Abstract
The majority of new HIV infections occur in sub-Saharan Africa and other developing countries with scarce financial resources and meagre health system infrastructure. Expansion of Antiretroviral Therapy (ART) may overwhelm health services capacity, result in suboptimal care, and divert attention from crucial preventive measures. A 2009 WHO guideline recommends earlier initiation of ART for adults and adolescents, treatment at CD4 counts of 350 cells/mm3 along with universal testing. For sub-Saharan Africa, WHO previously recommended a threshold of 200 cells/mm3. Despite vast potential benefits of early ART initiation at individual and community levels, it does not necessary follow that clinical experience in industrialised countries can be replicated in resource-limited settings with moderate to high HIV burdens. Adherence to the 2009 WHO guidelines is unlikely to be sustainable without guarantees of adequate national and donor support – something all developing countries need to consider before adoption of the new policy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Most of the world's new HIV infections occur in sub-Saharan Africa, where financial resources, laboratory infrastructure, and health-care workers are scarce compared to higher income settings.
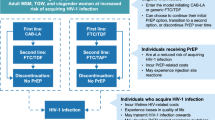
Because mathematical modelling showed that in communities with very high HIV prevalence, universal HIV testing and immediate Antiretroviral Therapy (ART) for everyone diagnosed with HIV could reduce new infections by 20 per cent within 10 years,1 WHO changed its guidelines in 2009. WHO's recommendation advanced initiation of treatment to a 350 cells/mm3 CD4 count. Current practice in low resource settings is 200 cells/mm3. Although the 2009 recommendations are intended to increase access to CD4 testing and use of viral load (VL) monitoring, ART use will not be denied where these monitoring tests are not available.2
What are the expected effects of early ART initiation on the immune system of individuals and then the impact at community level? Absence of large prospective trials gives rise to many uncertainties. The clinical experience underpinning the rationale for starting ART earlier, at higher CD4 counts, comes largely from industrialised countries and may not apply in resource-poor settings with a moderate to high HIV burden.
This policy shift may require a resource shift in the health system to offer ART, cutting back perhaps on voluntary counselling and testing (often called VCT) and on information, education, and communication elements.
As many research questions need to be addressed before implementation, it may be premature to assess the cost-effectiveness of the new policy. In moderate to high HIV settings, would the new guidance put public health objectives ahead of individual patient care?
Human immunodeficiency virus (HIV)
HIV causes acquired immunodeficiency syndrome (AIDS), a condition where the immune system begins to fail, permitting life-threatening opportunistic infections. HIV infection has an acute phase. The virus replicates rapidly immediately following exposure to HIV. Simultaneously CD4 cells decline temporarily, suggesting diminished immune capacity. A latency period of variable length follows, during which viral replication stabilises, as does the CD4 count. The final stage, AIDS, occurs when CD4+T-cell numbers decline below 200 cells per μL, a level below which an individual's immune system is too enfeebled to fight against opportunistic diseases. Death usually ensues.
Since the early 1990s, higher-income countries have been in the enviable position to offer ART to HIV-infected individuals. The first generation of ART drugs was toxic, prone to viral resistance, and poorly tolerated. With the new generation, HIV patients lead healthier, longer lives, making HIV a manageable, chronic disease. Current US and EU guidelines recommend treatment for patients with CD4+ counts lower than 350 cells/μL. These individuals are asymptomatic and their immune system continues to function.
Previous WHO guidelines for resource-poor settings advised ART be given only to symptomatic individuals, patients with severe immuno-suppression, or CD4 counts below 200 cells/mm3. At this lower CD4 level, many patients already had AIDS. The guideline was neither protective of individuals nor of the population.
Benefits
WHO's rationale in the new guidance for starting ART treatment at higher CD4 counts – while an HIV+ person remains asymptomatic – rests observational cohort studies plus theoretical assumptions.
Damage to the immune system
Because recovery is unlikely, early initiation of ART is thought to protect against immune system damage, as measured by CD4 cells. Maintaining higher CD4+ cell counts and preventing apparently irreversible immune system damage would, in principle, reduce AIDS-related morbidity and mortality.
