Data analysis.
The analyses were carried out for the total population and separately for male and female participants. Variables of interest were compared between the VLBW and NBW participants using the two-sample t test for continuous measures and χ2 for categorical measures.
We initially performed univariate linear regression analyses to examine the effects of the potential correlates of SBP and DBP at age 20 y. Multiple regression analyses then were performed via two models. In model 1, we included VLBW and NBW individuals to examine the independent effect of birth weight status on BP. The specific effect of intrauterine growth on BP could not be examined in model 1 as the birth weight and gestational age of the NBW control individuals was obtained by maternal report at age 8 y and was not believed to be reliable. The effect of birth weight status (VLBW versus NBW) thus was examined in this model, with VLBW status representing the overall impact of being born VLBW rather than of intrauterine growth per se. In model 2, we included only VLBW individuals to examine the independent effect of intrauterine growth (birth weight for gestational age z score) on BP. We used the regression models suggested by Lucas et al. (24). for these analyses. They included 1) an early model that included early size, which we defined as birth weight status in model 1 and birth weight z score in model 2; 2) a later model that included later size, which we defined as the 20-y weight and height z scores; 3) a combined model that included early and later size; and 4) an interaction model that considered the interaction between early and later size. Adjustment for later size is equivalent to adjustment for change in size between birth and 20 y, i.e. postnatal catch-up growth (24). In both models, we adjusted for maternal education, race, and gender where appropriate. In separate models, we also examined the singleton births separately and the effect of being born SGA (birth weight <−2 SD for gestational age) and of growth attainment at the expected date of delivery (i.e. the 40 wk weight z score).
RESULTS
Comparison of VLBW and NBW individuals.
The 195 VLBW individuals had a mean birth weight of 1189 g and mean gestational age of 29.8 wk; male and female VLBW individuals did not differ in maternal sociodemographic status or in infant birth or perinatal data (Table 1). The 208 control individuals had a mean birth weight of 3277 g. They were born at term gestation (≥37 wk), but specific information on their gestational age was not available. The VLBW and NBW individuals did not differ in race or gender (54% VLBW versus 53% NBW were black, and 46% versus 47% were white; 53% VLBW versus 49% NBW were male, and 47% VLBW versus 51% NBW were female). More of the mothers of VLBW participants than mothers of the control individuals had less than a high school education (17 versus 10%, respectively; p < 0.05). They did not differ in marital status (married 59 versus 64%) or race (54 versus 53% black).
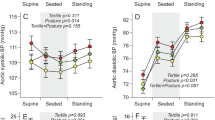
The total VLBW population, including male and female individuals, had a significantly higher SBP than the NBW control individuals (114.2 ± 11 versus 111.9 ± 13; p < 0.05). DBP did not differ significantly between groups (73.1 ± 9 versus 72.6 ± 9).
VLBW male individuals did not differ from their NBW control individuals in SBP or DBP. They had a significantly lower 20-y weight, height, and BMI than the NBW control individuals, as previously reported (6). VLBW female individuals had a significantly higher SBP than the NBW control individuals (110 versus 107 mm Hg; p = 0.03) but did not differ in DBP or in 20-y weight, height, or BMI (see Table 2).
There were no significant differences between the VLBW and NBW groups in the rates of hypertension, i.e. levels of SBP of 140 and above, of DBP of 90 and above, or of prehypertension (systolic BP 120–139) (25). However, significantly more VLBW female individuals had a DBP between 80 and 89 compared with the NBW control individuals (Table 3). The results were similar when the first or second SBP or DBP measurements rather then the mean of the two measurements were considered.
Univariate correlates of SBP and DBP.
Among VLBW male individuals, significant correlates (p < 0.05) of SBP included cesarean section delivery, birth length z score, change in length/height z score between birth and 20 y, and 20-y weight and BMI z scores. Significant correlates of DBP included the maternal DBP, 8- and 20-y weight and BMI z scores, and change in weight z score between birth and 20 y (Tables 4 and 5).
