Abstract
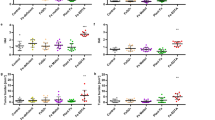
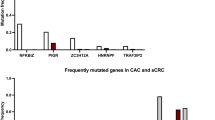
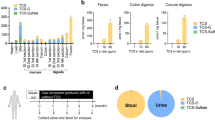
Individuals diagnosed with ulcerative colitis face a significantly increased risk of developing colorectal dysplasia and cancer during their lifetime. To date, little attention has been given to the development of a chemopreventive intervention for this high-risk population. The mouse model of dextran sulfate sodium (DSS) -induced colitis represents an excellent preclinical system in which to both characterize the molecular events required for tumor formation in the presence of inflammation and assess the ability of select agents to inhibit this process. Cyclic administration of DSS in drinking water results in the establishment of chronic colitis and the development of colorectal dysplasias and cancers with pathological features that resemble those of human colitis-associated neoplasia. The incidence and multiplicity of lesions observed varies depending on the mouse strain used (ie, Swiss Webster, C57BL/6J, CBA, ICR) and the dose (0.7%–5.0%) and schedule (1–15 cycles with or without a subsequent recovery period) of DSS. The incidence of neoplasia can be increased and its progression to invasive cancer accelerated significantly by administering DSS in combination with a known colon carcinogen (azoxymethane (AOM), 2-amino-3-methylimidazo[4,5-f]quinoline (IQ), 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP)) or iron. More recent induction of colitis-associated neoplasia in genetically defined mouse strains has provided new insight into the role of specific genes (ie, adenomatous polyposis coli (Apc), p53, inducible nitric oxide synthase (iNOS), Msh2) in the development of colitis-associated neoplasias. Emerging data from chemopreventive intervention studies document the efficacy of several agents in inhibiting DSS-induced neoplasia and provide great promise that colitis-associated colorectal neoplasia is a preventable disease.
Similar content being viewed by others
Article PDF
References
Eaden JA, Abrams KR, Mayberry JF . The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut 2001; 48: 526–35.
Hardy RG, Meltzer SJ, Jankowski JA . ABC of colorectal cancer. Molecular basis for risk factors. BMJ 2000; 321: 886–9.
Harpaz N, Talbot IC . Colorectal cancer in idiopathic inflammatory bowel disease. Sem Diagn Pathol 1996; 13: 339–57.
Ekbom A, Helmick C, Zack M, Adami HO . Ulcerative colitis and colorectal cancer. A population-based study. N Engl J Med 1990; 323: 1228–33.
Rutter MD, Saunders BP, Wilkinson KH, Rumbles S, Schofield G, Kamm MA, et al. Thirty-year analysis of a colonoscopic surveillance program for neoplasia in ulcerative colitis. Gastroenterology 2006; 130: 1030–8.
Farrell RJ, Peppercorn MA . Ulcerative colitis. Lancet 2002; 359: 331–40.
Kiesslich R, Fritsch J, Holtmann M, Koehler HH, Stolte M, Kanzler S, et al. Methylene blue-aided chromoendoscopy for the detection of intraepithelial neoplasia and colon cancer in ulcer-ative colitis. Gastroenterology 2003; 124: 880–8.
Kern SE, Redston M, Seymour AB, Caldas C, Powell SM, Kornacki S, et al. Molecular genetic profiles of colitis-associated neoplasms. Gastroenterology 1994; 107: 420–8.
Aust DE, Terdiman J P, Willenbucher R F, Chang CG, Molinaro-Clark A, Baretton GB, et al. The APC/β-catenin pathway in ulcerative colitis-related colorectal carcinomas. A mutational analysis. Cancer 2002; 94: 1421–7.
Hussain SP, Amstad P, Raja K, Ambs S, Nagashima M, Bennett WP, et al. Increased p53 mutation load in noncancerous colon tissue from ulcerative colitis: a cancer-prone chronic inflammatory disease. Cancer Res 2000; 60: 3333–7.
