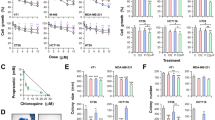
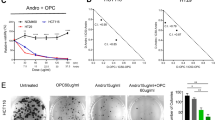
Abstract
In an effort to develop effective alternative strategies that increase the therapeutic efficacy and minimize the systemic toxicity of chemotherapeutic agents, more efforts are being directed towards the investigation of dietary supplements and other phytotherapeutic agents for their synergistic efficacy in combination with anticancer drugs. One such agent is silibinin, which has shown promising chemopreventive and anticancer effects in various in vitro and in vivo studies. The present review summarizes the effects of the combination of silibinin and chemotherapeutic drugs on the growth inhibition, cell cycle regulation, and apoptosis induction in prostate, breast, and lung cancer systems. Together, the results indicate a synergistic effect of silibinin on growth inhibition, reversal of chemoresistance, apoptosis induction, and a strong increase in G2–M checkpoint arrest when given in combination with these drugs. These results are highly significant with respect to the combined chemotherapy approach, wherein the criteria for combination is that the response has to be synergistic and that the drugs should not share common mechanisms of resistance and not overlap in their major side-effects.
Similar content being viewed by others
Article PDF
References
Hajdu SI . 2000 years of chemotherapy of tumors. Cancer 2005; 103: 1097–102.
Goodman LS, Gilman A, Brunton LL, Lazo JS, Parker KL . Goodman & Gilman's the pharmacological basis of therapeutics. 11th ed. New York: McGraw-Hill; 2006.
Cohen SM, Lippard SJ . Cisplatin: from DNA damage to cancer chemotherapy. Prog Nucleic Acid Res Mol Biol 2001; 67: 93–130.
Boulikas T, Vougiouka M . Cisplatin and platinum drugs at the molecular level. Oncol Rep 2003; 10: 1663–82.
Brabec V, Kasparkova J . Modifications of DNA by platinum complexes. Relation to resistance of tumors to platinum antitu-mor drugs. Drug Resist Updat 2005; 8: 131–46.
Binaschi M, Bigioni M, Cipollone A, Rossi C, Goso C, Maggi CA, et al. Anthracyclines: selected new developments. Curr Med Chem Anticancer Agents 2001; 1: 113–30.
Minotti G, Menna P, Salvatorelli E, Cairo G, Gianni L . Anthracy-clines: molecular advances and pharmacologic developments in antitumor activity and cardiotoxicity. Pharmacol Rev 2004; 56: 185–229.
Arola OJ, Saraste A, Pulkki K, Kallajoki M, Parvinen M, Voipio-Pulkki LM . Acute doxorubicin cardiotoxicity involves cardiomyo-cyte apoptosis. Cancer Res 2000; 60: 1789–92.
Petrylak DP, Tangen CM, Hussain MH, Lara PN Jr, Jones JA, Taplin ME, et al. Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N Engl J Med 2004; 351: 1513–20.
Tannock IF, de Wit R, Berry WR, Horti J, Pluzanska A, Chi KN, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 2004; 351: 1502–12.
Sherr CJ . Cell cycle control and cancer. Harvey Lect 2000; 96: 73–92.
Cooper GM, Hausman RE . The cell : a molecular approach. 3rd ed. Washington, D C: ASM Press, Sinauer Associates; 2004.
Vermeulen K, Van Bockstaele DR, Berneman ZN . The cell cycle: a review of regulation, deregulation and therapeutic targets in cancer. Cell Prolif 2003; 36: 131–49.
Paulovich AG, Toczyski DP, Hartwell LH . When checkpoints fail. Cell 1997; 88: 315–21.
Russell P . Checkpoints on the road to mitosis. Trends Biochem Sci 1998; 23: 399–402.
Vousden KH . Activation of the p53 tumor suppressor protein. Biochim Biophys Acta 2002; 1602: 47–59.
Sherr CJ, Roberts JM . CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev 1999; 13: 1501–12.