Among VLBW female individuals, significant correlates of SBP included the maternal DBP, cesarean section delivery, the 20-mo length z score, 8- and 20-y weight and height z scores, and change in weight z score between birth and 20 y. Significant correlates of DBP included maternal DBP, the birth weight z score, 20-y weight z score, and change in weight z score between birth and 20 y. The correlation between black race and DBP bordered on significance (p = 0.05).
Within the VLBW group, there were no significant differences in SBP or DBP between single or multiple births or between children who were born appropriate for gestational age or SGA.
Multivariate Analyses
Model 1: effect of VLBW on BP.
This model examined the independent effect of birth weight status (VLBW versus NBW) on BP after adjustment for maternal education and race and gender, where applicable. Examination of the total population revealed that the difference in SBP between the VLBW and NBW individuals bordered on significance (p = 0.08) but became highly significant (difference = 3.5 mm Hg; confidence interval 1.4–5.6; p = 0.001) after also adjusting for later size, i.e. 20-y weight and height (Table 6). Tests for birth weight–gender interactions with SBP and DBP in this model were not significant. However, because of clinical implications, we also examined gender-specific differences in BP. These revealed that among male individuals, there was a significant difference in SBP only after adjustment for later size (mean difference 3.2 mm Hg), whereas among the female individuals, the difference was significant both unadjusted and adjusted for later size (3.4 and 3.8, respectively). DBP did not differ between the VLBW and NBW individuals. Examination of the full Lucas model also revealed that 20-y weight but not height had a significant effect on both SBP and DBP both unadjusted and adjusted for birth weight status. There was no significant interaction between 20-y weight or height and birth weight status (data not shown). Examination of the singleton births revealed results similar to those described above.
Model 2: effect of intrauterine growth on BP within the VLBW cohort.
This model examined the independent effect of intrauterine growth (defined by the birth weight z score) on BP within the VLBW cohort adjusting for maternal education, race, and gender where applicable (see Table 7). Examination of the total population revealed that the birth weight z score had no effect on SBP or DBP unadjusted or adjusted for later size. The tests for interaction indicated that the effects were different for male and female individuals when SBP was considered (p = 0.06 unadjusted and p = 0.01 adjusted for 20-y weight and height) but not for DBP. However, despite the significant interaction, the effect of the birth weight z score on SBP was not significant when the male and female individuals were examined separately (Table 7). Among female individuals the β coefficient of the birth weight z score was significant both unadjusted and adjusted for current size, indicating that the higher the birth weight z score (i.e. the better the intrauterine growth), the lower the DBP. Birth weight z score had no effect on DBP among male individuals. Complete results of the Lucas model also revealed that 20-y weight but not height had a significant effect on SBP and DBP with and without adjustment for the birth weight z score. There was no significant interaction between 20-y weight or height and birth weight z score (data not shown).
The results were similar when the analyses were performed for singleton births only. Intrauterine growth measured categorically as SGA status (i.e. birth weight <−2 SD) had no significant effect on SBP or DBP in male or female individuals. Growth at 40 wk, the expected term date of delivery, similarly had no effect on SBP or DBP (data not shown). The results in models 1 and 2 did not change after inclusion of maternal BP in the analyses.
DISCUSSION
As the initial VLBW survivors of neonatal intensive care have reached young adulthood, there is increasing interest in the predictors of possible chronic sequelae later in life. We examined the effects of VLBW on BP in a cohort of 20-y-old VLBW individuals who were born between 1977 and 1979. Results revealed that VLBW individuals had a higher SBP than the NBW control individuals. These differences bordered on significance after adjustment for maternal race and education and gender and were significant after adjustment for later body size (i.e. 20-y weight and height). Among female individuals, the increase in BP was evident after adjustment for maternal education and race and both with and without adjustment for later size. Among male individuals, however, the effect of VLBW on BP was significant only after adjustment for later size. Birth weight status (i.e. VLBW versus NBW) had no effect on DBP in male or female individuals. Intrauterine growth within the VLBW cohort did not have an effect on SBP in male or female individuals when considered continuously as the birth weight z score or categorically as birth weight <−2 SD. However, among female individuals, intrauterine growth had a significant effect on DBP when measured continuously as the birth weight z score both adjusted and unadjusted for current size. Growth before 40 wk conceptual age, the equivalent to intrauterine growth among term-born infants, had no effect on SBP or DBP in male or female individuals.
