Abstract
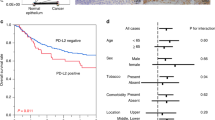
CDC25A, CDC25B and CDC25C belong to a family of protein phosphatases which activate the cyclin-dependent kinase at different points of the cell cycle. According to accumulating evidence, CDC25A and CDC25B seem to possess oncogenic properties. We have analysed these expressions by immunohistochemistry, western blot and RT-PCR in a series of 100 patients with squamous cell carcinoma of the oesophagus. When compared with non-cancerous cells, CDC25A and CDC25B were strongly expressed in the cytoplasm of cancer cells, with positive (+) classification in 46% (46 cases) and 48% (48 cases), respectively. There was no significant correlation between CDC25A and CDC25B expression, nor was there any association with the expression of other cell cycle-regulating molecules, including cyclin D1, Rb, p16INK4, p27KIP1and PCNA (proliferating cell nuclear antigen). CDC25A (+), as well as CDC25B (+), was more frequently found in patients with deeper tumour invasion and lymph node metastasis, while tumour size was correlated only with CDC25A expression. Postoperative survival was significantly poorer for CDC25A (+) patients than CDC25A (–) patients, but was not affected by the CDC25B status. Nuclear localization of CDC25A was observed in 51 cases (51%), regardless of its cytoplasmic expression, and was not associated with clinico-pathological factors or prognosis. Multivariate analysis revealed only the CDC25A status to be an independent significant prognostic factor among these biological and clinico-pathological factors. CDC25A but not CDC25B may be a new prognostic factor for squamous cell carcinoma of the oesophagus. Thus, regulation of the G1 checkpoint in the cell cycle may be important in oesophageal carcinogenesis, which may also involve many other oncogenes. © 2001 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Chen X and Prywes R (1999) Serum-induced expression of cdc25A gene by relief of E2F-mediated repression. Mol Cell Biol 19: 4695–4702
Chomczynski P and Sacchi N (1987) Single-step method RNA isolation by acid guanidiniumthiocyanate-phenol-chloroform extraction. Anal Biochem 162: 156–159
Chretien S, Dubart A, Beaupain D, Raich N, Grandchamp B, Rosa J, Goossens M and Romeo PH (1988) Alternative transcription and splicing of the human porphobilinogen deaminase gene result either in tissue-specific or in housekeeping expression. Proc Natl Acad Sci USA 85: 6–10
Ciaparrone M, Yamamoto H, Yao Y, Sgambato A, Cattoretti G, Tomita N, Monden M, Rotterdam H and Weinstein I (1998) Localization and expression of p27KIP1 in multistage colorectal carcinogenesis. Cancer Res 58: 114–122
Coco Martin JM, Balkenende A, Verschoo T, Lallemand F and Michalides R (1999) Cyclin D1 overexpression enhances radiation-induced apoptosis and radiosensitivity in a breast tumor cell line. Cancer Res 59: 1134–1140
Dixon D, Moyana T and King MJ (1998) Elevated expression of the cdc25A protein phosphatase in colon cancer. Exp-Cell-Res 240: 236–243
Fukuoka K, Shindoh M, Yamashita T, Fujinaga K, Amemia A and Totsuka Y (1996) High-risk HPV-positive human cancer cell lines show different sensitivity to cisplatin-induced apoptosis corelated with the p21Waf/Cip1 level. Cancer Lett 108: 15–23
Galaktionov K and Beach D (1991) Specific activation of cdc25 tyrosine phosphatases by B-type cyclins: evidence for multiple roles of mitotic cyclins. Cell 67: 1181–1194
Galaktionov K, Lee AK, Eckstein J, Draetta G, Meckler J, Loda M and Beach D (1995) CDC25 phosphatases as potential human oncogenes. Science 269: 1575–1577
Galaktionov K, Chen X and Beach D (1996) Cdc25 cell-cycle phosphatase as a target of c-myc. Nature (Lond) 382: 511–517
Gabrielli BG, De Souza CP, Tonks ID, Clark JM, Hayward NK and Ellem KA (1996) Cytoplasmic accumulation of cdc25B phosphatase in mitosis triggerrs centrosomal microtubule nucleation in HeLa cells. J Cell Sci 109: 1081–1093
Gasparotto D, Maestro R, Piccinin S, Vukosavljevic T, Barzan L, Sulfaro S and Boiocchi M (1997) Overexpression of CDC25A and CDC25B in head and neck cancers. Cancer Res 57: 2366–2368
Hernandez S, Hernandez L, Bea S, Cazorla M, Fernandez PL, Nadal A, Muntane J, Mallofre C, Montserrat E, Cardesa A and Campo E (1998) cdc25 cell cycle-activating phosphatases and c-myc expression in human non-Hodgikin lymphomas. Cancer Res 58: 1762–1767
Hunter T and Pines J (1994) Cyclins and cancer. II: Cyclin D and CDK inhibitors come of age. Cell 79: 573–582
Kang SH, Bang YJ, Jong HS, Kim NK and Kim SJ (1999) Rapid induction of p21 Waf1 but delayed down regulation of cdc25A in the TGF-beta-induced cell cycle arrest of gastric carcinoma cells. Br J Cancer 80: 1144–1149
