Abstract
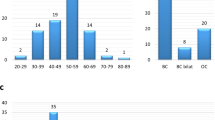
We have analysed 81 families with a history of breast and/or ovarian cancer for the presence of germline mutations in BRCA2 with a number of different mutation screening techniques. The protein truncation test (PTT) for exons 10 and 11 detected four different frame-shifting mutations in six of these families. Four of the remaining 75 families had given positive linkage evidence for being due to BRCA2. In these families the entire coding region was analysed by single-strand conformational polymorphism, leading to the detection of a non-sense and a splice-site mutation in two of them. While these studies were in progress, Southern analysis of BRCA1 revealed that in our study-population of 81 families, 15 families were segregating either the exon 13 or exon 22 deletion in BRCA1 (Petrij-Bosch et al (1997) Nat Genet: 341–345). This prompted us to examine BRCA2 in the remaining 58 families by Southern analysis, using two different restriction enzymes. No aberrations were found in the restriction patterns. Thus, contrary to BRCA1, large genomic rearrangements within the BRCA2 gene do not represent a major mutation mechanism among Dutch breast cancer families. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Cornelis R, Vasen H, Meijers-Heijboer H, Ford D, Van Vliet M, Van Tilborg A, Cleton F, Klijn J, Menko F, Meera Khan P, Cornelisse C and Devilee P (1995) Age at diagnosis as an indicator of eligibility for BRCA1 DNA-testing in familial breast cancer. Hum Genet 95: 539–544
Ford D, Easton DF, Stratton M, Narod S, Goldgar D, Devilee P, Bishop DT, Weber B, Lenoir G, Chang-Claude J, Sobol H, Teare MD, Struewing J, Arason A, Scherneck S, Peto J, Rebbeck TR, Tonin P, Neuhausen S, Barkardottir R, Eyfjord J, Lynch H, Ponder BA, Gayther SA, Birch JM, Lindblom A, Stoppa-Lyonnet D, Bignon Y, Borg A, Hamann U, Haites N, Scott RJ, Maugard CM, Vasen H, Seitz S, Cannon-Albright LA, Schofield A and Zelada-Hedman M the Breast Cancer Linkage Consortium: (1998) Genetic heterogeneity and penetrance analysis of the BRCA1 and BRCA2 genes in breast cancer families. Am J Hum Genet 62: 676–689
Gayther SA, Mangion J, Russell P, Seal S, Barfoot R, Ponder BAJ, Stratton MR and Easton D (1997) Variation of risks of breast and ovarian cancer associated with different germline mutations of the BRCA2 gene. Nat Genet 15: 103–105
Hogervorst F, Cornelis R, Bout M, Van Vliet M, Oosterwijk J, Olmer R, Bakker B, Klijn J, Vasen H, Meijers-Heijboer H, Menko F, Cornelisse C, Den Dunnen J, Devilee P and Van Ommen G-J (1995) Rapid detection of BRCA1 mutations by the Protein Truncation Test. Nat Genet 10: 208–212
Lancaster JM, Wooster R, Mangion J, Phelan CM, Cochran C, Gumbs C, Seal S, Barfoot R, Collins N, Bignell G, Patel S, Hamoudi R, Larsson C, Wiseman RW, Berchuck A, Iglehart JD, Marks JR, Ashworth A, Stratton MR and Futreal PA (1996) BRCA2 mutations in primary breast and ovarian cancers. Nature Genet 13: 238–240
Miki Y, Katagiri T, Kasumi F, Yoshimoto T and Nakamura Y (1996) Mutation analysis in the BRCA2 gene in primary breast cancers. Nature Genet 13: 245–247
Miller SA, Dykes DD and Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16: 1215–1215
Neuhausen S, Gilewski T, Norton L, Tran T, McGuire P, Swensen J, Hampel H, Borgen P, Brown K, Skolnick M, Shattuck-Eidens D, Jhanwar S, Goldgar D and Offit K (1996) Recurrent BRCA2 6174delT mutations in Askenazi Jewish women affected by breast cancer. Nature Genet 13: 126–128
Neuhausen SL, Godwin AK, Gershoni-Baruch R, Schubert E, Garber J, Stoppa-Lyonnet D, Olah E, Csokay B, Serova O, Lalloo F, Osorio A, Stratton M, Offit K, Boyd J, Caligo MA, Scott RJ, Schofield A, Teugels E, Schwab M, Cannon-Albright L, Bishop T, Easton D, Benitez J, King MC, Ponder BA, Weber B, Devilee P, Borg Å, Narod SA and Goldgar D (1998) Haplotype and phenotype analysis of nine recurrent BRCA2 mutations in 111 families: results of an international study. Am J Hum Genet 62: 1381–1388
Nordling M, Karlsson P, Wahlstrom J, Engwall Y, Wallgren A and Martinsson T (1998) A large deletion disrupts the exon 3 transcription activation domain of the BRCA2 gene in a breast/ovarian cancer family. Cancer Res 58: 1372–1375
Peelen T, Cornelis RC, Van Vliet M, Petrij-Bosch A, Cleton-Jansen A-M, Meijers-Heijboer H, Klijn JGM, Vasen HFA, Cornelisse CJ and Devilee P (1996) The majority of 22 Dutch high-risk breast cancer families are due to either BRCA1 or BRCA2. Eur J Hum Genet 4: 225–230
