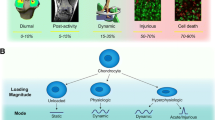
Abstract
The critical importance of mechanical signals to the health and maintenance of articular cartilage has been well demonstrated. Tissue engineers have taken a cue from normal cartilage physiology and incorporated the use of mechanical stimulation into their attempts to engineer functional cartilage. However, the specific types of mechanical stimulation that are most beneficial, and the mechanisms that allow a chondrocyte to perceive and respond to those forces, have yet to be elucidated. To develop a better understanding of these processes, it is necessary to examine the mechanical behavior of the single chondrocyte. This paper reviews salient topics related to chondrocyte biomechanics and mechanotransduction, and attempts to put this information into a context both appropriate and useful to cartilage tissue engineering. It also describes the directions this exciting field is taking, and lays out a vision for future studies that could have a significant impact on our understanding of cartilage health and disease. © 2003 Biomedical Engineering Society.
Similar content being viewed by others
REFERENCES
Bachrach, N. M., W. B. Valhmu, E. Stazzone, A. Ratcliffe, W. M. Lai, and V. C. Mow. Changes in proteoglycan synthesis of chondrocytes in articular cartilage are associated with the timedependent changes in their mechanical environment. J. Biomech.28:15611569, 1995.
Bader, D. L., T. Ohashi, M. M. Knight, D. A. Lee, and M. Sato. Deformation properties of articular chondrocytes: A critique of three separate techniques. Biorheology39:6978, 2002.
Bonassar, L. J., A. J. Grodzinsky, E. H. Frank, S. G. Davila, N. R. Bhaktav, and S. B. Trippel. The effect of dynamic compression on the response of articular cartilage to insulinlike growth factor I. J. Orthop. Res.19:1117, 2001.
Bonassar, L. J., A. J. Grodzinsky, A. Srinivasan, S. G. Davila, and S. B. Trippel. Mechanical and physicochemical regulation of the action of insulinlike growth factor I on articular cartilage. Arch. Biochem. Biophys.379:5763, 2000.
Breuls, R. G., B. G. Sengers, C. W. Oomens, C. V. Bouten, and F. P. Baaijens. Predicting local cell deformations in engineered tissue constructs: a multilevel finite element approach. J. Biomech. Eng.124:198207, 2002.
Browning, J. A., R. E. Walker, A. C. Hall, and R. J. Wilkins. Modulation of Na+ × H+ exchange by hydrostatic pressure in isolated bovine articular chondrocytes. Acta Physiol. Scand.166:3945, 1999.
Buschmann, M. D., Y. A. Gluzband, A. J. Grodzinsky, and E. B. Hunziker. Mechanical compression modulates matrix biosynthesis in chondrocyte/agarose culture. J. Cell. Sci.108:14971508, 1995.
Buschmann, M. D., E. B. Hunziker, Y. J. Kim, and A. J. Grodzinsky. Altered aggrecan synthesis correlates with cell and nucleus structure in statically compressed cartilage. J. Cell. Sci.109:499508, 1996.
Buschmann, M. D., Y. J. Kim, M. Wong, E. Frank, E. B. Hunziker, and A. J. Grodzinsky. Stimulation of aggrecan synthesis in cartilage explants by cyclic loading is localized to regions of high interstitial fluid flow. Arch. Biochem. Biophys.366:17, 1999.
Carver, S. E., and C. A. Heath. Increasing extracellular matrix production in regenerating cartilage with intermittent physiological pressure. Biotechnol. Bioeng.62:166174, 1999.
Carver, S. E., and C. A. Heath. Influence of intermittent pressure, fluid flow, and mixing on the regenerative properties of articular chondrocytes. Biotechnol. Bioeng.65:274281, 1999.
D'Andrea, P., A. Calabrese, I. Capozzi, M. Grandolfo, R. Tonon, and F. Vittur. Intercellular Ca2+ waves in mechanically stimulated articular chondrocytes. Biorheology37:7583, 2000.
Durrant, L. A., C. W. Archer, M. Benjamin, and J. R. Ralphs. Organization of the chondrocyte cytoskeleton and its response to changing mechanical conditions in organ culture. J. Anat.194:343353, 1999.
Freeman, P. M., R. N. Natarajan, J. H. Kimura, and T. P. Andriacchi. Chondrocyte cells respond mechanically to compressive loads. J. Orthop. Res.12:311320, 1994.
