Abstract
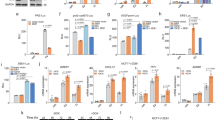
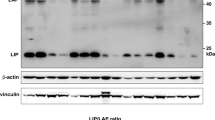
The transcription factor nuclear factor kappa B (NF-κB) is constitutively active in both cancer cells and stromal cells of breast cancer; however, the precise role of activated NF-κB in cancer progression is not known. Using parental MCF10A cells and a variant that expresses the myoepithelial marker p63 stably overexpressing the constitutively active p65 subunit of NF-κB (MCF10A/p65), we show that NF-κB suppresses the expression of epithelial specific genes E-cadherin and desmoplakin and induces the expression of the mesenchymal specific gene vimentin. P65 also suppressed the expression of p63 and the putative breast epithelial progenitor marker cytokeratin 5/6. MCF10A/p65 cells were phenotypically similar to cells undergoing epithelial to mesenchymal transition (EMT). MCF10A/p65 cells failed to form characteristic acini in three-dimensional Matrigel. Analysis of parental and MCF10A/p65 cells for genes previously shown to be involved in EMT revealed elevated expression of ZEB-1 and ZEB-2 in MCF10A/p65 cells compared to parental cells. In transient transfection assays, p65 increased ZEB-1 promoter activity. Furthermore, MCF10A cells overexpressing ZEB-1 showed reduced E-cadherin and p63 expression and displayed an EMT phenotype. The siRNA against ZEB-1 or ZEB-2 reduced the number of viable MCF10A/p65 but not parental cells, suggesting the dependence of MCF10A/p65 cells to ZEB-1 and ZEB-2 for cell cycle progression or survival. MCF10A cells chronically exposed to tumor necrosis factor alpha (TNFα), a potent NF-κB inducer, also exhibited the EMT-like phenotype and ZEB-1/ZEB-2 induction, both of which were reversed following TNFα withdrawal.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Abbreviations
- EMT:
-
epithelial to mesenchymal transition
- IL-1:
-
interleukin-1
- MnSOD:
-
manganese superoxide dismutase
- NF-κB:
-
nuclear factor kappaB
- NLS:
-
nuclear localization signal
- TGFβ:
-
transforming growth factor beta
- TNF:
-
tumor necrosis factor
References
Adriance MC, Inman JL, Petersen OW, Bissell MJ . (2005). Myoepithelial cells: good fences make good neighbors. Breast Cancer Res 7: 190–197.
Akimaru H, Hou DX, Ishii S . (1997). Drosophila CBP is required for dorsal-dependent twist gene expression. Nat Genet 17: 211–214.
Barbareschi M, Pecciarini L, Cangi MG, Macri E, Rizzo A, Viale G et al. (2001). p63, a p53 homologue, is a selective nuclear marker of myoepithelial cells of the human breast. Am J Surg Pathol 25: 1054–1060.
Bhat-Nakshatri P, Newton TR, Goulet Jr R, Nakshatri H . (1998). NF-kappaB activation and interleukin 6 production in fibroblasts by estrogen receptor-negative breast cancer cell-derived interleukin 1alpha. Proc Natl Acad Sci USA 95: 6971–6976.
Bhat-Nakshatri P, Sweeney CJ, Nakshatri H . (2002). Identification of signal transduction pathways involved in constitutive NF-kappaB activation in breast cancer cells. Oncogene 21: 2066–2078.
Bhowmick NA, Chytil A, Plieth D, Gorska AE, Dumont N, Shappell S et al. (2004). TGF-beta signaling in fibroblasts modulates the oncogenic potential of adjacent epithelia. Science 303: 848–851.
Biswas DK, Cruz AP, Gansberger E, Pardee AB . (2000). Epidermal growth factor-induced nuclear factor kappa B activation: a major pathway of cell-cycle progression in estrogen-receptor negative breast cancer cells. Proc Natl Acad Sci USA 97: 8542–8547.
Biswas DK, Shi Q, Baily S, Strickland I, Ghosh S, Pardee AB et al. (2004). NF-kappa B activation in human breast cancer specimens and its role in cell proliferation and apoptosis. Proc Natl Acad Sci USA 101: 10137–10142.
Biswas DK, Singh S, Shi Q, Pardee AB, Iglehart JD . (2005). Crossroads of estrogen receptor and NF-kappaB signaling. Sci STKE 2005: pe27.
Boecker W, Moll R, Dervan P, Buerger H, Poremba C, Diallo RI et al. (2002). Usual ductal hyperplasia of the breast is a committed stem (progenitor) cell lesion distinct from atypical ductal hyperplasia and ductal carcinoma in situ. J Pathol 198: 458–467.