No consensus exists on the ideal point to start ART, as no large prospective study has been completed. The UK Collaborative HIV Cohort Study found cumulative increase in CD4+ cell count to be dependent on the baseline CD4+ cell count. Only patients who initiated therapy at nadir CD4+ cell counts >350 cells/mm3 achieved eventual CD4+ cell counts similar – but not equal – to those observed in HIV-negative patients.3 The ART Cohort Collaboration found risks for AIDS and death significantly less when therapy was started at CD4 counts between 200 and 350 cells/mm3, as compared to CD4 counts no higher than 200 cells/mm.3, 4
Reduction of plasma VL
A correlation exists between the concentration of HIV particles in blood, called VL and CD4+ count. If VL is kept low, immune function is more likely preserved and disease progression delayed. Early ART not only reduces the VL and slows disease progression; treatment is thought to limit the size of the reservoir latent infected cells5 (cells in which the virus is present but not replicating, potentially a mechanism responsible for viral persistence). Patients treated in the first 3 weeks of infection may be able to marshal maximal HIV-specific immune responses to facilitate clearance of this reservoir.6, 7, 8
Intra-patient viral diversity
ART may become less effective owing to viral mutations that accumulate. Later initiation, at CD4 counts less than 350, may have allowed more mutations. HIV diversity is a direct consequence of replication rates, high mutation rates intrinsic to the virus, and selective pressures encountered within the host.9 Initiating ART sooner may restrain increased viral diversity, keeping the virus population more susceptible to drugs.
Reduction of opportunistic infections
Earlier initiation may reduce HIV-associated complications – tuberculosis (TB) and other opportunistic infections. In developing countries, latent TB infections are common. Without ART, HIV-infected patients with latent TB are very likely to develop active TB; perhaps 10 per cent per year, versus a 10 per cent lifetime risk for otherwise healthy persons with latent TB infections.10 Reactivation of TB may be directly related to loss of interferon-producing CD4 T-cells, as this cytokine is important to human host response against TB.11
Community-level Benefits
Biological effects of transmission
Early ART initiation affects transmission in important ways. VL predicts the risk of heterosexual transmission of HIV. Transmission is rare among persons with fewer than 1500 copies of HIV-1 RNA/ml.12 Thus universal HIV testing and early initiation of ART at the community level in sub-Saharan Africa, by greatly reducing the VL, might mitigate a severe generalised HIV epidemic.
HIV plasma VL rises transiently owing to immune stimulation from opportunistic infections that increase HIV transmission. In low-income countries, a systematic review measured plasma VL concentrations and the effect of treating co-infections. Mean plasma VL decreased after the treatment of co-infecting pathogens in all 18 studies.13 Starting ART at higher CD4 counts may be more effective than pathogen-specific interventions; it would also diminish HIV transmission.
Less TB in the community
Early ART initiation may lead to a diminished incidence of TB, while reducing HIV transmission in the community. A randomised trial in Abidjan, Ivory Coast, found early ART for HIV-infected adults diminished incidence of TB and other bacterial infections.14 In Tanzania, where the HIV/AIDS epidemic is associated with a 60 per cent increase in active TB,15 early ART may help TB control.
Psychological effects
If HIV is perceived as a manageable condition, this may encourage individuals at high risk to be re-tested. Similarly, targeting patients to embark earlier on ART may reduce new HIV infections, including those caused by mother-to-child transmission. Reduction of HIV-associated morbidity and mortality from opportunistic infections, especially TB – the leading cause of death related to HIV in sub-Saharan Africa – may also improve the quality of life and outlook of the community.
Economic effects
Beyond reducing negative microeconomic impacts on households, early ART initiation may increase productivity in certain economic sectors. It may, for example, increase availability of HIV-infected health professionals to work on AIDS prevention and treatment.
Uncertainties Facing Lower Resource Countries
Resource-poor countries face uncertainties about early ART initiation because new guidance is based on clinical experiences and studies in industrialised countries. These uncertainties warrant careful consideration before a country adopts of the 2009 WHO guidance.
