Abstract
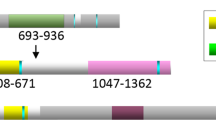
An unusual group of carcinomas, here termed nuclear protein in testis (NUT) midline carcinomas (NMC), are characterized by translocations that involve NUT, a novel gene on chromosome 15. In about 2/3rds of cases, NUT is fused to BRD4 on chromosome 19. Using a candidate gene approach, we identified two NMCs harboring novel rearrangements that result in the fusion of NUT to BRD3 on chromosome 9. The BRD3–NUT fusion gene encodes a protein composed of two tandem chromatin-binding bromodomains, an extra-terminal domain, a bipartite nuclear localization sequence, and almost the entirety of NUT that is highly homologous to BRD4–NUT. The function of NUT is unknown, but here we show that NUT contains nuclear localization and export sequences that promote nuclear-cytoplasmic shuttling via a leptomycin-sensitive pathway. In contrast, BRD3–NUT and BRD4–NUT are strictly nuclear, implying that the BRD moiety retains NUT in the nucleus via interactions with chromatin. Consistent with this idea, FRAP studies show that BRD4, BRD4–NUT and BRD3–NUT have significantly slower rates of lateral nuclear diffusion than that of NUT. To investigate the functional role of BRD–NUT fusion proteins in NMCs, we investigated the effects of siRNA-induced BRD3–NUT and BRD4–NUT withdrawal. Silencing of these proteins in NMC cell lines resulted in squamous differentiation and cell cycle arrest. Together, these data suggest that BRD–NUT fusion proteins contribute to carcinogenesis by associating with chromatin and interfering with epithelial differentiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Dey A, Chitsaz F, Abbasi A, Misteli T, Ozato K . (2003). The double bromodomain protein Brd4 binds to acetylated chromatin during interphase and mitosis. Proc Natl Acad Sci USA 100: 8758–8763.
Farina A, Hattori M, Qin J, Nakatani Y, Minato N, Ozato K . (2004). Bromodomain protein Brd4 binds to GTPase-activating SPA-1, modulating its activity and subcellular localization. Mol Cell Biol 24: 9059–9069.
Fornerod M, Ohno M, Yoshida M, Mattaj IW . (1997). CRM1 is an export receptor for leucine-rich nuclear export signals. Cell 90: 1051–1060.
French CA, Kutok JL, Faquin WC, Toretsky JA, Antonescu CR, Griffin CA et al. (2004). Midline carcinoma of children and young adults with NUT rearrangement. J Clin Oncol 22: 4135–4139.
French CA, Miyoshi I, Aster JC, Kubonishi I, Kroll TG, Dal Cin P et al. (2001). BRD4 bromodomain gene rearrangement in aggressive carcinoma with translocation t(15;19). Am J Pathol 159: 1987–1992.
French CA, Miyoshi I, Kubonishi I, Grier HE, Perez-Atayde AR, Fletcher JA . (2003). BRD4–NUT fusion oncogene: a novel mechanism in aggressive carcinoma. Cancer Res 63: 304–307.
Haruki N, Kawaguchi KS, Eichenberger S, Massion PP, Gonzalez A, Gazdar AF et al. (2005). Cloned fusion product from a rare t(15;19)(q13.2;p13.1) inhibit S phase in vitro. J Med Genet 42: 558–564.
Jang MK, Mochizuki K, Zhou M, Jeong HS, Brady JN, Ozato K . (2005). The bromodomain protein Brd4 is a positive regulatory component of P-TEFb and stimulates RNA polymerase II-dependent transcription. Mol Cell 19: 523–534.
Kees UR, Mulcahy MT, Willoughby ML . (1991). Intrathoracic carcinoma in an 11-year-old girl showing a translocation t(15;19). Am J Pediatr Hematol Oncol 13: 459–464.
Maruyama T, Farina A, Dey A, Cheong J, Bermudez VP, Tamura T et al. (2002). A mammalian bromodomain protein, brd4, interacts with replication factor C and inhibits progression to S phase. Mol Cell Biol 22: 6509–6520.
Mertens F, Wiebe T, Adlercreutz C, Mandahl N, French CA . (2006). Successful treatment of a child with t(15;19)-positive tumor. Pediatr Blood Cancer; e-pub ahead of print; 2 January 2007.
Ossareh-Nazari B, Bachelerie F, Dargemont C . (1997). Evidence for a role of CRM1 in signal-mediated nuclear protein export. Science 278: 141–144.
Ottinger M, Christalla T, Nathan K, Brinkmann MM, Viejo-Borbolla A, Schulz TF . (2006). The Kaposi’s Sarcoma-associated Herpesvirus LANA-1 interacts with the short variant of BRD4 and releases cells from a BRD4- and BRD2/RING3- induced G1 cell cycle arrest. J Virol 80: 10772–10786.
Toretsky JA, Jenson J, Sun CC, Eskenazi AE, Campbell A, Hunger SP . (2003). Translocation (11;15;19): a highly specific chromosome rearrangement associated with poorly differentiated thymic carcinoma in young patients. Am J Clin Oncol 26: 300–306.
Yang Z, Yik JH, Chen R, He N, Jang MK, Ozato K et al. (2005). Recruitment of P-TEFb for stimulation of transcriptional elongation by the bromodomain protein Brd4. Mol Cell 19: 535–545.
Acknowledgements
We thank Drs Andrew Weng and Stephen Blacklow for helpful advice and discussion. CAF is supported by a grants from the National Cancer Institute.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
French, C., Ramirez, C., Kolmakova, J. et al. BRD–NUT oncoproteins: a family of closely related nuclear proteins that block epithelial differentiation and maintain the growth of carcinoma cells. Oncogene 27, 2237–2242 (2008). https://doi.org/10.1038/sj.onc.1210852
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210852
Keywords
This article is cited by
-
Top IHC/ISH Hacks for and Molecular Surrogates of Poorly Differentiated Sinonasal Small Round Cell Tumors
Head and Neck Pathology (2024)
-
Molecularly defined sinonasal malignancies: an overview with focus on the current WHO classification and recently described provisional entities
Virchows Archiv (2024)
-
Sinonasal NUT Carcinoma: A Call for Increased Attention to a Vital Clinical Diagnosis
Indian Journal of Surgical Oncology (2024)
-
Unusual lung tumors—from morphology to genetics
Modern Pathology (2022)
-
Therapeutic impact of BET inhibitor BI 894999 treatment: backtranslation from the clinic
British Journal of Cancer (2022)