Brentnall TA, Crispin DA, Bronner M P, Cherian S P, Hueffed M, Rabinovitch PS, et al. Microsatellite instability in nonneoplastic mucosa from patients with chronic ulcerative colitis. Cancer Res 1996; 56: 1237–40.
Chang CL, Marra G, Chauhan DP, Ha HT, Chang DK, Ricciardiello L, et al. Oxidative stress inactivates the human DNA mismatch repair system. Am J Physiol Cell Physiol 2002; 283: C148–54.
Fogt F, Vortmeyer AO, Goldman H, Giordano TJ, Merino MJ, Zhuang Z . Comparison of genetic alterations in colonic adenoma and ulcerative colitis-associated dysplasia and carcinoma. Hum Pathol 1998; 29: 131–6.
Yashiro M, Carethers JM, Laghi L, Saito K, Slezak P, Jaramillo E, et al. Genetic pathways in the evolution of morphologically distinct colorectal neoplasms. Cancer Res 2001; 61: 2676–83.
Wong NACS, Harrison DJ . Colorectal neoplasia in ulcerative colitis—recent advances. Histopathology 2001; 39: 221–34.
Ohkusa T . Production of experimental ulcerative colitis in hamsters by dextran sulfate sodium and change in intestinal microflora. Jpn J Gastroenterol 1985; 82: 1327–36.
Okayasu I, Hatekayama S, Yamada M, Ohkusa T, Inagaki Y, Nakaya R . A novel method in the induction of reliable experimental acute and chronic ulcerative colitis in mice. Gastroenterology 1990; 98: 694–702.
Cooper HS, Murthy SNS, Shah RS, Sedergran DJ . Clinicopathologic study of dextran sulfate sodium experimental murine colitis. Lab Invest 1993; 69: 238–49.
Tanaka T, Kohno H, Suzuki R, Hata K, Sugie S, Niho N, et al. Dextran sodium sulfate strongly promotes colorectal carcinogenesis in ApcMin/+ mice: inflammatory stimuli by dextran sodium sulfate results in development of multiple colonic neoplasms. Int J Cancer 2006; 118: 25–34.
Cooper HS, Murthy S, Kido K, Yoshitake H, Flanigan A . Dysplasia and cancer in the dextran sulfate sodium mouse colitis model. Relevance to colitis-associated neoplasia in the human: a study of histopathology, B-catenin and p53 expression and the role of inflammation. Carcinogenesis 2000; 21: 757–68.
Nosho K, Yamamoto H, Adachi Y, Endo T, Hinoda Y, Imai K . Gene expression profiling of colorectal adenomas and early invasive carcinomas by cDNA array analysis. Br J Cancer 2005; 92: 1193–200.
Cooper HS, Everley L, Chang W-C, Pfeiffer G, Lee B, Murthy S, et al. The role of mutant Apc in the development of dysplasia and cancer in the mouse model of dextran sulfate sodium-induced colitis. Gastroenterology 2001; 121: 1407–16.
Coudry RA, Cooper HS, Gary M, Lubet RA, Chang WCL, Clapper ML . Correlation of inhibition of colitis-associated dysplasia by celecoxib with degree of inflammation in the mouse model of DSS-induced colitis. Proc Am Assoc Cancer Res 2004; 45: 548.
Tanaka T, Kohno H, Suzuki R, Yamada Y, Sugie S, Mori H . A novel inflammation-related mouse colon carcinogenesis model induced by azoxymethane and dextran sodium sulfate. Cancer Sci 2003; 94: 965–73.
Suzuki R, Kohno H, Sugie S, Nakagama H, Tanaka T . Strain differences in the susceptibility to azoxymethane and dextran sodium sulfate-induced colon carcinogenesis in mice. Carcinogenesis 2006; 27: 162–9.