Gooch, K. J., T. Blunk, D. L. Courter, A. L. Sieminski, P. M. Bursac, G. VunjakNovakovic, and L. E. Freed. IGFI and mechanical environment interact to modulate engineered cartilage development. Biochem. Biophys. Res. Commun.286:909915, 2001.
Gray, M. L., A. M. Pizzanelli, A. J. Grodzinsky, and R. C. Lee. Mechanical and physiochemical determinants of the chondrocyte biosynthetic response. J. Orthop. Res.6:777792, 1988.
Guilak, F.Compressioninduced changes in the shape and volume of the chondrocyte nucleus. J. Biomech.28:15291541, 1995.
Guilak, F., L. Alexopoulos, R. Nielsen, H. P. TingBeall, and M. Haider. The biomechanical properties of the chondrocyte pericellular matrix: micropipette aspiration of mechanically isolated chondrons. Trans. Orthop. Res. Soc.27:107, 2003.
Guilak, F., G. R. Erickson, and H. P. TingBeall. The effects of osmotic stress on the viscoelastic and physical properties of articular chondrocytes. Biophys. J.82:720727, 2002.
Guilak, F., W. R. Jones, H. P. TingBeall, and G. M. Lee. The deformation behavior and mechanical properties of chondrocytes in articular cartilage. Osteoarthritis Cartilage7:5970, 1999.
Guilak, F., and V. C. Mow. The mechanical environment of the chondrocyte: A biphasic finite element model of cellmatrix interactions in articular cartilage. J. Biomech.33:16631673, 2000.
Guilak, F., A. Ratcliffe, and V. C. Mow. Chondrocyte deformation and local tissue strain in articular cartilage: A confocal microscopy study. J. Orthop. Res.13:410421, 1995.
Guilak, F., M. Sato, C. M. Stanford, and R. A. Brand. Cell mechanics. J. Biomech.33:12, 2000.
Guilak, F., J. R. Tedrow, and R. Burgkart. Viscoelastic properties of the cell nucleus. Biochem. Biophys. Res. Commun.269:781786, 2000.
Haider, M. A., and F. Guilak. An axisymmetric boundary integral model for incompressible linear viscoelasticity: Application to the micropipette aspiration contact problem. J. Biomech. Eng.122:236244, 2000.
Hall, A. C.Differential effects of hydrostatic pressure on cation transport pathways of isolated articular chondrocytes. J. Cell Physiol.178:197204, 1999.
Hansen, U., M. Schunke, C. Domm, N. Ioannidis, J. Hassenpflug, T. Gehrke, and B. Kurz. Combination of reduced oxygen tension and intermittent hydrostatic pressure: A useful tool in articular cartilage tissue engineering. J. Biomech.34:941949, 2001.
Idowu, B. D., M. M. Knight, D. L. Bader, and D. A. Lee. Confocal analysis of cytoskeletal organization within isolated chondrocyte subpopulations cultured in agarose. Histochem. J.32:165174, 2000.
Jin, M., E. H. Frank, T. M. Quinn, E. B. Hunziker, and A. J. Grodzinsky. Tissue shear deformation stimulates proteoglycan and protein biosynthesis in bovine cartilage explants. Arch. Biochem. Biophys.395:4148, 2001.
Jones, W. R., H. P. TingBeall, G. M. Lee, S. S. Kelley, R. M. Hochmuth, and F. Guilak. Alterations in the Young's modulus and volumetric properties of chondrocytes isolated from normal and osteoarthritic human cartilage. J. Biomech.32:119127, 1999.
Jortikka, M. O., J. J. Parkkinen, R. I. Inkinen, J. Karner, H. T. Jarvelainen, L. O. Nelimarkka, M. I. Tammi, and M. J. Lammi. The role of microtubules in the regulation of proteoglycan synthesis in chondrocytes under hydrostatic pressure. Arch. Biochem. Biophys.374:172180, 2000.
Kim, Y. J., L. J. Bonassar, and A. J. Grodzinsky. The role of cartilage streaming potential, fluid flow, and pressure in the stimulation of chondrocyte biosynthesis during dynamic compression. J. Biomech.28:10551066, 1995.
Kim, Y. J., R. L. Sah, A. J. Grodzinsky, A. H. Plaas, and J. D. Sandy. Mechanical regulation of cartilage biosynthetic behavior: physical stimuli. Arch. Biochem. Biophys.311:112, 1994.
