Abstract
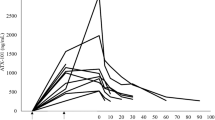
Pre-clinical studies indicate that cisplatin encapsulated in STEALTH®liposomes (SPI-77) retains anti-tumour activity, but has a much reduced toxicity, compared to native cisplatin. A phase I study was conducted to determine the toxicity and pharmacokinetics of SPI-77 administered to children with advanced cancer not amenable to other treatment. Paediatric patients were treated at doses ranging from 40 to 320 mg m−2by intravenous infusion every 4 weeks. Blood samples taken during, and up to 3 weeks after, administration and plasma and ultrafiltrate were prepared immediately. Urine was collected, when possible, for 3 days after administration. SPI-77 administration was well tolerated with the major toxicity being an infusion reaction which responded to modification of the initial infusion rate of SPI-77. Limited haematological toxicity and no nephrotoxicity were observed. No responses to treatment were seen during the course of this phase I study. Measurement of total plasma platinum showed that cisplatin was retained in the circulation with a half life of up to 134 h, with maximum plasma concentrations approximately 100-fold higher than those reported following comparable doses of cisplatin. Comparison of plasma and whole blood indicated that cisplatin was retained in the liposomes and there was no free platinum measurable in the ultrafiltrate. Urine recovery was less than 4% of the dose administered over 72 h. Results from this phase I study indicate that high doses of liposomal cisplatin can safely be given to patients, but further studies are required to address the issue of reformulation of liposomally bound cisplatin. © 2001 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Bramwell VH, Burgers M, Sneath R, Souhami R, van Oosterom AT, Voûte PA, Rouesse J, Spooner D, Craft AW, Somers R, Pringle J, Malcolm AJ, van der Eijken J, Thomas D, Uscinska B, Machin D and van Glabbeke M (1992) A comparison of two short intensive adjuvant chemotherapy regimens in operable osteosarcoma of the limbs in children and young adults: the first study of the European Osteosarcoma Intergroup. J Clin Oncol 10: 1579–1531
Buckner JC, Peethambaram PP, Smithson WA, Groover RV, Schomberg PJ, Kimmel DW, Raffel C, OFallon JR, Neglia J and Shaw EG (1999) Phase II trial of primary chemotherapy followed by reduced-dose radiation for CNS germ cell tumors. J Clin Oncol 17: 933–940
Bues-Charbit M, Gentet JC, Bernard JL, Breant V, Cano JP and Raybaud C (1987) Continuous infusion of high-dose cisplatin in children: pharmacokinetics of free and total platinum. Eur J Cancer Clin Oncol 23: 1649–1652
Cavaletti G, Marzorati L, Bogliun G, Colombo N, Marzola M, Pittelli MR and Tredici G (1992) Cisplatin-induced peripheral neurotoxicity is dependent on total dose-intensity and single dose-intensity. Cancer 69: 203–207
Dominici C, Petrucci F, Caroli S, Alimonti A, Clerico A and Castello MA (1989) A pharmacokinetic study of high-dose continuous infusion cisplatin in children with solid tumors. J Clin Oncol 7: 100–107
Gill PS, Espina BM, Muggia F, Cabriales S, Tulpule A, Esplin JA, Liebman HA, Forssen E, Ross ME and Levine AM (1995) Phase I/II clinical and pharmacological evaluation of liposomal daunorubicin. J Clin Oncol 13: 996–1003
Krakoff IH (1979) Nephrotoxicity of cis-dichlorodiammineplatinum (II). Cancer Treat Rep 63: 1523–1525
Manaka RC and Wolf W (1978) Distribution of cis -platin in blood. Chem Biol Interactions 22: 353–358