Takahashi M, Nakatsugi S, Sugimura T, Wakabayashi K . Frequent mutations of the β-catenin gene in mouse colon tumors induced by azoxymethane. Carcinogenesis 2000; 21: 1117–20.
Tanaka T, Suzuki R, Kohno H, Sugie S, Takahashi M, Wakabayashi K . Colonic adenocarcinomas rapidly induced by the combined treatment with 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine and dextran sodium sulfate in male ICR mice possess β-catenin gene mutations and increases immunoreactivity for β-catenin, cyclooxygenase-2 and inducible nitric oxide synthase. Carcinogenesis 2005; 26: 229–38.
Aust DE, Terdiman JP, Willenbucher RF, Chew K, Ferrell L, Florendo C, et al. Altered distribution of β-catenin, and its binding proteins E-cadherin and APC, in ulcerative colitis-related colorectal cancers. Mod Pathol 2001; 14: 29–39.
Souza RF, Yin J, Smolinski KN, Zou TT, Wang S, Shi YQ, et al. Frequent mutation of the E2F-4 cell cycle gene in primary human gastrointestinal tumors. Cancer Res 1997; 57: 2350–3.
Sugimura T . Nutrition and dietary carcinogens. Carcinogenesis 2000; 21: 387–95.
Nagao M . A new approach to risk estimation of food-borne carcinogens—heterocyclic amines—based on molecular information. Mutat Res 1999; 431: 3–12.
Sugimura T . Overview of carcinogenic heterocyclic amines. Mutat Res 1997; 376: 211–9.
Nishikawa A, Imazawa T, Kuroiwa Y, Kitamura Y, Kanki K, Ishii Y, et al. Induction of colon tumors in C57BL/6J mice fed MeIQx, IQ, or PhIP followed by dextran sulfate sodium treatment. Toxicol Sci 2005; 84: 243–8.
Carrier J, Aghdassi E, Cullen J, Allard JP . Iron supplementation increases disease activity and vitamin E ameliorates the effect in rats with dextran sulfate sodium-induced colitis. J Nutr 2002; 132: 3146–50.
Seril DN, Liao J, Ho KL, Warsi A, Yang CS, Yang GY . Dietary iron supplementation enhances DSS-induced colitis and associated colorectal carcinoma development in mice. Dig Dis Sci 2002; 47: 1266–78.
Cooper HS, Chang WCL, Coudry R, Gary MA, Everley L, Spittle CS, et al. Generation of a unique strain of multiple intestinal neoplasia (Apc+/Min-FCCC) mice with significantly increased numbers of colorectal adenomas. Mol Carcinog 2005; 44: 31–41.
Burmer GC, Crispin DA, Kolli VR, Haggitt RC, Kulander BG, Rubin CE, et al. Frequent loss of a p53 allele in carcinomas and their precursors in ulcerative colitis. Cancer Commun 1991; 3: 167–72.
Chang WC, Coudry RA, Clapper ML, Zhang X, Williams KL, Spittle CS, et al. Loss of p53 enhances the induction of colitis-associated neoplasia by dextran sulfate sodium. Carcinogenesis 2007 June 8; [Epub ahead of print].
Fujii S, Fujimori T, Kawamata H, Takeda J, Kitajima K, Omotehara F, et al. Development of colonic neoplasia in p53 deficient mice with experimental colitis induced by dextran sulphate sodium. Gut 2004; 53: 710–6.
Wink DA, Vodovotz Y, Laval J, Laval F, Dewhirst MW, Mitchell JB . The multifaceted roles of nitric oxide in cancer. Carcinogenesis 1998; 19: 711–21.
Jaiswal M, LaRusso NF, Gores GJ . Nitric oxide in gastrointestinal epithelial cell carcinogenesis: linking inflammation to oncogenesis. Am J Physiol 2001; 281: G626–G34.