Karlsson C, Katich S, Hagting A, Hoffmann I and Pines J (1999) Cdc25B and cdc25C differ markedly in their properties as initiators mitosis. J Cell Biol 3: 573–584
Kokunai T and Tamaki N (1999) Relationship between expression of p21Waf/Cip1 and radioresistance in human gliomas. Jpn J Cancer Res 90: 638–646
Kudo Y, Yasui W, Ue T, Yamamoto S, Yokozaki H, Nikai H and Tahara E (1997) Overexpression of cyclin-dependent kinase-activating cdc25B phosphatase in human gastric carcinomas. Jpn J Cancer Res 88: 947–952
Lu SH, Hsieh LL, Luo FC and Weinstein IB (1988) Amplification of the EGF receptor and c-myc genes in human esophageal cancers. Int J Cancer 42: 502–505
Miyata H, Doki Y, Hitoshi S, Inoue M, Yano M, Fujiwara Y, Yamamoto H, Nishioka K, Kentaro K and Monden M (2000) CDC25B and p53 are independently implicated in radiation sensitivity for human esophageal cancers. Clin Cancer Res 6: 4859–4865
Nagata A, Igarashi M, Junno S, Suto K and Okayama H (1991) An additional homolog of fission yeast cdc25+ gene occurs in humans and is highly expressed in some cancer cells. New Biol 3: 959–968
Nagel S, Schmidt S, Thiede C, Huhn D and Neubauer A (1996) Quantification of Bcr-Abl transcripts in chronic myelogenous leukemia (CML) using standardized, internally controlled, competitive differential PCR(CD-PCR). Nucleic Acids Res 24: 4102–4103
Okami J, Yamamoto H, Fujiwara Y, Tsujie M, Kondo M, Noura S, Oshima S, Nagano H, Dono K, Umeshita K, Ishikawa O, Sakon M, Matsuura N and Monden M (1999) Overexpression of cyclooxygenase-2 in carcinoma of the pancreas. Clin Cancer Res 5: 2018–2024
Pines J (1995) Cyclins and cyclin-dependent kinasees: theme and variations. Adv Cancer Res 66: 181–212
Sadhu K, Reed SI, Richardson H and Russell P (1990) Human homolog of fission yeast cdc25 mitotic inducer is predominantly expressed in G2. Proc Natl Acad Sci USA 87: 5139–5143
Saha P, Eichbaum Q, Silberman ED, Mayer B and Dutta A (1997) p21CIP1and Cdc25A: Competition between an Inhibitor and an Activator of Cyclin-Dependent Kinases. Mol Cell Biol 17: 4338–4345
Shamma A, Doki Y, Shiozaki H, Tsujinaka T, Inoue M, Yano M, Kimura Y, Yamamoto M and Monden M (1998) Effect of cyclin D1 and associated proteins on proliferation of esophageal squamous cell carcinoma. Int J Oncology 13: 455–460
Sherr CJ (1994) Mammalian G1 cyclins. Cell 79: 551–555
Takemasa I, Yamamoto H, Tomita N, Sekimoto M, Ohue M, Noura S, Miyake Y, Aihara T, Tamaki Y, Sakita I, Matsuura N and Monden M (2000) Assessment for increased expression of cdc25B phosphatase as a novel prognostic marker in human colorectal carcinomas. Cancer Res 60: 3043–3050
TNM classification by UICC,
Warenius HM, Seabra LA and Maw P (1996) Sensitivity to cis- biamminedichloroplatinum in human cancer cells is related to expression of cyclin D1 but not c-raf-1 protein. Int J Cancer 67: 224–231
Wu W, Fan YH, Kemp BL, Walsh G and Mao L (1998) Overexpression of cdc25A and cdc25B is frequent in primary non-small cell lung cancer but is not associated with overexpression of c-myc. Cancer Res 58: 4082–4085
Yamamoto H, Soh J-W, Shirin H, Xing W-Q, Lim JT, Yao Y, Slosberg E, Tomita N, Schieren I and Weinstein IB (1999) Comparative effects of overexpression of p27KIP1 and p21CIP1/WAFT on growth and differentiation in human colon carcinoma cells. Oncogene 18: 103–115
Yao Y, Slosberg E, Wang L, Hibshoosh H, Zhang Y, Xing W and Weinstein B (1999) Increased susceptibility to carcinogen-induced mammary tumors in MMTV-Cdc25B transgenic mice. Oncogene 18: 5159–5166
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Nishioka, K., Doki, Y., Shiozaki, H. et al. Clinical significance of CDC25A and CDC25B expression in squamous cell carcinomas of the oesophagus. Br J Cancer 85, 412–421 (2001). https://doi.org/10.1054/bjoc.2001.1934
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2001.1934
Keywords
This article is cited by
-
Lung squamous cell carcinoma and lung adenocarcinoma differential gene expression regulation through pathways of Notch, Hedgehog, Wnt, and ErbB signalling
Scientific Reports (2020)
-
Phosphatases and kinases regulating CDC25 activity in the cell cycle: clinical implications of CDC25 overexpression and potential treatment strategies
Molecular and Cellular Biochemistry (2016)
-
The regulatory roles of phosphatases in cancer
Oncogene (2014)
-
Part II—mechanism of adaptation: A549 cells adapt to high concentration of nitric oxide through bypass of cell cycle checkpoints
Tumor Biology (2014)
-
Systematic review and meta-analysis of tumor biomarkers in predicting prognosis in esophageal cancer
BMC Cancer (2013)