Toxicity
The least expensive regimens used today in resource-rich settings were introduced in 1996, when an international panel recommended ART for all HIV-infected patients with a CD4+ count lower than 500 cells/mm.3, 16 Concerns about resistance,17 inadequate adherence,18 and toxic effects,19 however, commonly delayed initiation of treatment until later in the disease course.
Timing
The optimal time to begin therapy remains uncertain.20, 21 Observational data may be problematic, owing to our inability to control for all confounding factors. As it was not a randomised trial, the North American AIDS Cohort Collaboration on Research and Design study, despite its strengths, cannot be the basis for advice that everyone with HIV should start receiving ART. Patients in the study who began therapy early might have differed in important ways from those who deferred therapy.22
Toxicity due to longer use
Toxicities associated with long-term use of more toxic first generation drugs and with newer, more expensive, more potent, and better tolerated ARTs have not been quantified in large prospective randomised control trials. International clinical trials assessing when to start ART will not be available for several years. Trials of early ART initiation in South Africa may well demonstrate reduced morbidity and mortality, improved survival, and be cost-effective,23 but they address only short-term outcomes. They offer little insight into optimal timing in resource-limited settings.
Inflammatory syndrome (IRIS)
IRIS describes inflammatory disorders associated with paradoxical worsening of pre-existing infections in HIV-infected individuals following the initiation of ART.24 Unreliable diagnosis of TB, both latent and active, in HIV-infected individuals constitutes a special consideration for early ART initiation in countries with high endemic TB rates and moderate to high HIV burdens. Failure to treat latent TB infections can mean that ART treatment, via reconstitution, or the reversal of HIV-related immune system decline, makes it possible to transmit TB to others.25
Health systems in successful regimes
Successful clinical experiences, the basis for recommending early ART at higher CD4 counts, come largely from industrialised countries where specialised services are common: technology to assess CD4 cell counts and VL, monitor toxicity, conduct genotype resistance testing – along with many treatment options.
Regular CD4 count monitoring
Regular CD4 count monitoring means that ART can be started at a particular CD4 count, commonly 350. Tested every 3 months, patients not yet symptomatic, but at risk of progression are less likely to be missed. Many countries need to expand CD4 monitoring laboratory facilities, outlined in the 2009 WHO recommendations. But access to ART is not to be denied where monitoring tests are unavailable.
VL monitoring
VL monitoring assists in chronic HIV care management. Virological change can be followed only with VL monitoring. When unavailable, the decision of when to switch a patient from a first-line to a second-line treatment regimen must be based on changes in CD4 count or on WHO stage 4 clinical criteria. CD4 count changes correlate with VL across populations, but for individual patients they cannot be relied on to identify virological failure.26
Toxicity monitoring
If ART is started at higher CD4 counts and will be used for long periods, severe drug toxicities must be detected and followed, demanding expanded laboratory capacity: tests for Zidovudine (ZDV) haematoxicity and Nevirapine hepatoxicity, currently available in affluent countries.
Genotyping
Performing genotype resistance testing before starting treatment can help guide a physician's choice of regimen and increase patients’ quality-adjusted life expectancy.27 Where such genotyping is not available, patients may remain on failing regimens longer, often increasing viral drug resistance and compromising second-line drug options.
Limitations on drug options
In high-income countries, robust anti-drug-resistant protease inhibitors are available as first- or second-line drugs for newly treated, naive patients. Elsewhere drug options are limited. Treatment interruptions due to drug shortages or severe toxicity encourage drug resistance that facilitates HIV progression. Participants on CD4+ cell-guided episodic treatment face disease progression twice as rapid as those receiving continuous ART.28
Demands on Health Systems: Resource Poor, Moderate to High HIV Burden
Resource-poor settings with low HIV burdens face the clinical limitations described above. Countries with moderate to high HIV burdens face additional burdens and potential concerns.
Expansion of health-care personnel
ART initiation at CD4 counts of 350 require more staff, facilities, and comprehensive training in management of people living with AIDS – including training to predict when toxicity is likely and indicators for change in therapy.