Seril DN, Liao J, Yang GY . Colorectal carcinoma development in inducible nitric oxide synthase-deficient mice with dextran sulfate sodium-induced ulcerative colitis. Mol Carcinog 2007; 46: 341–53.
Fleisher AS, Esteller M, Harpaz N, Leytin A, Rashid A, Xu Y, et al. Microsatellite instability in inflammatory bowel disease-associated neoplastic lesions is associated with hypermethylation and diminished expression of the DNA mismatch repair gene, hMLH1. Cancer Res 2000; 60: 4864–8.
Cawkwell L, Sutherland F, Murgatroyd H, Jarvis P, Gray S, Cross D, et al. Defective hMSH2/hMLH1 protein expression is seen infrequently in ulcerative colitis associated colorectal cancers. Gut 2000; 46: 367–9.
Kohonen-Corish MR, Daniel JJ, te Riele H, Buffinton GD, Dahlstrom JE . Susceptibility of Msh2-deficient mice to inflammation-associated colorectal tumors. Cancer Res 2002; 62: 2092–7.
Oshima M, Dinchuk JE, Kargman SL, Oshima H, Hancock B, Kwong E, et al. Suppression of intestinal polyposis in ApcΔ716 knockout mice by inhibition of cyclooxygenase 2 (COX-2). Cell 1996; 87: 803–9.
Steinbach G, Lynch P, Phillips RKS, Wallace MH, Hawk E, Gordon GB, et al. The effect of celecoxib, a cyclooxygenase-2 inhibitor, in familial adenomatous polyposis. N Engl J Med 2000; 342: 1946–52.
Mahadevan U, Loftus EV, Tremaine WJ, Sandborn WJ . Safety of selective cyclooxygenase-2 inhibitors in inflammatory bowel disease. Am J Gastroenterol 2002; 97: 910–4.
Kapicioglu S, Baki AH, Sari M, Ozdemir F, Kavgaci H . Does nimesulide induce gastric mucosal damage? “A double-blind randomized placebo-controlled trial”. Hepatogastroenterology 2000; 47: 1183–5.
Kohno H, Suzuki R, Sugie S, Tanaka T . Suppression of colitis-related mouse colon carcinogenesis by a COX-2 inhibitor and PPAR ligands. BMC Cancer 2005; 5: 46.
Niho N, Takahashi M, Kitamura T, Shoji Y, Itoh M, Noda T, et al. Concomitant suppression of hyperlipidemia and intestinal polyp formation in Apc-deficient mice by peroxisome proliferator-activated receptor ligands. Cancer Res 2003; 63: 6090–5.
Girnun GD, Smith WM, Drori S, Sarraf P, Mueller E, Eng C, et al. APC-dependent suppression of colon carcinogenesis by PPARγ. Proc Natl Acad Sci USA 2002; 99: 13771–6.
Lefebvre AM, Chen I, Desreumaux P, Najib J, Fruchart JC, Geboes K, et al. Activation of the peroxisome proliferator-activated receptor γ promotes the development of colon tumors in C57BL/6J-APCMin/+ mice. Nat Med 1998; 4: 1053–7.
Saez E, Tontonoz P, Nelson MC, Alvarez JG, Ming UT, Baird SM, et al. Activators of the nuclear receptor PPAR? enhance colon polyp formation. Nat Med 1998; 4: 1058–61.
Tanaka T, Kohno H, Yoshitani S, Takashima S, Okumura A, Murakami A, et al. Ligands for peroxisome proliferator-activated receptors α and γ inhibit chemically induced colitis and formation of aberrant crypt foci in rats. Cancer Res 2001; 61: 2424–8.
Gary MA, Coudry RA, Cooper HS, Litwin S, Concha AC, Lubet RA, et al. 5-Aminosalicylic acid inhibits colitis-associated dysplasia in the mouse model of AOM/DSS-induced colitis. Proc Am Assoc Cancer Res Third Annual International Conference on Frontiers in Cancer Prevention Research 2004; 45: A81.
