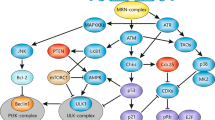
Abstract
Bleomycin is a cytotoxic antibiotic that generates DNA double-strand breaks (DSB) and DNA single-strand breaks (SSB). It is possible to introduce known quantities of bleomycin molecules into cells. Low amounts kill the cells by a slow process termed mitotic cell death, while high amounts produce a fast process that has been termed pseudoapoptosis. We previously showed that these types of cell death are a direct consequence of the DSB generated by bleomycin. Here, we use deglyco-bleomycin, a bleomycin derivative lacking the carbohydrate moiety. Although this molecule performs the same nucleophilic attacks on DNA as bleomycin, we show that deglyco-bleomycin is at least 100 times less toxic to Chinese hamster fibroblasts than bleomycin. In fact, deglyco-bleomycin treatment results in apoptosis induction. In contrast, however, deglyco-bleomycin was found to generate almost exclusively SSB. Our results suggest that more than 150 000 SSB per cell are required to trigger apoptosis in Chinese hamster fibroblasts and that SSB are 300 times less toxic than DSB. Taken together with previous studies on bleomycin, our data demonstrates that cells can die by apoptosis, mitotic cell death, or pseudoapoptosis, depending on the number of DNA breaks and on the ratio of SSB to DSB. © 2001 Cancer Research Campaign http://www.bjcancer.com
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Bailly C, Kenani A and Waring MJ (1995) Analogue versus digital recognition of DNA by bleomycin: an effect of the carbohydrate moiety. FEBS Lett 372: 144–147
Cohen-Jonathan E, Bernhard EJ and McKenna WG (1999) How does radiation kill the cells? Curr Opin Chem Biol 3: 77–83
Cole A, Shonka F, Corry PM and Cooper WG (1975) CHO cell repair of single strand and double strand breaks induced by gamma and alpha radiations. Molecular mechanisms for the repair of DNA, II
Cole A, Meyn RE, Chen R, Corry PM and Hillelman W (1980) Mechanisms of cell injury. Meyn RE, Withers HR (eds), Radiation Biology in Cancer Research, pp. 33–58, Raven Press Ltd: New York
Cullinan EB, Gawron LS, Rustum YM and Beerman TA (1991) Extrachromosomal chromatin: Novel target for bleomycin cleavage in cells and solid tumors. Biochemistry 30: 3055–3061
Foray N, Fertil B, Alsbeih MGA, Badie C, Chavaudra N, Iliakis G and Malaise EP (1996) Dose-rate effect on radiation-induced DNA double strand breaks in the human fibroblast HF19 cell line. Int J Radiat Biol 69: 241–249
Goodhead DT (1989) The initial physical damage produced by ionizing radiations. Int J Radiat Biol 56: 623–634
Grimwade J and Beerman T (1986) Measurement of bleomycin, neocarzinostatin, and auromomycin cleavage of cell-free and intracellular simian virus 40 DNA and chromatin. Mol Pharmacol 30: 358–363
Kenani A, Bailly C, Helbecque N, Catteau JP, Houssin R, Bernier JL and Hénichart JP (1988a) The role of the gulose-mannose part of bleomycin in inactivation of iron-molecular oxygen complex. Biochem J 253: 497–504
Kenani A, Lambling G and Henichart JP (1988b) A convenient method for the cleavage of the D-mannosyl-L-gulose disaccharide from bleomycin A2 . Carbohydr Res 177: 81–89
Kolesnick RN, Haimovitz-Friedman A and Fuks Z (1994) The sphingomyelin signal transduction pathway mediates apoptosis for tumor necrosis factor, Fas, and ionizing radiation. Biochem Cell Biol 72: 471–474
Mir LM, Tounekti O and Orlowski S (1996) Bleomycin: revival of an old drug. Gen Pharmacol 27: 745–748
Oppenheimer NJ, Chang C, Chang L-H, Ehrenfeld GM, Rodriguez LO and Hecht SM (1982) Deglyco-bleomycin Degradation of DNA and formation of a structurally unique Fe(II)-Co complex. J Biol Chem 257: 1606–1609