Brantley DM, Chen CL, Muraoka RS, Bushdid PB, Bradberry JL, Kittrell F et al. (2001). Nuclear factor-kappaB (NF-kappaB) regulates proliferation and branching in mouse mammary epithelium. Mol Biol Cell 12: 1445–1455.
Cao Y, Bonizzi G, Seagroves TN, Greten FR, Johnson R, Schmidt EV et al. (2001). IKKalpha provides an essential link between RANK signaling and cyclin D1 expression during mammary gland development. Cell 107: 763–775.
Charafe-Jauffret E, Ginestier C, Monville F, Finetti P, Adelaide J, Cervera N et al. (2006). Gene expression profiling of breast cell lines identifies potential new basal markers. Oncogene 25: 2273–2284.
Clarke CL, Sandle J, Parry SC, Reis-Filho JS, O'Hare MJ, Lakhani SR . (2004). Cytokeratin 5/6 in normal human breast: lack of evidence for a stem cell phenotype. J Pathol 204: 147–152.
Clarkson RW, Heeley JL, Chapman R, Aillet F, Hay RT, Wyllie A et al. (2000). NF-kappaB inhibits apoptosis in murine mammary epithelia. J Biol Chem 275: 12737–12742.
Cogswell PC, Guttridge DC, Funkhouser WK, Baldwin Jr AS . (2000). Selective activation of NF-kappaB subunits in human breast cancer: potential roles for NF-kappaB2/p52 and for Bcl-3. Oncogene 19: 1123–1131.
Comijn J, Berx G, Vermassen P, Verschueren K, van Grunsven L, Bruyneel E et al. (2001). The two-handed E box binding zinc finger protein SIP1 downregulates E-cadherin and induces invasion. Mol Cell 7: 1267–1278.
Debnath J, Mills KR, Collins NL, Reginato MJ, Muthuswamy SK, Brugge JS . (2002). The role of apoptosis in creating and maintaining luminal space within normal and oncogene-expressing mammary acini. Cell 111: 29–40.
Dimri GP, Lee X, Basile G, Acosta M, Scott G, Roskelley C et al. (1995). A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci USA 92: 9363–9367.
Dontu G, El-Ashry D, Wicha MS . (2004). Breast cancer, stem/progenitor cells and the estrogen receptor. Trends Endocrinol Metab 15: 193–197.
Eger A, Aigner K, Sonderegger S, Dampier B, Oehler S, Schreiber M et al. (2005). DeltaEF1 is a transcriptional repressor of E-cadherin and regulates epithelial plasticity in breast cancer cells. Oncogene 24: 2375–2385.
Elloul S, Elstrand MB, Nesland JM, Trope CG, Kvalheim G, Goldberg I et al. (2005). Snail, Slug, and Smad-interacting protein 1 as novel parameters of disease aggressiveness in metastatic ovarian and breast carcinoma. Cancer 103: 1631–1643.
Ghosh S, Karin M . (2002). Missing pieces in the NF-kappaB puzzle. Cell 109 (Suppl): S81–S96.
Gordon LA, Mulligan KT, Maxwell-Jones H, Adams M, Walker RA, Jones JL . (2003). Breast cell invasive potential relates to the myoepithelial phenotype. Int J Cancer 106: 8–16.
Guaita S, Puig I, Franci C, Garrido M, Dominguez D, Batlle E et al. (2002). Snail induction of epithelial to mesenchymal transition in tumor cells is accompanied by MUC1 repression and ZEB1 expression. J Biol Chem 277: 39209–39216.
Huber MA, Azoitei N, Baumann B, Grunert S, Sommer A, Pehamberger H et al. (2004). NF-kappaB is essential for epithelial-mesenchymal transition and metastasis in a model of breast cancer progression. J Clin Invest 114: 569–581.
Ip MM, Shoemaker SF, Darcy KM . (1992). Regulation of rat mammary epithelial cell proliferation and differentiation by tumor necrosis factor-alpha. Endocrinology 130: 2833–2844.
Irie HY, Pearline RV, Grueneberg D, Hsia M, Ravichandran P, Kothari N et al. (2005). Distinct roles of Akt1 and Akt2 in regulating cell migration and epithelial–mesenchymal transition. J Cell Biol 171: 1023–1034.
Janda E, Lehmann K, Killisch I, Jechlinger M, Herzig M, Downward J et al. (2002). Ras and TGF[beta] cooperatively regulate epithelial cell plasticity and metastasis: dissection of Ras signaling pathways. J Cell Biol 156: 299–313.
Jonkers J, Berns A . (2004). Oncogene addiction: sometimes a temporary slavery. Cancer Cell 6: 535–538.
Karin M, Lin A . (2002). NF-kappaB at the crossroads of life and death. Nat Immunol 3: 221–227.