Grimm GR, Dirisina R, Lee G, Tang Y, Manjali J, Hanson NB, et al. 5-ASA suppressed induction of dysplasia on the AOM/DSS model: Correlation with inhibition of Wnt/beta-catenin transcriptional activity. Gastroenterology 2005; 128 ( Suppl 2): A200.
Seril DN, Liao J, Ho KL, Yang CS, Yang GY . Inhibition of chronic ulcerative colitis-associated colorectal adenocarcinoma development in a murine model by N-acetylcysteine. Carcinogenesis 2002; 23: 993–1001.
Morris AJ, Malbon CC . Physiological regulation of G protein-linked signaling. Physiol Rev 1999; 79: 1373–430.
Goldstein JL, Brown MS . Regulation of low-density lipoprotein receptors: implications for pathogenesis and therapy of hyper-cholesterolemia and atherosclerosis. Circulation 1987; 76: 504–7.
Suzuki S, Tajima T, Sassa S, Kudo H, Okayasu I, Sakamoto S . Preventive effect of fluvastatin on ulcerative colitis-associated carcinogenesis in mice. Anticancer Res 2006; 26: 4223–8.
Raboy V . Myo-Inositol-1,2,3,4,5,6-hexakisphosphate. Phytochemistry 2003; 64: 1033–43.
Fox CH, Eberl M . Phytic acid (IP6), novel broad spectrum anti-neoplastic agent: a systematic review. Complement Ther Med 2002; 10: 229–34.
Liao J, Seril DN, Yang AL, Lu GG, Yang GY . Inhibition of chronic ulcerative colitis associated adenocarcinoma development in mice by inositol compounds. Carcinogenesis 2007; 28: 446–54.
Tanaka T, Kawabata K, Kakumoto M, Makita H, Hara A, Mori H, et al. Citrus auraptene inhibits chemically induced colonic aberrant crypt foci in male F344 rats. Carcinogenesis 1997; 18: 2155–61.
Kohno H, Suzuki R, Curini M, Epifano F, Maltese F, Gonzales SP, et al. Dietary administration with prenyloxycoumarins, auraptene and collinin, inhibits colitis-related colon carcinogenesis in mice. Int J Cancer 2006; 118: 2936–42.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by USPHS grants CN05121, CA099122, and CA06927 from the National Cancer Institute, an ACS Institutional Research Grant, and by an appropriation from the Commonwealth of Pennsylvania. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the National Cancer Institute or the American Cancer Society.
Rights and permissions
About this article
Cite this article
Clapper, M., Cooper, H. & Chang, WC. Dextran sulfate sodium-induced colitis-associated neoplasia: a promising model for the development of chemopreventive interventions. Acta Pharmacol Sin 28, 1450–1459 (2007). https://doi.org/10.1111/j.1745-7254.2007.00695.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00695.x
Keywords
This article is cited by
-
Using aspirin to prevent and treat cancer
Inflammopharmacology (2024)
-
Studies on the mechanism of local and extra-intestinal tissue manifestations in AOM-DSS-induced carcinogenesis in BALB/c mice: role of PARP-1, NLRP3, and autophagy
Naunyn-Schmiedeberg's Archives of Pharmacology (2023)
-
o-Aminoazotoluene, 7,12-dimethylbenz[a]anthracene, and N-ethyl-N-nitrosourea, which are mutagenic but not carcinogenic in the colon, rapidly induce colonic tumors in mice with dextran sulfate sodium-induced colitis
Genes and Environment (2022)
-
Green tea EGCG effectively alleviates experimental colitis in middle-aged male mice by attenuating multiple aspects of oxi-inflammatory stress and cell cycle deregulation
Biogerontology (2022)
-
Moderate DNA hypomethylation suppresses intestinal tumorigenesis by promoting caspase-3 expression and apoptosis
Oncogenesis (2021)