This is the largest study of the effects of VLBW status on BP to date. Its strengths include that we considered gender-specific differences and confounding factors such as maternal BP and catch-up growth. Possible weaknesses include that we did not measure nocturnal or ambulatory BP, and thus an effect of heightened anxiety during measure of the BP in female VLBW individuals cannot be excluded (26). We also have no information on the fathers' BP, maternal smoking during pregnancy (27), or on early nutrition, including breastfeeding (28). Because male individuals have higher birth weights than female individuals, our use of the Usher and McLean growth norms (20), which are not gender specific, may have led to an overestimation of the birth weight z score among VLBW male individuals and a corresponding underestimation among VLBW female individuals who have lower birth weights than male individuals. However, additional analyses using the gender-specific birth weight norms of Alexander et al. (29) and Kramer et al. (30) revealed results similar to those obtained using the norms of Usher and McLean (20). Because we had no information on the specific gestational age of the NBW population, we could not calculate their birth weight z score and thus could not examine the effect of their in utero growth on BP. We thus examined the effect of birth weight status per se (VLBW versus NBW) on BP, in which birth weight status represents the overall experience of being born of low birth weight, including the postnatal period. The follow-up rate of VLBW individuals was 78% compared with only 64% of NBW individuals, which might bias the results. The VLBW and NBW groups were similar in terms of maternal sociodemographic characteristics at 8 y of age, when the NBW group was recruited. From 8 to 20 y of age, both groups had greater losses to follow-up among children whose mothers had less education, but more of the losses occurred in the NBW group than in the VLBW group. This resulted in a discrepancy in maternal education between the VLBW and NBW groups at age 20 y. To control for this bias, we adjusted for maternal education in all the analyses.
Reviews of the relationship between birth weight and BP among predominantly term-born populations have revealed that SBP increases with decreasing birth weight, the effect size being 2–3 mm Hg/kg birth weight (31,32). This association between birth weight and BP has been used as evidence to support the “fetal origins hypothesis” of the effects of the early environment on adult cardiovascular and other chronic disease (33). However, the results of the majority of studies on the relationship between birth weight and BP have been based on analyses that adjusted for current weight. This takes into account the change in body size between birth and the time of study, i.e. catch-up growth. Adjustment for current body size increases the strength of the association between birth weight and later BP and is considered to give misleading results (24,34–37). Huxley et al. (37) on reexamination of all of the available data recently concluded that studies that report a strong inverse association between birth weight and BP may reflect the impact of random error, selective emphasis of certain results, inappropriate adjustment for current body size, and lack of adjustment for other confounding factors such as sociodemographic status and maternal BP (36,37). Schluchter (38) undertook a meta-analysis and similarly concluded that publication bias and heterogeneity of studies may have influenced the reported results.
Similar to our results, other studies of preterm births, the majority being VLBW, have reported a significantly higher mean SBP among preterm survivors compared with term-born NBW control individuals (7,8,39–41). The mean difference in SBP is ∼3 mm Hg. This difference has been documented during adolescence (8,39) and young adulthood (7,40,41). Those who specifically examined the association between intrauterine growth and SBP did not find a significant relationship when intrauterine growth was measured as birth weight z score (7,42), birth weight ratio (39,43), or categorically as SGA (40). Similarly, no association has been found between the child's birth weight z score at the time of neonatal discharge, i.e. effect of intrauterine and neonatal growth (42,44).
Doyle et al. (7), whose cohort is most comparable to ours in year of birth, birth weight, and age of study, followed 145 VLBW and 38 NBW individuals to at least 18 y of age and performed both sphygmomanometer and ambulatory BP measurements. He found that VLBW individuals had a significantly higher mean SBP and DBP than the NBW control individuals and in addition that significantly more of the VLBW individuals had an ambulatory SBP above the 95th percentile. Similar to our findings, he did not find a significant relationship between BP and the birth weight z score or an effect of multiple birth. He earlier reported a significantly higher SBP at age 14 y among individuals who received antenatal steroids to accelerate pulmonary maturity (45). Dessens et al. (9), however, reported lower SBP for 20-y-old preterm survivors who received antenatal steroids. VLBW individuals in our cohort did not receive ante- or postnatal steroids.