Peelen T, Van Vliet M, Petrij-Bosch A, Mieremet R, Szabo C, Van den Ouweland AMW, Hogervorst F, Brohet R, Ligtenberg MJL, Teugels E, Van der Luijt R, Van der Hout AH, Gille JJP, Pals G, Jedema I, Olmer R, Van Leeuwen I, Newman B, Plandsoen M, Van der Est M, Brink G, Hageman S, Arts PJW, Bakker MM, Willems HW, Van der Looij E, Neyns B, Bonduelle M, Jansen R, Oosterwijk JC, Sijmons R, Smeets HJM, Van Asperen CJ, Meijers-Heijboer H, Klijn JGM, De Greve J, King MC, Menko FH, Brunner HG, Halley D, Van Ommen G-JB, Vasen HFA, Cornelisse CJ, Van’t Veer LJ, De Knijff P, Bakker E and Devilee P (1997) A high proportion of novel mutations in BRCA1 with strong founder effects among Dutch and Belgian hereditary breast and ovarian cancer families. Am J Hum Genet 60: 1041–1049
Petrij-Bosch A, Peelen T, Van Vliet M, Van Eijk R, Olmer R, Drüsedau M, Hogervorst FBL, Hageman S, Arts PJW, Ligtenberg MJL, Meijers-Heijboer H, Klijn JGM, Vasen HFA, Cornelisse CJ, Van't Veer LJ, Bakker E, Van Ommen G-JB and Devilee P (1997) BRCA1 genomic deletions are major founder mutations in Dutch breast cancer patients. Nature Genet 17: 341–345
Phelan CM, Lancaster JM, Tonin P, Gumbs C, Cochran C, Carter R, Ghadirian P, Perret C, Moslehi R, Dion F, Faucher M-C, Dole K, Karimi S, Foulkes W, Lounis H, Warner E, Goss P, Anderson D, Larsson C, Narod SA and Futreal PA (1996) Mutation analysis of the BRCA2 gene in 49 site-specific breast cancer families. Nature Genet 13: 120–122
Puget N, Torchard D, Serova-Sinilnikova OM, Lynch HT, Feunteun J, Lenoir GM and Mazoyer S (1997) A 1-kb Alu-mediated germ-line deletion removing BRCA1 exon 17. Cancer Res 57: 828–831
Puget N, Sinilnikova OM, Stoppa-Lyonnet D, Audoynaud C, Pag Lynch HT, Goldgar D, Lenoir GM and Mazoyer S (1999a) An Alu-mediated 6-kb duplication in the BRCA1 gene: a new founder mutation?. Am J Hum Genet 64: 300–302
Puget N, Stoppalyonnet D, Sinilnikova OM, Pages S, Lynch HT, Lenoir GM and Mazoyer S (1999b) Screening for germ-line rearrangements and regulatory mutations in BRCA1 led to the identification of four new deletions. Cancer Res 59: 455–461
Rahman N and Stratton MR (1998) The genetics of breast cancer susceptibility. Annu Rev Genet 32: 95–121
Ramus SJ, Kotejarai Z, Friedman LS, Van der Looij M, Gayther SA, Csokay B, Ponder BA and Olah E (1997) Analysis of BRCA1 and BRCA2 mutations in Hungarian families with breast or breast-ovarian cancer. Am J Hum Genet 60: 1242–1246
Serova-Sinilnikova OM, Boutrand L, Stoppa-Lyonnet D, Bressac-Depaillerets B, Dubois V, Lasset C, Janin N, Bignon YJ, Longy M, Maugard C, Lidereau R, Leroux D, Frebourg T, Mazoyer S and Lenoir GM (1997) BRCA2 mutations in hereditary breast and ovarian cancer in France. Am J Hum Genet 60: 1236–1239
Smith TM, Lee MK, Szabo CI, Jerome N, Mceuen M, Taylor M, Hood L and King MC (1996) Complete genomic sequence and analysis of 117 kb of human DNA containing the gene BRCA1. Genome Res 6: 1029–1049
Szabo CI and King MC (1997) Population genetics of BRCA1 and BRCA2. Am J Hum Genet 60: 1013–1020
Thorlacius S, Olafsdottir G, Tryggvadottir L, Neuhausen S, Jonasson JG, Tavtigian SV, Tulinius H, Ögmundsdottir HM and Eyfjörd JE (1996) A single BRCA2 mutation in male and female breast cancer families from Iceland with varied cancer phenotypes. Nature Genet 13: 117–119
Tonin PN, Mesmasson AM, Futreal PA, Morgan K, Mahon M, Foulkes WD, Cole DEC, Provencher D, Ghadirian P and Narod SA (1998) Founder BRCA1 and BRCA2 mutations in French Canadian breast and ovarian cancer families. Am J Hum Genet 63: 1341–1351
Vehmanen P, Friedman LS, Eerola H, Sarantaus L, Pyrhonen S, Ponder BAJ, Muhonen T and Nevanlinna H (1997) A low proportion of BRCA2 mutations in Finnish breast cancer families. Am J Hum Genet 60: 1050–1058
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Peelen, T., Vliet, M., Bosch, A. et al. Screening for BRCA2 mutations in 81 Dutch breast–ovarian cancer families. Br J Cancer 82, 151–156 (2000). https://doi.org/10.1054/bjoc.1999.0892
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.1999.0892
Keywords
This article is cited by
-
Functional evaluation of five BRCA2 unclassified variants identified in a Sri Lankan cohort with inherited cancer syndromes using a mouse embryonic stem cell-based assay
Breast Cancer Research (2020)
-
The germline mutational landscape of BRCA1 and BRCA2 in Brazil
Scientific Reports (2018)
-
Large BRCA1 and BRCA2 genomic rearrangements in Polish high-risk breast and ovarian cancer families
Molecular Biology Reports (2013)
-
Molecular and clinical characterization of an in frame deletion of uncertain clinical significance in the BRCA2 gene
Breast Cancer Research and Treatment (2012)
-
Large genomic rearrangements of the BRCA1 and BRCA2 genes: review of the literature and report of a novel BRCA1 mutation
Breast Cancer Research and Treatment (2011)