Surveillance of HIV drug resistance
Drug resistance, low owing to limited ART use, may spread when a national treatment protocol adopts initiation at <350 CD4 accompanied by a scale-up of HIV testing. A greater portion of newly diagnosed patients may have drug resistant HIV, further limiting treatment options. At worst, ART would be ineffective. In recently infected persons, it may no longer be possible to rely on the minimum-resource strategy recommended by WHO to measure drug resistance at the population level.
Expanded use of VCT
Health-care restructuring may deplete resources allotted to VCT. Universal access to treatment requires universal knowledge of HIV status. In Tanzania, for example, only 10 per cent of people know their HIV status.29 To expand knowledge, both testing and counselling are needed.
Today management, called syndromic, depends on symptoms. If management is to begin with HIV+ but asymptomatic people who present with elevated CD4 cell counts, more services will be required in clinics.
Both TB and sexually transmitted diseases (STD) are closely linked to HIV prevalence. TB and STD programmes can be entry points for HIV counselling and testing. A study of STD management in primary care in the Mwanza region of Tanzania uncovered a 40 per cent reduction of HIV incidence.30 Limited health service utilisation in Africa may undermine strategies that depend on provider-initiated, routine HIV testing.31 Tanzania has experimented with community-based HIV testing – such as door-to-door testing – to find asymptomatic patients. Uganda found this strategy useful, particularly in communities with high HIV prevalence and low knowledge of HIV status; similar strategies are currently being rolled out in other sub-Saharan countries.32
Prevention activities and structural issues
HIV prevention activities are crucial. They may be short-changed by early ART initiation that divert resources. Stigma and discrimination against AIDS victims continue to discourage use of counselling and testing services. Thus efforts to scale up HIV testing must create an enabling atmosphere in which communities can recognise and address the social, cultural, and economic factors that put health in jeopardy, particularly the health of young women. A qualitative study in Tanzania revealed widespread public health benefits of ART in the presence of community-level interventions to ensure acceptability. Programmes must combat a general perception among community opinion leaders that as patients using ART regain health, they continue spreading the disease.33
Specific public knowledge needs
Specific information, education, and communication programmes may be needed to assist management of asymptomatic HIV patients. Countries with moderate to high HIV burdens can design programmes to accompany early ART initiation and prevent paradoxical increases in HIV incidence owing to increases in riskier behaviour during the infectious period. Behavioural surveillance can help assess the impact of ART availability on sexual behaviour. A national programme can also help establish a culture of repeat testing, avoiding a paradoxical increase of HIV incidence due to a false sense of protection.
Knowledge Gaps
Research should address several questions before implementation of the WHO guideline in which HIV disease burden is moderate, as in Tanzania.
Epidemiologic situation
Greater understanding of the epidemiological situation can help direct resources effectively, but, in the absence of knowledge of HIV status, this is impossible. Limited and understaffed health systems in sub-Saharan Africa make it unlikely that routine or mandatory testing per se would achieve drastic improvement in HIV/AIDS prevention or care.31 Will repeat testing of higher risk individuals, for example, be given priority over testing people considered to be at low risk for the first time?
Cost effectiveness
Without an understanding of the epidemiological situation it remains difficult to study cost effectiveness of programme elements, such as long-term CD4 and toxicity monitoring or to uncover failing regimens and resultant spread of primary drug resistance. Nevirapine, Stavudine, and Lamivudine, priced at approxmiately $150 USD a year, will be used for longer periods. Studies may uncover new toxicity and resistance. Will this lead to a switch to less toxic drugs like Efavirenz and Lopinavir priced up to $400 USD a year? Will better transport and logistics infrastructure be needed than was the case for first-line fixed dose combinations?
Lower immunity levels in developing countries
Immunological characteristics of HIV patients may need to be studied in the absence of data from prospective controlled studies. To what extent is the response to second-line therapy diminished in patients left for long periods on virologically failing regimens? Do lower immunity levels caused by poor nutritional status compromise early ART initiation's effectiveness and compound the risk of toxicity? As more children in moderate to high HIV burden settings receive treatment, what is the impact of long-term treatment toxicities on the health of these children?