Correlates of BP in normative populations include gender, race, a family history of hypertension, and other genetic and environmental effects (27,46). Perinatal correlates include maternal anemia (47), pregnancy-induced hypertension, and maternal diet (48–50). Correlates during childhood and adolescence include sodium intake during infancy (51), type of early nutrition (52–55), somatic growth (56,57), obesity, and sexual maturity (46).
It has been postulated that the increase in SBP among preterm survivors may be explained by postnatal experiences associated with prematurity. These include neonatal illness and/or therapy, nutrition, and possibly stress (7,40). There is very little information on these relationships. Singhal et al. (42) reported a significantly lower BP among 13-y-old adolescents who were born preterm and received breast milk compared with term or preterm adolescents who received formula, but protein and calorie intake had no effect on the children's BP. In this longitudinal study, breast milk had no effect on SBP when the children were 8 y old (44). The results of Singhal et al. need replication because only one quarter of the subjects were followed to age 13 y (44). Calculation of the neonatal sodium intake in this population had no effect on BP when the children were 18 mo of age (58).
Morley et al. (59) reported that SBP in 7- to 9-y-old preterm children who were born to smoking compared with nonsmoking mothers was significantly lower among those who were born before 33 wk gestation and significantly higher among those who were born at or after 33 wk gestation. Similar to our results, they did not find a significant relationship between pregnancy-induced hypertension and the children's BP. The nephron number hypothesis of the relationship between poor renal growth and the risk for developing later hypertension also needs to be considered in our cohort (60). Nephrogenesis occurs predominantly between 32 and 36 wk gestation and is completed 4–6 wk before term gestation (61,62). Poor neonatal growth, which is prevalent among VLBW infants during the extrauterine preterm period, may have had detrimental effects on nephrogenesis (15). Kistner et al. (41) reported a significantly higher BP in a small group of 15 young adult women who were born at <32 wk gestation but did not find differences in GFR, renal plasma flow, or urinary albumin excretion when they compared them with 18 term-born SGA and 17 NBW control subjects. This suggests that hyperfiltration in individuals with a lower nephron number maintains a normal filtration rate that may predispose to long-term renal consequences (63). Kistner et al. also reported abnormal retinal vascularization in the preterm group of women as evidenced by significantly longer retinal arterioles and a reduced number of vascular branching points (64). They and others suggested that this abnormal retinal vascularization, which is independent of retinopathy of prematurity, may indicate a general effect of prematurity on the vascular system (64,65). Elastin synthesis, reported to be limited primarily to the fetal and perinatal period between 20 and 40 wk gestation, may possibly also be affected (66).
The greater effect of birth weight on BP in female individuals that we have found has previously been noted by others (67). Murray et al. (68) also reported a negative relationship between birth weight and pulse wave velocity, a measure of arterial compliance in young adult women but not in men. We previously reported that catch-up growth occurred by age 20 y among the VLBW women but not among the men (6). This may partly explain the gender-specific effect of birth weight status (VLBW versus NBW) on SBP and the effect of growth in utero (i.e. birth weight z score) on DBP among our VLBW female individuals. The positive relationship between the velocity of weight gain during childhood and adolescence and BP is well recognized in normative populations (46,56,57,69,70). Catch-up growth in children of lower birth weight (71), in children who were born SGA (72), or after postnatal failure to thrive (73) is similarly associated with higher BP levels.
We conclude that VLBW young adult women have a significantly higher SBP than NBW control subjects. This together with their greater catch-up in growth during childhood and adolescence puts them at greater risk for future cardiovascular sequelae. It has been shown that the higher the BP in a nonhypertensive population, the greater the risk for development of hypertension in later life (74,75). The mean difference in SBP of 3 mm Hg in population BP, although minor, may be of significance for the development of later life hypertension, coronary heart disease, and resultant death as a result of cardiovascular disease (76). Anticipatory guidance and periodic monitoring of BP of VLBW survivors, especially among women, thus is advisable.