Orlowski S and Mir LM (1993) Cell electropermeabilization: a new tool for biochemical and pharmacological studies. Biochim Biophys Acta 1154: 51–63
Orlowski S, Belehradek, Jr J, Paoletti C and Mir LM (1988) Transient electropermeabilization of cells in culture. Increase of the cytotoxicity of anticancer drugs. Biochem Pharmacol 37: 4727–4733
Peitsch MC, Mannhertz HG and Tschopp J (1994) The apoptosis endonucleases: cleaning up after cell death?. Trends Cell Biol 4: 37–41
Poddevin B, Orlowski S, Belehradek, Jr J and Mir LM (1991) Very high cytotoxicity of bleomycin introduced into the cytosol of cells in culture. Biochem Pharmacol 42, S. 67–75
Povirk LF, Wübker W, Köhnlein W and Hutchinson F (1977) DNA double-strand breaks and alkali-labile bonds produced by bleomycin. Nucleic Acids Res 4: 3573–3580
Povirk LF, Han YH and Steighner RJ (1989) Structure of bleomycin-induced DNA double-strand breaks: predominance of blunt ends and single-base 5′ extensions. Biochemistry 28: 5808–5814
Santana P, Pena LA, Haimovitz-Friedman A, Martin S, Green D, McLoughlin M, Cordon-Cardo C, Schuchman EH, Fuks Z and Kolesnick R (1996) Acid sphingomyelinase-deficient human lymphoblasts and mice are defective in radiation-induced apoptosis. Cell 86: 189–199
Smith CA, Williams GT, Kingston R, Jenkinson EJ and Owen JJ (1989) Antibodies to CD3/T-cell receptor complex induce death by apoptosis in immature T cells in thymic cultures. Nature 337: 181–184
Sugiyama H, Kilskukie RE, Chang L-H, Ma LT, Hecht SM, van der Marel GA and van Boom JH (1986) DNA strand scission by bleomycin: catalytic cleavage and strand selectivity. J Am Chem Soc 108: 3852–3854
Tomei LD (1991) Apoptosis: A program for death or survival?. Tomei LD, Cope FO (eds), Apoptosis: The molecular basis of cell death, pp. 279–316, Cold Spring Harbor Laboratory Press: New York
Tounekti O, Pron G, Belehradek, Jr J and Mir LM (1993) Bleomycin, an apoptosis-mimetic drug that induces two types of cell death depending on the number of molecules internalized. Cancer Res 53: 5462–5469
Tounekti O, Belehradek, Jr J and Mir LM (1995) Relationships between DNA fragmentation, chromatin condensation and changes in flow cytometry profiles detected during apoptosis. Exp Cell Res 217: 506–516
White E (1996) Life, death, and the pursuit of apoptosis. Genes & Dev 10: 1–15
Yoshida A, Ueda T, Wano Y and Nakamura T (1993) DNA damage and cell killing by camptothecin and its derivative in human leukemia HL-60 cells. Jpn J Cancer Res 84: 566–573
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Tounekti, O., Kenani, A., Foray, N. et al. The ratio of single- to double-strand DNA breaks and their absolute values determine cell death pathway. Br J Cancer 84, 1272–1279 (2001). https://doi.org/10.1054/bjoc.2001.1786
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2001.1786
Keywords
This article is cited by
-
Allium sativum Essential Oil Supplementation Reverses the Hepatic Inflammation, Genotoxicity and Apoptotic Effects in Swiss Albino Mice Intoxicated with the Lead Nitrate
Biological Trace Element Research (2024)
-
Genotoxicity and oxidative stress induction by calcium hydroxide, calcium titanate or/and yttrium oxide nanoparticles in mice
Scientific Reports (2023)
-
Suppression of tumor growth and apoptosis induction by pomegranate seed nano-emulsion in mice bearing solid Ehrlich carcinoma cells
Scientific Reports (2023)
-
Alleviation of Cadmium Chloride–Induced Acute Genotoxicity, Mitochondrial DNA Disruption, and ROS Generation by Chocolate Coadministration in Mice Liver and Kidney Tissues
Biological Trace Element Research (2022)
-
Acute Oral Administration of Cerium Oxide Nanoparticles Suppresses Lead Acetate–Induced Genotoxicity, Inflammation, and ROS Generation in Mice Renal and Cardiac Tissues
Biological Trace Element Research (2022)