Muggia FM, Hainsworth JD, Jeffers S, Miller P, Groshen S, Tan M, Roman L, Uziely B, Muderspach L, Garcia A, Burnett A, Greco FA, Morrow CP, Paradiso LJ and Liang LJ (1997) Phase II study of liposomal doxorubicin in refractory ovarian cancer: antitumor activity and toxicity modification by liposomal encapsulation. J Clin Oncol 15: 987–993
Newman MS, Colbern GT, Working PK, Engbers C and Amantia MA (1999) Comparative pharmacokinetics, tissue distribution, and therapeutic effectiveness of cisplatin encapsulated in long-circulating, pegylated liposomes (SPI-077) in tumor-bearing mice. Cancer Chemother Pharmacol 43: 1–7
Northfelt DW, Dezube BJ, Thommes JA, Levine R, Von Roenn JH, Dosik GM, Rios A, Krown SE, DuMond C and Mamelok RD (1997) Efficacy of pegylated-liposomal doxorubicin in the treatment of AIDS-related Kaposi’s sarcoma after failure of standard chemotherapy. J Clin Oncol 15: 653–659
Papahadjopoulous D, Allen TN, Gabizon A, Mayhew E, Matthay K, Huang SK, Lee KD, Woodle MC, Lasic DD, Redemann C and Martin FJ (1991) Sterically stabilized liposomes: Improvement in pharmacokinetics and antitumor therapeutic efficacy. Proc Natl Acad Sci (USA) 88: 11460–11464
Pearson AD, Craft AW, Pinkerton CR, Meller ST and Reid MM (1992) High-dose rapid schedule chemotherapy for disseminated neuroblastoma. Eur J Cancer 28A: 1654–1659
Peng B, English M, Boddy AV, Price L, Wyllie R, Pearson ADJ, Tilby MJ and Newell DR (1997) Cisplatin pharmacokinetics in children. Eur J Cancer 33: 1823–1828
Pratt CB, Hayes A, Green AA, Evans WE, Senzer N, Howarth CB, Ransom JL and Crom W (1981) Pharmacokinetic evaluation of cisplatin in children with malignant solid tumors: a phase II study. Cancer Treat Rep 65: 1021–1026
Rahman A, Treat J, Roh JK, Potkul LA, Alvord WG, Forst D and Woolley PV (1990) A phase I clinical trial and pharmacokinetic evaluation of liposome encapsualted doxorubicin. J Clin Oncol 8: 1093–1100
Schellens JHM, Meerum Terwogt J, Groenewegen G, Blijham GH, Ten Bokkel Huinink WW, Smart M, Maliepaard M Floot B, Welbank H and Beijnen JH (1998) Phase I and pharmacologic study of SPI-77, a novel stealth liposomal encapsulated formulation of cisplatin (CDDP). Eighty-ninth annual meeting, Proceedings American Association Cancer Research 39: P2218
Uziely B, Jeffers S, Isacson R, Kutsch K, Wei-Tsao D, Yehoshua Z, Libson E, Muggia FM and Gabizon A (1995) Liposomal doxorubicin (Dox-SL™): Antitumor activity and unique toxicities during two complementary phase I studies. J Clin Oncol 13: 1777–1785
Vaage J, Mayhew E, Lasic D and Martin F (1992) Therapy of primary and metastatic mouse mammary carcinoma with doxorubicin encapsulated in long circulating liposomes. Int J Cancer 51: 942–948
Vermorken JB, Kaptein TS, Hart AAM and Pinedo HM (1983) Ototoxicity of cisdiamminedichloroplatinum (II). Influence of dose, schedule and mode of administration. Eur J Cancer Clin Oncol 19: 53–58
Woodle MC and Lasic DD (1992) Sterically stabilized liposomes. Biochem Biophys Acta 1113: 171
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Veal, G., Griffin, M., Price, E. et al. A phase I study in paediatric patients to evaluate the safety and pharmacokinetics of SPI-77, a liposome encapsulated formulation of cisplatin. Br J Cancer 84, 1029–1035 (2001). https://doi.org/10.1054/bjoc.2001.1723
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2001.1723
Keywords
This article is cited by
-
Medication-Induced Nephrotoxicity in Children
Current Pediatrics Reports (2020)
-
Platinum-based drugs: past, present and future
Cancer Chemotherapy and Pharmacology (2016)