Knight, M. M., D. A. Lee, and D. L. Bader. The influence of elaborated pericellular matrix on the deformation of isolated articular chondrocytes cultured in agarose. Biochim. Biophys. Acta1405:6777, 1998.
Knight, M. M., J. M. Ross, A. F. Sherwin, D. A. Lee, D. L. Bader, and C. A. Poole. Chondrocyte deformation within mechanically and enzymatically extracted chondrons compressed in agarose. Biochim. Biophys. Acta1526:141146, 2001.
Knight, M. M., J. van de Breevaart Bravenboer, D. A. Lee, G. J. van Osch, H. Weinans, and D. L. Bader. Cell and nucleus deformation in compressed chondrocytealginate constructs: Temporal changes and calculation of cell modulus. Biochim. Biophys. Acta1570:18, 2002.
Koay, E. J., A. C. Shieh, and K. A. Athanasiou. Creep indentation of single cells. (in review).
Lai, W. M., V. C. Mow, D. D. Sun, and G. A. Ateshian. On the electric potentials inside a charged soft hydrated biological tissue: streaming potential versus diffusion potential. J. Biomech. Eng.122:336346, 2000.
Langelier, E., R. Suetterlin, C. D. Hoemann, U. Aebi, and M. D. Buschmann. The chondrocyte cytoskeleton in mature articular cartilage: Structure and distribution of actin, tubulin, and vimentin filaments. J. Histochem. Cytochem.48:13071320, 2000.
LeBaron, R. G., and K. A. Athanasiou. Extracellular matrix cell adhesion peptides: Functional applications in orthopedic materials. Tissue Eng.6:85103, 2000.
Lee, D. A., M. M. Knight, J. F. Bolton, B. D. Idowu, M. V. Kayser, and D. L. Bader. Chondrocyte deformation within compressed agarose constructs at the cellular and subcellular levels. J. Biomech.33:8195, 2000.
Lee, D. A., T. Noguchi, M. M. Knight, L. O'Donnell, G. Bentley, and D. L. Bader. Response of chondrocyte subpopulations cultured within unloaded and loaded agarose. J. Orthop. Res.16:726733, 1998.
Lee, H. S., S. J. MillwardSadler, M. O. Wright, G. Nuki, and D. M. Salter. Integrin and mechanosensitive ion channeldependent tyrosine phosphorylation of focal adhesion proteins and betacatenin in human articular chondrocytes after mechanical stimulation. J. Bone Miner. Res.15:15011509, 2000.
Mauck, R. L., M. A. Soltz, C. C. Wang, D. D. Wong, P. H. Chao, W. B. Valhmu, C. T. Hung, and G. A. Ateshian. Functional tissue engineering of articular cartilage through dynamic loading of chondrocyteseeded agarose gels. J. Biomech. Eng.122:252260, 2000.
MillwardSadler, S. J., M. O. Wright, L. W. Davies, G. Nuki, and D. M. Salter. Mechanotransduction via integrins and interleukin4 results in altered aggrecan and matrix metalloproteinase 3 gene expression in normal, but not osteoarthritic, human articular chondrocytes. Arthritis Rheum.43:20912099, 2000.
MillwardSadler, S. J., M. O. Wright, H. Lee, K. Nishida, H. Caldwell, G. Nuki, and D. M. Salter. Integrinregulated secretion of interleukin 4: A novel pathway of mechanotransduction in human articular chondrocytes. J. Cell Biol.145:183189, 1999.
Mow, V. C., S. C. Kuei, W. M. Lai, and C. G. Armstrong. Biphasic creep and stress relaxation of articular cartilage in compression. Theory and experiments. J. Biomech. Eng.102:7384, 1980.
Parkkinen, J. J., J. Ikonen, M. J. Lammi, J. Laakkonen, M. Tammi, and H. J. Helminen. Effects of cyclic hydrostatic pressure on proteoglycan synthesis in cultured chondrocytes and articular cartilage explants. Arch. Biochem. Biophys.300:458465, 1993.
Parkkinen, J. J., M. J. Lammi, H. J. Helminen, and M. Tammi. Local stimulation of proteoglycan synthesis in articular cartilage explants by dynamic compression. J. Orthop. Res.10:610620, 1992.
Sah, R. L., Y. J. Kim, J. Y. Doong, A. J. Grodzinsky, A. H. Plaas, and J. D. Sandy. Biosynthetic response of cartilage explants to dynamic compression. J. Orthop. Res.7:619636, 1989.
