Abstract
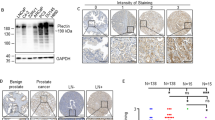
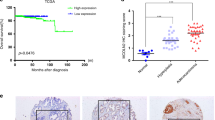
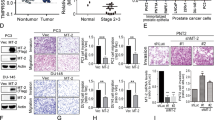
SUMO-specific protease 1 (SENP1) is a member of de-SUMOylation protease family and has an important role in the regulation of androgen receptor-dependent transcription and hypoxia signaling. This activity profile of SENP1 prompted us to investigate whether SENP1 is involved in the pathogenesis of prostate cancer. In previous studies, we have detected the overexpression of SENP1 in both precancerous prostate intraepithelial neoplasia (PIN) lesions and prostate cancer tissue samples from patients. Whereas our whole-animal model has demonstrated that SENP1 induction is critical for prostate cell transformation, the role of SENP1 in prostate cancer progression is still unknown. In this study, we show that SENP1 expression directly correlates with prostate cancer aggressiveness and reccurrence, by analyzing more than 150 prostate cancer specimens. Modulating SENP1 level dictates colony formation of prostate cancer cell lines, tumor growth in nude mice and also prostate cancer cell migration and invasion. Silencing SENP1 level in highly metastatic prostate cancer cells perturbs their ability to metastasize to the bone and initiates secondary tumors. Mechanistically, the expression of two critical bone remodeling proteins, matrix metalloproteinase 2 (MMP2) and MMP9, is regulated by SENP1 through the HIF1α signaling pathway. All these results show the contribution of SENP1 to the progression of prostate cancer, and suggest that SENP1 may be a prognostic marker and a therapeutic target for metastasis in prostate cancer patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Yeh ET . SUMOylation and De-SUMOylation: wrestling with life's processes. J Biol Chem 2009; 284: 8223–8227.
Hay RT . SUMO: a history of modification. Mol Cell 2005; 18: 1–12.
Kim JH, Choi HJ, Kim B, Kim MH, Lee JM, Kim IS et al. Roles of sumoylation of a reptin chromatin-remodelling complex in cancer metastasis. Nat Cell Biol 2006; 8: 631–639.
Yang Y, Fu W, Chen J, Olashaw N, Zhang X, Nicosia SV et al. SIRT1 sumoylation regulates its deacetylase activity and cellular response to genotoxic stress. Nat Cell Biol 2007; 9: 1253–1262.
Yu L, Ji W, Zhang H, Renda MJ, He Y, Lin S et al. SENP1-mediated GATA1 deSUMOylation is critical for definitive erythropoiesis. J Exp Med 2010; 207: 1183–1195.
Cheng J, Wang D, Wang Z, Yeh ET . SENP1 enhances androgen receptor-dependent transcription through desumoylation of histone deacetylase 1. Mol Cell Biol 2004; 24: 6021–6028.
Cheng J, Kang X, Zhang S, Yeh ET . SUMO-specific protease 1 is essential for stabilization of HIF1alpha during hypoxia. Cell 2007; 131: 584–595.
Kaikkonen S, Jaaskelainen T, Karvonen U, Rytinki MM, Makkonen H, Gioeli D et al. SUMO-specific protease 1 (SENP1) reverses the hormone-augmented SUMOylation of androgen receptor and modulates gene responses in prostate cancer cells. Mol Endocrinol 2009; 23: 292–307.
Yamaguchi T, Sharma P, Athanasiou M, Kumar A, Yamada S, Kuehn MR . Mutation of SENP1/SuPr-2 reveals an essential role for desumoylation in mouse development. Mol Cell Biol 2005; 25: 5171–5182.
Bawa-Khalfe T, Cheng J, Wang Z, Yeh ET . Induction of the SUMO-specific protease 1 transcription by the androgen receptor in prostate cancer cells. J Biol Chem 2007; 282: 37341–37349.
Cheng J, Bawa T, Lee P, Gong L, Yeh ET . Role of desumoylation in the development of prostate cancer. Neoplasia 2006; 8: 667–676.
Bawa-Khalfe T, Cheng J, Lin SH, Ittmann MM, Yeh ET . SENP1 induces prostatic intraepithelial neoplasia through multiple mechanisms. J Biol Chem 2010; 285: 25859–25866.