Abbreviations
- BMI:
-
body mass index
- BP:
-
blood pressure
- DBP:
-
diastolic blood pressure
- NBW:
-
normal birth weight
- SBP:
-
systolic blood pressure
- SGA:
-
small for gestational age
- VLBW:
-
very low birth weight
References
Taylor HG, Klein N, Minich NM, Hack M 2000 Middle-school-age outcomes in children with very low birthweight. Child Dev 71: 1495–1511
Saigal S, den Ouden L, Wolke D, Hoult L, Paneth N, Streiner DL, Whitaker A, Pinto-Martin J 2003 School-age outcomes in children who were extremely low birth weight from four international population-based cohorts. Pediatrics 112: 943–950
Hack M, Breslau N, Aram D, Weissman B, Klein N, Borawski-Clark E 1992 The effect of very low birth weight and social risk on neurocognitive abilities at school age. J Dev Behav Pediatr 13: 412–420
Hack M, Flannery DJ, Schluchter M, Cartar L, Borawski E, Klein N 2002 Outcomes in young adulthood for very-low-birth-weight infants. N Engl J Med 346: 149–157
Cooke RW 2004 Health, lifestyle, and quality of life for young adults born very preterm. Arch Dis Child 89: 201–206
Hack M, Schluchter M, Cartar L, Rahman M, Cuttler L, Borawski E 2003 Growth of very low birthweight infants to age 20 years. Pediatrics 112: e30–e38. Available: www.pediatrics.org/cgi/content/full/112/1/e30
Doyle LW, Faber B, Callanan C, Morley R 2003 Blood pressure in late adolescence and very low birth weight. Pediatrics 111: 252–257
Pharoah POD, Stevenson CJ, West CR 1998 Association of blood pressure in adolescence with birthweight. Arch Dis Child Fetal Neonatal Ed 79: F114–F118
Dessens AB, Smolders-de Haas H, Koppe JG 2000 Twenty-year follow-up of antenatal corticosteroid treatment. Pediatrics 105: E77. Available: www.pediatrics.org/cgi/content/full/105/6/e77
Singhal A, Fewtrell M, Cole TJ, Lucas A 2003 Low nutrient intake and early growth for later insulin resistance in adolescents born preterm. Lancet 361: 1089–1097
Hack M, Youngstrom EA, Cartar L, Schluchter M, Taylor HG, Flannery D, Klein N, Borawski E 2004 Behavioral outcomes and evidence of psychopathology among very low birth weight infants at age 20 years. Pediatrics 114: 932–940
Forsen T, Eriksson JG, Tuomilehto J, Osmond C, Barker DJ 1999 Growth in utero and during childhood among women who develop coronary heart disease: longitudinal study. BMJ 319: 1403–1407
Fall CH, Osmond C, Barker DJ, Clark PM, Hales CN, Stirling Y, Meade TW 1995 Fetal and infant growth and cardiovascular risk factors in women. BMJ 310: 428–432
Klaus MH, Fanaroff AA 1979 Care of the High Risk Neonate. W.B. Saunders, Philadelphia
Hack M, Merkatz IR, Gordon D, Jones PK, Fanaroff AA 1982 The prognostic significance of postnatal growth in low-birth weight infants. Am J Obstet Gynecol 143: 693–699
Perloff D, Grim C, Flack J, Frohlich ED, Hill M, McDonald M, Morgenstern BZ 1993 Human blood pressure determination by sphygmomanometry. Circulation 88: 2460–2470
1995 Physical status: the use and interpretation of anthropometry: a report of a WHO expert committee. WHO Tech Rep Ser 854: 1–452
Freeman JV, Cole TJ, Chinn S, Jones PR, White EM, Preece MA 1995 Cross sectional stature and weight reference curves for the UK, 1990. Arch Dis Child 73: 17–24
Hardy R, Wadsworth M, Kuh D 2000 The influence of childhood weight and socioeconomic status on change in adult body mass index in a British national birth cohort. Int J Obes Relat Metab Disord 24: 725–734
Usher R, McLean F 1969 Intrauterine growth of live-born Caucasian infants at sea level: standards obtained from measurements in 7 dimensions of infants born between 25 and 44 weeks of gestation. J Pediatr 74: 901–910
Kuczmarski RJ, Ogden CL, Grummer-Strawn LM, Flegal KM, Guo SS, Wei R, Mei Z, Curtain LR, Roche AF, Johnson CL 2000 CDC growth charts: United States. Adv Data 8: 1–27