Genovese C, Trani D, Caputi M, Claudio PP . Cell cycle control and beyond: emerging roles for the retinoblastoma gene family. Oncogene 2006; 25: 5201–9.
DiPaola RS . To arrest or not to G(2)-M Cell-cycle arrest : commentary re: A K. Tyagi et al. Silibinin strongly synergizes human prostate carcinoma DU145 cells to doxorubicin-induced growth inhibition, G(2)-M arrest, and apoptosis. Clin cancer res, 8: 3512–3519, 2002. Clin Cancer Res 2002; 8: 3311–4.
Hirose Y, Berger MS, Pieper RO . Abrogation of the Chk1-medi-ated G(2) checkpoint pathway potentiates temozolomide-induced toxicity in a p53-independent manner in human glioblastoma cells. Cancer Res 2001; 61: 5843–9.
Shapiro GI, Supko JG, Patterson A, Lynch C, Lucca J, Zacarola PF, et al. A phase II trial of the cyclin-dependent kinase inhibitor flavopiridol in patients with previously untreated stage IV non-small cell lung cancer. Clin Cancer Res 2001; 7: 1590–9.
Devita VT, Hellman S, Rosenberg RA . Cancer: Principles and Practice of Oncology. 7th ed. Pliladelphia-PA: Lippincott Williams and Wilkins; 2005.
Kelloff GJ . Perspectives on cancer chemoprevention research and drug development. Adv Cancer Res 2000; 78: 199–334.
Sporn MB, Suh N . Chemoprevention of cancer. Carcinogenesis 2000; 21: 525–30.
Wellington K, Jarvis B . Silymarin: a review of its clinical properties in the management of hepatic disorders. BioDrugs 2001; 15: 465–89.
Kaur M, Agarwal R . Silymarin and epithelial cancer chemoprevention: How close we are to bedside? Toxicol Appl Pharmacol ( Published online Nov, 15th 2006).
Singh R P, Agarwal R . Prostate cancer prevention by silibinin. Curr Cancer Drug Targets 2004; 4: 1–11.
Singh R P, Agarwal R . Mechanisms of action of novel agents for prostate cancer chemoprevention. Endocr Relat Cancer 2006; 13: 751–78.
Singh R P, Agarwal R . Prostate cancer chemoprevention by silibinin: bench to bedside. Mol Carcinog 2006; 45: 436–42.
Vogel G, Trost W, Braatz R, Odenthal KP, Brusewitz G, Antweiler H, et al. Pharmacodynamics, site and mechanism of action of silymarin, the antihepatoxic principle from Silybum mar. (L) Gaertn. 1. Acute toxicology or tolerance, general and specific (liver-) pharmacology. Arzneimittelforschung 1975; 25: 82–9.
Mereish KA, Bunner DL, Ragland DR, Creasia DA . Protection against microcystin-LR-induced hepatotoxicity by Silymarin: biochemistry, histopathology, and lethality. Pharm Res 1991; 8: 273–7.
Zi X, Agarwal R . Silibinin decreases prostate-specific antigen with cell growth inhibition via G1 arrest, leading to differentiation of prostate carcinoma cells: implications for prostate cancer intervention. Proc Natl Acad Sci USA 1999; 96: 7490–5.
Zi X, Zhang J, Agarwal R, Pollak M . Silibinin up-regulates insulin-like growth factor-binding protein 3 expression and inhibits proliferation of androgen-independent prostate cancer cells. Cancer Res 2000; 60: 5617–20.
Deep G, Singh RP, Agarwal C, Kroll DJ, Agarwal R . Silymarin and silibinin cause G1 and G2-M cell cycle arrest via distinct circuitries in human prostate cancer PC3 cells: a comparison of flavanone silibinin with flavanolignan mixture silymarin. Oncogene 2006; 25: 1053–69.