Keyes WM, Wu Y, Vogel H, Guo X, Lowe SW, Mills AA . (2005). p63 deficiency activates a program of cellular senescence and leads to accelerated aging. Genes Dev 19: 1986–1999.
Kim DW, Sovak MA, Zanieski G, Nonet G, Romieu-Mourez R, Lau AW et al. (2000). Activation of NF-kappaB/Rel occurs early during neoplastic transformation of mammary cells. Carcinogenesis 21: 871–879.
Kumar S, Kishimoto H, Chua HL, Badve S, Miller KD, Bigsby RM et al. (2003). Interleukin-1 alpha promotes tumor growth and cachexia in MCF-7 xenograft model of breast cancer. Am J Pathol 163: 2531–2541.
Kuphal S, Poser I, Jobin C, Hellerbrand C, Bosserhoff AK . (2004). Loss of E-cadherin leads to upregulation of NFkappaB activity in malignant melanoma. Oncogene 23: 8509–8519.
Kurose K, Gilley K, Matsumoto S, Watson PH, Zhou XP, Eng C . (2002). Frequent somatic mutations in PTEN and TP53 are mutually exclusive in the stroma of breast carcinomas. Nat Genet 32: 355–357.
Lu T, Sathe SS, Swiatkowski SM, Hampole CV, Stark GR . (2004). Secretion of cytokines and growth factors as a general cause of constitutive NFkappaB activation in cancer. Oncogene 23: 2138–2145.
McKeon F . (2004). p63 and the epithelial stem cell: more than status quo? Genes Dev 18: 465–469.
Meng F, Liu L, Chin PC, D'Mello SR . (2002). Akt is a downstream target of NF-kappa B. J Biol Chem 277: 29674–29680.
Mills KR, Reginato M, Debnath J, Queenan B, Brugge JS . (2004). Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) is required for induction of autophagy during lumen formation in vitro. Proc Natl Acad Sci USA 101: 3438–3443.
Moody SE, Perez D, Pan TC, Sarkisian CJ, Portocarrero CP, Sterner CJ et al. (2005). The transcriptional repressor Snail promotes mammary tumor recurrence. Cancer Cell 8: 197–209.
Nakshatri H, Bhat-Nakshatri P, Martin DA, Goulet Jr RJ, Sledge Jr GW . (1997). Constitutive activation of NF-kappaB during progression of breast cancer to hormone-independent growth. Mol Cell Biol 17: 3629–3639.
Nakshatri H, Goulet Jr RJ . (2002). NF-kappaB and breast cancer. Curr Probl Cancer 26: 282–309.
Nieto MA . (2002). The snail superfamily of zinc-finger transcription factors. Nat Rev Mol Cell Biol 3: 155–166.
Nozaki S, Sledge Jr GW, Nakshatri H . (2000). Cancer cell-derived interleukin 1alpha contributes to autocrine and paracrine induction of pro-metastatic genes in breast cancer. Biochem Biophys Res Commun 275: 60–62.
Ozturk N, Erdal E, Mumcuoglu M, Akcali KC, Yalcin O, Senturk S et al. (2006). Reprogramming of replicative senescence in hepatocellular carcinoma-derived cells. Proc Natl Acad Sci USA 103: 2178–2183.
Pahl HL . (1999). Activators and target genes of Rel/NF-kappaB transcription factors. Oncogene 18: 6853–6866.
Perou CM, Sorlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA et al. (2000). Molecular portraits of human breast tumours. Nature 406: 747–752.
Petersen OW, Nielsen HL, Gudjonsson T, Villadsen R, Rank F, Niebuhr E et al. (2003). Epithelial to mesenchymal transition in human breast cancer can provide a nonmalignant stroma. Am J Pathol 162: 391–402.
Pikarsky E, Porat RM, Stein I, Abramovitch R, Amit S, Kasem S et al. (2004). NF-kappaB functions as a tumour promoter in inflammation-associated cancer. Nature 431: 461–466.
Postigo AA . (2003). Opposing functions of ZEB proteins in the regulation of the TGFbeta/BMP signaling pathway. EMBO J 22: 2443–2452.
Postigo AA, Dean DC . (2000). Differential expression and function of members of the zfh-1 family of zinc finger/homeodomain repressors. Proc Natl Acad Sci USA 97: 6391–6396.
Radisky DC, Bissell MJ . (2004). Cancer. Respect thy neighbor!. Science 303: 775–777.
Rapp UR, Gotz R, Albert S . (2006). BuCy RAFs drive cells into MEK addiction. Cancer Cell 9: 9–12.
Rizki A, Bissell MJ . (2004). Homeostasis in the breast: it takes a village. Cancer Cell 6: 1–2.