Cotten M, Oh W, McDonald S, Fanaroff AA, Duara S, Laptook A, Carlo WA, Stoll BJ, Poole K, Wright LL, Goldberg RN 2002 Predictors of prolonged hospital stay (PHS) for extremely low birth weight (ELBW) infants. [abstract]. Pediatr Res 51: 375A
Ross G, Krauss AN, Auld PA 1983 Growth achievement in low-birth-weight premature infants: relationship to neurobehavioral outcome at one year. J Pediatr 103: 105–108
Lucas A, Fewtrell MS, Cole TJ 1999 Fetal origins of adult disease-the hypothesis revisited. BMJ 319: 245–249
Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL, Jones DW, Materson BJ, Oparil S, Wright JT, Roccella EJ Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure 2003 Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. National Heart, Lung, and Blood Institute; National High Blood Pressure Education Program Coordinating Committee. Hypertension 42: 1206–1252
Light KC, Girdler SS, Sherwood A, Bragdon EE, Brownley KA, West SG, Hinderliter AL 1999 High stress responsivity predicts later blood pressure only in combination with positive family history and high life stress. Hypertension 33: 1458–1464
Blane D, Hart CL, Smith GD, Gillis CR, Hole DJ, Hawthorne VM 1996 Association of cardiovascular disease risk factors with socioeconomic position during childhood and during adulthood. BMJ 313: 1434–1438
Booth I 2001 Does the duration of breast feeding matter?. BMJ 322: 625–626
Alexander GR, Himes JH, Kaufman RB, Mor J, Kogan M 1996 A United States National Reference for fetal growth. Obstet Gynecol 87: 163–168
Kramer MS, Platt RW, Wen SW, Joseph KS, Allen A, Abrahamowicz M, Blondel B, Breart G ; Fetal/Infant Health Study Group of the Canadian Perinatal Surveillance System 2001 A new and improved population-based Canadian reference for birth weight for gestational age. Pediatrics 108: E35. Available: www.pediatric.org/cig/content/full/108/2/e35
Law CM, Shiell AW 1996 Is blood pressure inversely related to birth weight? The strength of evidence from a systematic review of the literature. J Hypertens 14: 935–941
Huxley RR, Shiell AW, Law CM 2000 The role of size at birth and postnatal catch-up growth in determining systolic blood pressure: a systematic review of the literature. J Hypertens 18: 815–831
Barker DJP 1998 Mothers, Babies and Health in Later Life. Churchill, Livingston, Edinburgh
Paneth N, Susser M 1995 Early origin of coronary heart disease (the “Barker hypothesis”). BMJ 310: 411–412
Leon DA 1998 Fetal growth and adult disease. Eur J Clin Nutr 52( suppl 1): S72–S82
Kramer MS 2000 Invited commentary: association between restricted fetal growth and adult chronic disease: is it causal? Is it important?. Am J Epidemiol 152: 605–608
Huxley R, Neil A, Collins R 2002 Unraveling the fetal origins hypothesis: is there really an inverse association between birthweight and subsequent blood pressure?. Lancet 360: 659–665
Schluchter MD 2003 Publication bias and heterogeneity in the relationship between systolic blood pressure, birth weight, and catch-up growth—a meta analysis. J Hypertens 21: 1–7
Stevenson CJ, West CR, Pharoah PO 2001 Dermatoglyphic patterns, very low birth weight, and blood pressure in adolescence. Arch Dis Child Fetal Neonatal Ed 84: F18–F22
Irving RJ, Belton NR, Elton RA, Walker BR 2000 Adult cardiovascular risk factors in premature babies. Lancet 355: 2135–2136
Kistner A, Celsi G, Vanpee M, Jacobson SH 2000 Increased blood pressure but normal renal function in adult women born preterm. Pediatr Nephrol 15: 215–220
Singhal A, Cole TJ, Lucas A 2001 Early nutrition in preterm infants and later blood pressure: two cohorts after randomized trials. Lancet 357: 413–419