Semenza GL . Defining the role of hypoxia-inducible factor 1 in cancer biology and therapeutics. Oncogene 2010; 29: 625–634.
Nemeth JA, Yousif R, Herzog M, Che M, Upadhyay J, Shekarriz B et al. Matrix metalloproteinase activity, bone matrix turnover, and tumor cell proliferation in prostate cancer bone metastasis. J Natl Cancer Inst 2002; 94: 17–25.
Kessenbrock K, Plaks V, Werb Z . Matrix metalloproteinases: regulators of the tumor microenvironment. Cell 2010; 141: 52–67.
Littlepage LE, Sternlicht MD, Rougier N, Phillips J, Gallo E, Yu Y et al. Matrix metalloproteinases contribute distinct roles in neuroendocrine prostate carcinogenesis, metastasis, and angiogenesis progression. Cancer Res 2010; 70: 2224–2234.
Trudel D, Fradet Y, Meyer F, Harel F, Tetu B . Significance of MMP-2 expression in prostate cancer: an immunohistochemical study. Cancer Res 2003; 63: 8511–8515.
Sehgal G, Hua J, Bernhard EJ, Sehgal I, Thompson TC, Muschel RJ . Requirement for matrix metalloproteinase-9 (gelatinase B) expression in metastasis by murine prostate carcinoma. Am J Pathol 1998; 152: 591–596.
Gohji K, Fujimoto N, Hara I, Fujii A, Gotoh A, Okada H et al. Serum matrix metalloproteinase-2 and its density in men with prostate cancer as a new predictor of disease extension. Int J Cancer 1998; 79: 96–101.
Stearns M, Stearns ME . Evidence for increased activated metalloproteinase 2 (MMP-2a) expression associated with human prostate cancer progression. Oncology Research 1996; 8: 69–75.
Zhou LX, Li T, Huang YR, Sha JJ, Sun P, Li D . Application of histone modification in the risk prediction of the biochemical recurrence after radical prostatectomy. Asian J Androl 2010; 12: 171–179.
Qin H, Sun Y, Benveniste EN . The transcription factors Sp1, Sp3, and AP-2 are required for constitutive matrix metalloproteinase-2 gene expression in astroglioma cells. J Biol Chem 1999; 274: 29130–29137.
Xu Y, Zuo Y, Zhang H, Kang X, Yue F, Yi Z et al. Induction of SENP1 in endothelial cells contributes to hypoxia-driven VEGF expression and angiogenesis. J Biol Chem 2010; 285: 36682–36688.
Acknowledgements
We thank Dr E Benveniste (The University of Alabama at Birmingham) for the MMP2-Luc plasmid. This work was supported in part by National Natural Science Foundation of China (81071665 to JC, 30900740 to YZ), National Basic Research Program of China (973 Program) (2010CB912104 to JC), Shanghai Committee of Science and Technology (10140902000, 10410700900, 11XD1403200 to JC, 11DZ2260200), United States Department of Defense Grants (PC060932 to JC) and NIH (CA 239520 to ETHY). ETHY is the McNair Scholar of the Texas Heart Institute/St Luke’s Episcopal Hospital.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Wang, Q., Xia, N., Li, T. et al. SUMO-specific protease 1 promotes prostate cancer progression and metastasis. Oncogene 32, 2493–2498 (2013). https://doi.org/10.1038/onc.2012.250
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.250
Keywords
This article is cited by
-
The SUMO protease SENP1 promotes aggressive behaviors of high HIF2α expressing renal cell carcinoma cells
Oncogenesis (2022)
-
Human amniotic mesenchymal stem cells to promote/suppress cancer: two sides of the same coin
Stem Cell Research & Therapy (2021)
-
Autophagy provides a conceptual therapeutic framework for bone metastasis from prostate cancer
Cell Death & Disease (2021)
-
A SUMOylation-dependent HIF-1α/CLDN6 negative feedback mitigates hypoxia-induced breast cancer metastasis
Journal of Experimental & Clinical Cancer Research (2020)
-
Function and clinical significance of SUMOylation in type I endometrial carcinoma
Oncology and Translational Medicine (2017)