Singh R P, Dhanalakshmi S, Tyagi AK, Chan DC, Agarwal C, Agarwal R . Dietary feeding of silibinin inhibits advance human prostate carcinoma growth in athymic nude mice and increases plasma insulin-like growth factor-binding protein-3 levels. Cancer Res 2002; 62: 3063–9.
Dhanalakshmi S, Agarwal P, Glode LM, Agarwal R . Silibinin sensitizes human prostate carcinoma DU145 cells to cisplatin-and carboplatin-induced growth inhibition and apoptotic death. Int J Cancer 2003; 106: 699–705.
Tyagi AK, Singh RP, Agarwal C, Chan DC, Agarwal R . Silibinin strongly synergizes human prostate carcinoma DU145 cells to doxorubicin-induced growth inhibition, G2-M arrest, and apoptosis. Clin Cancer Res 2002; 8: 3512–9.
Flaig T W, Su LJ, Harrison G, Agarwal R, Glode LM . Silibinin synergizes with mitoxantrone to inhibit cell growth and induce apoptosis in human prostate cancer cells. Int J Cancer 2007; 120: 2028–33.
Tyagi AK, Agarwal C, Chan DC, Agarwal R . Synergistic anti-cancer effects of silibinin with conventional cytotoxic agents doxorubicin, cisplatin and carboplatin against human breast carcinoma MCF-7 and MDA-MB468 cells. Oncol Rep 2004; 11: 493–9.
Sharma G, Singh RP, Chan DC, Agarwal R . Silibinin induces growth inhibition and apoptotic cell death in human lung carcinoma cells. Anticancer Res 2003; 23: 2649–55.
Singh R P, Mallikarjuna GU, Sharma G, Dhanalakshmi S, Tyagi AK, Chan DC, et al. Oral silibinin inhibits lung tumor growth in athymic nude mice and forms a novel chemocombination with doxorubicin targeting nuclear factor kappaB-mediated inducible chemoresistance. Clin Cancer Res 2004; 10: 8641–7.
Das KC, White CW . Activation of NF-kappaB by antineoplastic agents. Role of protein kinase C. J Biol Chem 1997; 272: 14914–20.
Wang CY, Cusack JC Jr, Liu R, Baldwin AS Jr . Control of induc-ible chemoresistance: enhanced anti-tumor therapy through increased apoptosis by inhibition of NF-kappaB. Nat Med 1999; 5: 412–7.
Dhanalakshmi S, Singh RP, Agarwal C, Agarwal R . Silibinin inhibits constitutive and TNFalpha-induced activation of NF-kappaB and sensitizes human prostate carcinoma DU145 cells to TNFalpha-induced apoptosis. Oncogene 2002; 21: 1759–67.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original studies in our silymarin/silibinin and cancer chemoprevention program are funded by NCI RO1 grants (No CA64514, CA102514, CA104286, CA112304, and CA113876), DOD prostate cancer program funding, and a NCI RO3 grant (CA99079).
Rights and permissions
About this article
Cite this article
Raina, K., Agarwal, R. Combinatorial strategies for cancer eradication by silibinin and cytotoxic agents: efficacy and mechanisms. Acta Pharmacol Sin 28, 1466–1475 (2007). https://doi.org/10.1111/j.1745-7254.2007.00691.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00691.x
Keywords
This article is cited by
-
Silibinin efficacy in a rat model of pulmonary arterial hypertension using monocrotaline and chronic hypoxia
Respiratory Research (2019)
-
Discovery of novel aromatase inhibitors using a homogeneous time-resolved fluorescence assay
Acta Pharmacologica Sinica (2014)
-
Inhibition of silibinin on migration and adhesion capacity of human highly metastatic breast cancer cell line, MDA-MB-231, by evaluation of β1-integrin and downstream molecules, Cdc42, Raf-1 and D4GDI
Medical Oncology (2012)
-
Stimulation of neoplastic mouse lung cell proliferation by alveolar macrophage-derived, insulin-like growth factor-1 can be blocked by inhibiting MEK and PI3K activation
Molecular Cancer (2011)