Morley R, Lister G, Leeson-Payne C, Lucas A 1994 Size at birth and later blood pressure. Arch Dis Child 70: 536–537
Lucas A, Morley R 1994 Does early nutrition in infants born before term programme later blood pressure?. BMJ 309: 304–308
Doyle LW, Ford GW, Davis NM, Callanan C 2000 Antenatal corticosteroid therapy and blood pressure at 14 years of age in preterm children. Clin Sci (Lond) 98: 137–142
Gerber LM, Stern PM 1999 Relationship of body size and body mass to blood pressure: sex-specific and developmental influences. Hum Biol 71: 505–528
Bergel E, Haelterman E, Belizan J, Villar J, Carroli G 2000 Perinatal factors associated with blood pressure during childhood. Am J Epidemiol 151: 594–601
Roseboom TJ, van der Meulen JH, van Montfrans GA, Ravelli AC, Osmond C, Barker DJ, Bleker OP 2001 Maternal nutrition during gestation and blood pressure in later life. J Hypertens 19: 29–34
Campbell DM, Hall MH, Barker DJP, Cross J, Shiell AW, Godfrey KM 1996 Diet in pregnancy and the offspring's blood pressure 40 years later. Br J Obstet Gynaecol 103: 273–280
Churchill D, Perry IJ, Beevers DG 1997 Ambulatory blood pressure in pregnancy and fetal growth. Lancet 349: 7–10
Geleijnse JM, Hofman A, Witteman JC, Hazebroek AA, Valkenburg HA, Grobbee DE 1996 Long-term effects of neonatal sodium restriction on blood pressure. Hypertension 29: 913–917
Martin RM, McCarthy A, Smith GD, Davies DP, Ben-Shlomo Y 2003 Infant nutrition and blood pressure in early adulthood: the Barry Caerphilly Growth study. Am J Clin Nutr 77: 1489–1497
Martin RM, Ness AR, Gunnell D, Emmett P, Davey Smith G, ALSPAC Study Team 2004 Does breast-feeding in infancy lower blood pressure in childhood? The Avon Longitudinal Study of Parent and Children (ALSPAC). Circulation 109: 1259–1266
Forsyth JS, Willatts P, Agostoni C, Bissenden J, Casaer P, Boehm G 2003 Long chain polyunsaturated fatty acid supplementation in infant formula and blood pressure in later childhood: follow up of a randomized controlled trial. BMJ 326: 953–958
Owen CG, Whincup PH, Gilg JA, Cook DG 2003 Effect of breast feeding in infancy on blood pressure in later life: systematic review and meta-analysis. BMJ 327: 1189–1192
Falkner B, Hulman S, Kushner H 1998 Birth weight versus childhood growth as determinants of adult blood pressure. Hypertension 31: 145–150
Katz SH, Hediger ML, Schall JI, Bowers EJ, Barker WF, Aurand S, Eveleth PB, Gruskin AB, Parks JS 1980 Blood pressure, growth and maturation from childhood through adolescence. Mixed longitudinal analysis of the Philadelphia Blood Pressure Project. Hypertension 2: 55–69
Lucas A, Morley R, Hudson GJ, Bamford MF, Boon A, Crowle P, Dossetor JF, Pearse R 1988 Early sodium intake and later blood pressure in preterm infants. Arch Dis Child 63: 656–657
Morley R, Leeson C, Payne C, Lister G, Lucas A 1995 Maternal smoking and blood pressure in 7.5 to 8 year old offspring. Arch Dis Child 72: 120–124
Brenner BM, Garcia DL, Anderson S 1988 Glomeruli and blood pressure. Less of one, more the other?. Am J Hypertens 1: 335–347
Hinchliffe SA, Sargent PH, Howard CV, Chan YF, van Velszen D 1991 Human intrauterine renal growth expressed in absolute number of glomeruli assessed by the disector method and Cavalieri principle. Lab Invest 64: 777–784
Ingelfinger JR 2003 Is microanatomy destiny?. N Engl J Med 348: 99–100
Hostetter TH, Olson JL, Rennke HG, Venkatachalam MA, Brenner BM 2001 Hyperfiltration in remnant nephrons: a potentially adverse response to renal ablation. J Am Soc Nephrol 12: 1315–1325
Kistner A, Jacobson L, Jacobson SH, Svensson E, Hellstrom A 2002 Low gestational age associated with abnormal retinal vascularization and increased blood pressure in adult women. Pediatr Res 51: 675–680