TB clinical diagnostic algorithm
High TB endemicity overlaps with moderate to high HIV prevalence and TB is the leading cause of HIV-related deaths. A clinical diagnostic algorithm that reflects local conditions would help establish decision rules for treatment with Isoniazid of latent TB infections in HIV patients. At what CD4 count should HIV patients, who are tuberculin positive or have past TB history, start Isoniazid?
Risk factors for non-adherence
Adhering to life-long therapy is problematic for asymptomatic patients. What contributes to non-adherence? Are there cost effective mechanisms to enhance adherence?
GLOBAL ANALYTIC APPROACH
Continued political support
Universal HIV testing and starting ART at a CD4 count of 350 cell/mm requires a firm long-term political commitment globally. But is political commitment waning, set off by the global recessions and a growing sense that more lives would be saved by fighting other diseases?34 The Obama administration has, for example, focused on the new Global Health Initiative on Mother-and-child Health.
Migration of health workers
Industrialised countries that recruit health workers from sub-Saharan Africa could design and implement immediate measures to stem migration of badly needed health workers. Cooperation between sub-Saharan and industrial country governments can offer some relief, as out-migration undermines HIV prevention, treatment, and care.
CONCLUSION
What can we conclude about the new 2009 WHO guidance to improve health, reduce infections, and save lives by moving treatment initiation to 350 cells/mm3 from 200 cells/mm3, along with universal HIV testing for sub-Saharan Africa?
Too soon to assess cost effectiveness
Striving for every African adult and adolescent to know his or her HIV status, be re-tested in case of potential exposure,35 and receive immediate ART treatment are laudable goals. But are they feasible? Providing ART coverage to HIV patients at a 200 CD4 count already represents a challenge to health systems in sub-Saharan Africa. Very few asymptomatic patients present themselves for counselling and testing. Thus it may be premature to assess the cost-effectiveness of a policy change. Potential benefits should be weighed against potential risks of treatment-associated toxicity, drug resistance that decreases future treatment options, and transmission of resistant viral strains within the community.
HIV testing scale-up and ART in low resource countries
As low-resource countries scale-up HIV testing and they find more asymptomatic patients eligible for ART, we may expect to find greater stresses within health programmes and diversion of resources away from treatment of symptomatic HIV patients. The pain is likely to be worst in high HIV burden countries like Botswana and in high prevalence settings within moderate burden countries, such as the Iringa and Mwanza districts in Tanzania, rather than where HIV prevalence is low, as in Morocco.
Before implementing the new WHO guidance, less affluent countries with moderate to high HIV burdens must build an internal consensus to ensure that health system strengthening remains a priority.
Public health system losing authority
Because the old WHO guidance that prescribed treatment at a CD4 level of 200 was rarely achieved, will the new guidance have a substantial impact? Or will it result in the public health system losing its authority to deny treatment to anyone whose CD4 count is above the threshold?
Ethical concerns
An epidemic may make a compelling case for compromising ethical concerns. Implementing the 2009 WHO guidance in the face of suboptimal laboratory infrastructure may target one subgroup and divert resources from other HIV prevention, and from counselling and testing. Should countries expend resources on treating fewer people optimally or try to reach all people with more advanced HIV? Medical interventions to achieve population benefits rather than individual patient benefits might reflect public health at its best and its worst.36 It might fail to incorporate a human rights perspective focused on individuals while failing to achieve population goals. Surely, strides in public health may not be sustainable if both national commitment and donor support cannot be guaranteed.
References
Granich, R. et al (2009) Universal voluntary HIV testing with immediate antiretroviral therapy as a strategy for elimination of HIV transmission: A mathematical model. The Lancet 373 (9657): 48–57.
WHO. (2009) New HIV recommendations to improve health, reduce infections and save lives, http://www.who.int/mediacentre/news/releases/2009/world_aids_20091130/en/index.html, accessed 20 July 2010.