Romieu-Mourez R, Landesman-Bollag E, Seldin DC, Sonenshein GE . (2002). Protein kinase CK2 promotes aberrant activation of nuclear factor-kappaB, transformed phenotype, and survival of breast cancer cells. Cancer Res 62: 6770–6778.
Shin SR, Sanchez-Velar N, Sherr DH, Sonenshein GE . (2006). 7,12-dimethylbenz(a)anthracene treatment of a c-rel mouse mammary tumor cell line induces epithelial to mesenchymal transition via activation of nuclear factor-kappaB. Cancer Res 66: 2570–2575.
Sorlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H et al. (2001). Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 98: 10869–10874.
Sosic D, Richardson JA, Yu K, Ornitz DM, Olson EN . (2003). Twist regulates cytokine gene expression through a negative feedback loop that represses NF-kappaB activity. Cell 112: 169–180.
Sovak MA, Bellas RE, Kim DW, Zanieski GJ, Rogers AE, Traish AM et al. (1997). Aberrant nuclear factor-kappaB/Rel expression and the pathogenesis of breast cancer. J Clin Invest 100: 2952–2960.
Tait L, Soule HD, Russo J . (1990). Ultrastructural and immunocytochemical characterization of an immortalized human breast epithelial cell line, MCF-10. Cancer Res 50: 6087–6094.
Tarin D, Thompson EW, Newgreen DF . (2005). The fallacy of epithelial mesenchymal transition in neoplasia. Cancer Res 65: 5996–6000.
Thiery JP . (2003). Epithelial–mesenchymal transitions in development and pathologies. Curr Opin Cell Biol 15: 740–746.
Thompson EW, Newgreen DF, Tarin D . (2005). Carcinoma invasion and metastasis: a role for epithelial–mesenchymal transition? Cancer Res 65: 5991–5995.
Tickle C . (1998). Worlds in common through NF-kappaB. Nature 392: 547–549.
Vandewalle C, Comijn J, De Craene B, Vermassen P, Bruyneel E, Andersen H et al. (2005). SIP1/ZEB2 induces EMT by repressing genes of different epithelial cell–cell junctions. Nucleic Acids Res 33: 6566–6578.
Villadsen R . (2005). In search of a stem cell hierarchy in the human breast and its relevance to breast cancer evolution. Apmis 113: 903–921.
Welm B, Behbod F, Goodell MA, Rosen JM . (2003). Isolation and characterization of functional mammary gland stem cells. Cell Prolif 36 (Suppl 1): 17–32.
Yang J, Mani SA, Donaher JL, Ramaswamy S, Itzykson RA, Come C et al. (2004). Twist, a master regulator of morphogenesis, plays an essential role in tumor metastasis. Cell 117: 927–939.
Zhou BP, Deng J, Xia W, Xu J, Li YM, Gunduz M et al. (2004). Dual regulation of Snail by GSK-3beta-mediated phosphorylation in control of epithelial–mesenchymal transition. Nat Cell Biol 6: 931–940.
Acknowledgements
We thank Meei-Huey Jeng and George Sledge for providing the MCF10A(E) and MCF10A(M) cells, respectively, Tom Genetta for the ZEB-1 cDNA, Geert Berx for the ZEB-2 cDNA, and Louis Pelus for p63 cDNA. We are also grateful to Drs Attaya Suvannasankha, Zhuo Wang and Colin Crean for their critical review of our manuscript. HN is Marian J Morrison Investigator for Breast Cancer Research. This work is supported by the Department of Defense Grant DAMD17-01-1-0274 to H.N.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chua, H., Bhat-Nakshatri, P., Clare, S. et al. NF-κB represses E-cadherin expression and enhances epithelial to mesenchymal transition of mammary epithelial cells: potential involvement of ZEB-1 and ZEB-2. Oncogene 26, 711–724 (2007). https://doi.org/10.1038/sj.onc.1209808
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209808
Keywords
This article is cited by
-
RUNX1-IT1 acts as a scaffold of STAT1 and NuRD complex to promote ROS-mediated NF-κB activation and ovarian cancer progression
Oncogene (2024)
-
NF-κB signaling in neoplastic transition from epithelial to mesenchymal phenotype
Cell Communication and Signaling (2023)
-
Pancreatic cancer stemness: dynamic status in malignant progression
Journal of Experimental & Clinical Cancer Research (2023)
-
New ruthenium-xanthoxylin complex eliminates colorectal cancer stem cells by targeting the heat shock protein 90 chaperone
Cell Death & Disease (2023)
-
The extracellular matrix alteration, implication in modulation of drug resistance mechanism: friends or foes?
Journal of Experimental & Clinical Cancer Research (2022)