Hellström A, Hard AL, Niklasson A, Svensson E, Jacobsson B 1998 Abnormal retinal vascularization in preterm children as a general vascular phenomenon. Lancet 352: 1827
Martyn CN, Greenwald SE 1997 Impaired synthesis of elastin in walls of aorta and large conduit arteries during early development as an initiating event in pathogenesis of systemic hypertension. Lancet 350: 953–955
Taylor SJ, Whincup PH, Cook DG, Papacosta O, Walker M 1997 Size at birth and blood pressure: cross sectional study in 8–11 year old children. BMJ 314: 475–480
Murray LJ, Gallagher AM, Boreham CA, Savage M, Smith GD 2001 Sex specific difference in the relation between birth weight and arterial compliance in young adults: The Young Hearts Project. J Epidemiol Community Health 55: 665–666
Akahoshi M, Soda M, Carter RL, Nakashima E, Shimaoka K, Seto S, Yano K 1996 Correlation between systolic blood pressure and physical development in adolescence. Am J Epidemiol 144: 51–58
Lauer RM, Clarke WR 1984 Childhood risk factors for high adult blood pressure: the Muscatine Study. Pediatrics 84: 633–641
Horta BL, Barros FC, Victora CG, Cole TJ 2003 Early and late growth and blood pressure in adolescence. J Epidemiol Community Health 57: 226–230
Williams S, Poulton R 2002 Birth size, growth, and blood pressure between the ages of 7 and 26 years: failure to support the fetal origins hypothesis. Am J Epidemiol 155: 849–852
Walker SP, Gaskin P, Powell CA, Bennett FI, Forrester TE, Grantham-McGregor S 2001 The effects of birth weight and postnatal linear growth retardation on blood pressure at age 11–12 years. J Epidemiol Community Health 55: 394–398
Vasan RS, Larson MG, Leip EP, Kannel WB, Levy D 2001 Assessment of frequency of progression to hypertension in nonhypertensive participants in the Framingham Heart Study: a cohort study. Lancet 358: 1682–1686
Miura K, Daviglus ML, Dyer AR, Liu K, Garside DB, Stamler J, Greenland P 2001 Relationship of blood pressure to 25-year mortality due to coronary heart disease, cardiovascular diseases, and all causes in young adult men: the Chicago Heart Association Detection Project in Industry. Arch Intern Med 161: 1501–1508
Stamler J, Stamler R, Neaton JD 1993 Blood pressure, systolic and diastolic, and cardiovascular risks. US population data. Arch Intern Med 153: 598–615
Acknowledgements
We thank Blanche Caron, Debra Hoffman, Susan McGrath, Miriam Curran, Elizabeth Carter, and Terry Reid for assistance in compiling and analyzing the data and Alpher Torres for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by grants from the National Institutes of Health (RO1 HD34177 and M01 RR00080, General Clinical Research Center) and in part by a grant (96-46) from the Genentech Foundation for Growth and Development.
Rights and permissions
About this article
Cite this article
Hack, M., Schluchter, M., Cartar, L. et al. Blood Pressure Among Very Low Birth Weight (<1.5 kg) Young Adults. Pediatr Res 58, 677–684 (2005). https://doi.org/10.1203/01.PDR.0000180551.93470.56
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/01.PDR.0000180551.93470.56
This article is cited by
-
In infants with congenital heart disease autonomic dysfunction is associated with pre-operative brain injury
Pediatric Research (2022)
-
Autonomic nervous system development and its impact on neuropsychiatric outcome
Pediatric Research (2019)
-
Association among prematurity (<30 weeks’ gestational age), blood pressure, urinary albumin, calcium, and phosphate in early childhood
Pediatric Nephrology (2017)
-
Hypertensive Pregnancy Disorders and Future Renal Disease
Current Hypertension Reports (2014)
-
Is late-preterm birth a risk factor for hypertension in childhood?
European Journal of Pediatrics (2014)