Hughes, R., Sabin, C. and Sterne, J. (2007) Long-term trends in CD4 count in patients starting HAART: UK-CHIC study [Abstract P18.4/04]. 11th European AIDS Conference, Madrid, http://www.aidsmeds.com/articles/hiv_cd4_uk_2094_13356.shtml, accessed 25 October 2009.
May, M. et al (2007) Prognosis of HIV-1-infected patients up to 5 years after initiation of HAART: Collaborative analysis of prospective studies. AIDS 21 (9): 1185–1197.
Lori, F. et al (1999) Treatment of human immunodeficiency virus infection with hydroxyurea, didanosine, and a protease inhibitor before seroconversion is associated with normalized immune parameters and limited viral reservoir. Journal of Infectious Diseases 180 (6): 1827–1832.
Rosenberg, E.S. et al (2000) Immune control of HIV-1 after early treatment of acute infection. Nature 407 (6803): 523–526.
Oxenius, A. et al (2000) Early highly active antiretroviral therapy for acute HIV-1 infection preserves immune function of CD8+ and CD4+ T lymphocytes. Proceedings National Academy of Science of USA, 28 March 2000; 97(7), pp. 3382–3387. http://www.ncbi.nlm.nih.gov/pubmed/10737796, accessed 25 October 2009.
Malhotra, U. et al (2000) Effect of combination antiretroviral therapy on T-cell immunity in acute human immunodeficiency virus type 1 infection. Journal of Infectious Diseases 181 (1): 121–131.
Gottlieb, G.S. (2003) Early HIV-1 viral diversity associated with rapid disease progression. NLM Gateway Abstract 10th Conference Retroviral Opportunistic Infections Feb 10 http://gateway.nlm.nih.gov/MeetingAbstracts/ma?f=102261104.html, accessed 1 November 2009.
Selwyn, P.A., Hartel, D. and Lewis, V.A. et al (1989) A prospective study of the risk of tuberculosis among intravenous drug users with human immunodeficiency virus infection. New England Journal of Medicine 320: 545–550.
Schluger, N.W. and Rom, W.N. (1998) The host immune response to tuberculosis. American Journal of Respiratory and Critical Care Medicine 157 (3): 679–691.
Quinn, T.C. et al (2000) Viral load and heterosexual transmission of human immunodeficiency virus type 1. New England Journal of Medicine 342 (13): 921–929.
Modjarrad, K. and Vermund, S.H. (2010) Effect of treating co-infections on HIV-1 viral load: A systematic review. The Lancet Infectious Diseases 10 (7): 455–463.
Moh, R. (2007) Incidence and determinants of mortality and morbidity following early antiretroviral therapy initiation in HIV-infected adults in West Africa. AIDS 21 (18): 2483–2491.
USAID Tanzania. (2008) http://www.usaid.gov/our_work/global_health/id/tuberculosis/countries/africa/tanzania_profile.htm, accessed 25 October 2009.
Carpenter, C.C., Fischl, M.A. and Hammer, S.M. et al (1996) Antiretroviral therapy for HIV infection in 1996: Recommendations of an international panel. Journal of the American Medical Association 276 (2): 146–154.
Richman, D.D., Morton, S.C. and Wrin, T. et al (2004) The prevalence of antiretroviral drug resistance in the United States. AIDS 18 (10): 1393–1401.
Paterson, D.L., Swindells, S. and Mohr, J. et al (2000) Adherence to protease inhibitor therapy and outcomes in patients with HIV infection. Annals of Internal Medicine 133 (1): 21–30.
Carr, A., Samaras, K. and Burton, S. et al (1998) A syndrome of peripheral lipodystrophy, hyperlipidaemia and insulin resistance in patients receiving HIV protease inhibitors. AIDS 12 (7): F51–F58.
AIDS Info. (2008) Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents, http://www.aidsinfo.nih.gov/Guidelines/Default.aspx?MenuItem=Guideline, accessed 25 October 2009.