Salter, D. M., S. J. MillwardSadler, G. Nuki, and M. O. Wright. Integrininterleukin4 mechanotransduction pathways in human chondrocytes. Clin. Orthop. Relat. Res.391:S4960, 2001.
Sato, M., D. P. Theret, L. T. Wheeler, N. Ohshima, and R. M. Nerem. Application of the micropipette technique to the measurement of cultured porcine aortic endothelial cell viscoelastic properties. J. Biomech. Eng.112:263268, 1990.
Schinagl, R. M., D. Gurskis, R. M. Schinagl, A. C. Chen, and R. L. Sah. Depthdependent confined compression modulus of fullthickness bovine articular cartilage. J. Orthop. Res.15:499506, 1997.
Shin, D., and K. Athanasiou. Cytoindentation for obtaining cell biomechanical properties. J. Orthop. Res.17:880890, 1999.
Smith, R. L., B. S. Donlon, M. K. Gupta, M. Mohtai, P. Das, D. R. Carter, J. Cooke, G. Gibbons, N. Hutchinson, and D. J. Schurman. Effects of fluidinduced shear on articular chondrocyte morphology and metabolism. J. Orthop. Res.13:824831, 1995.
Smith, R. L., J. Lin, M. C. Trindade, J. Shida, G. Kajiyama, T. Vu, A. R. Hoffman, M. C. van der Meulen, S. B. Goodman, D. J. Schurman, and D. R. Carter. Timedependent effects of intermittent hydrostatic pressure on articular chondrocyte type II collagen and aggrecan mRNA expression. J. Rehabil. Res. Dev.37:153161, 2000.
Smith, R. L., S. F. Rusk, B. E. Ellison, P. Wessells, K. Tsuchiya, D. R. Carter, W. E. Caler, L. J. Sandell, and D. J. Schurman. stimulation of articular chondrocyte mRNA and extracellular matrix synthesis by hydrostatic pressure.J. Orthop. Res.14:5360, 1996.
Theret, D. P., M. J. Levesque, M. Sato, R. M. Nerem, and L. T. Wheeler. The application of a homogeneous halfspace model in the analysis of endothelial cell micropipette measurements. J. Biomech. Eng.110:190199, 1988.
Trickey, T. R., M. Lee, and T. Guilak. Viscoelastic properties of chondrocytes from normal and osteoarthritic human cartilage. J. Orthop. Res.18:891898, 2000.
VunjakNovakovic, G., I. Martin, B. Obradovic, S. Treppo, A. J. Grodzinsky, R. Langer, and L. E. Freed. Bioreactor cultivation conditions modulate the composition and mechanical properties of tissueengineered cartilage. J. Orthop. Res.17:130138, 1999.
Wang, C. C., X. E. Guo, D. Sun, V. C. Mow, G. A. Ateshian, and C. T. Hung. The functional environment of chondrocytes within cartilage subjected to compressive loading: A theoretical and experimental approach. Biorheology39:1125, 2002.
Wright, M. O., K. Nishida, C. Bavington, J. L. Godolphin, E. Dunne, S. Walmsley, P. Jobanputra, G. Nuki, and D. M. Salter. Hyperpolarization of cultured human chondrocytes following cyclical pressureinduced strain: Evidence of a role for alpha 5 beta 1 integrin as a chondrocyte mechanoreceptor. J. Orthop. Res.15:742747, 1997.
Wright, M. O., R. A. Stockwell, and G. Nuki. Response of plasma membrane to applied hydrostatic pressure in chondrocytes and fibroblasts. Connect. Tissue Res.28:4970, 1992.
Wu, J. Z., and W. Herzog. Finite element simulation of location and timedependent mechanical behavior of chondrocytes in unconfined compression tests. Ann. Biomed. Eng.28:318330, 2000.
Wu, J. Z., W. Herzog, and M. Epstein. Modeling of location and timedependent deformation of chondrocytes during cartilage loading. J. Biomech.32:563572, 1999.
Yellowley, C. E., C. R. Jacobs, Z. Li, Z. Zhou, and H. J. Donahue. Effects of fluid flow on intracellular calcium in bovine articular chondrocytes. Am. J. Physiol.273:C3036, 1997.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shieh, A.C., Athanasiou, K.A. Principles of Cell Mechanics for Cartilage Tissue Engineering. Annals of Biomedical Engineering 31, 1–11 (2003). https://doi.org/10.1114/1.1535415
Issue Date:
DOI: https://doi.org/10.1114/1.1535415