Hammer, S.M., Eron Jr, J.J. and Reiss, P. et al (2008) Antiretroviral treatment of adult HIV infection: 2008 Recommendations of the International AIDS Society-USA panel. Journal of the American Medical Association 300 (5): 555–570.
Sax, P.E. and Baden, L.R. (2009) When to start antiretroviral therapy – Ready when you are? New England Journal of Medicine 360 (18): 1897–1899.
Walensky, R.P. and Wolf, L.L. et al (2009) When to start antiretroviral therapy in resource-poor settings. Annals of Internal Medicine 151 (3): 157–166.
Shelburne, S.A., Montes, M. and Hamill, R.J. (2006) Immune reconstitution inflammatory syndrome: More answers, more questions. Journal of Antimicrobial Chemotherapy 57 (2): 167–170.
CDC NIH HIVMA/IDSA. (2008) Guidelines for prevention and treatment of opportunistic infections in HIV-infected adults and adolescents, http://www.aidsinfo.nih.gov/contentfiles/Adult_OI000999.pdf, accessed 12 December 2009.
Badri, M., Lawn, S. and Wood, R. (2008) Utility of CD4 cell counts for early prediction of virological failure during antiretroviral therapy in a resource-limited setting. BMC Infectious Diseases 8: 89.
Bio Medicine. (2005) Drug resistance testing in treatment naive HIV is cost effective, http://news.bio-medicine.org/medicine-news-3/Drug-resistance-testing-in-treatment-nave-HIV-patients-is-cost-effective-8520-1, accessed 17 November 2009.
El-Sadr, W.M. and Lundgren, J.D. et al (2006) The strategies for management of ART therapy (SMART) study group. CDR+ count-guided interruption of antiretroviral, http://www.nejm.org/doi/pdf/10.1056/NEJMoa062360, accessed 2 February 2009.
AMREF UK. (2009) HIV voluntary counselling and testing. Tanzania: http://uk.amref.org/what-we-do/hiv-voluntary-counselling-and-testing-tanzania, accessed 7 February 2009.
Tanzania Commission for HIV/AIDS. (2003) National multi-sectoral strategic framework on HIV/AIDS framework on HIV/AIDS 2003–2007 http://www.tanzania.go.tz/pdf/tacaidsnmf.pdf, accessed 6 February 6, 2009.
Asante, A.D. (2007) Scaling up HIV prevention: Why routine or mandatory testing is not feasible for sub-Saharan Africa. Bulletin World Health Organization 85 (8): 644–646.
Leach-Lemens, C. and Owuor, J. (2009) Scaling up HIV testing and counselling towards universal access: what works in resource-limited settings? AIDSMAP http://www.aidsmap.com/cms1330557.aspx, accessed 2 February 2010.
Roura, M., Urassa, M., Bsza, J., Mbata, D., Wringe, A. and Zaba, B. (2008) Scaling up stigma? The effects of ART roll-out on stigma and HIV testing. Early evidence from rural Tanzania. Sexually Transmitted Infections 85 (4): 308–312.
McNeil, D.G. (2010) At front lines, AIDS war is falling apart. New York Times, 9 May.
De Cock, K.M. et al (2003) A serostatus approach to HIV/AIDS prevention and care in Africa. The Lancet 362: 1847–1849.
Garnett, G. and Baggeley, R.F. (2008) Treating our way out of the HIV pandemic: Could we, would we, should we? The Lancet 373 (2009): 9–11.
Author information
Authors and Affiliations
Additional information
The author questions aspects of a 2009 WHO guideline recommending earlier initiation of antiretroviral therapy (ART) along with universal testing and suggests what all developing countries need to consider prior to adoption of the new policy.
Rights and permissions
About this article
Cite this article
Menon, S. Early initiation of antiretroviral therapy and universal HIV testing in sub-Saharan Africa: Has WHO offered a milestone for HIV prevention?. J Public Health Pol 31, 385–400 (2010). https://doi.org/10.1057/jphp.2010.29
Published:
Issue Date:
DOI: https://doi.org/10.1057/jphp.2010.29