Abstract
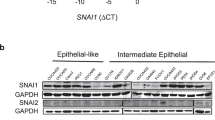
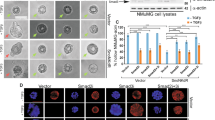
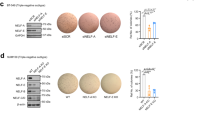
Epithelial-mesenchymal transition (EMT) has pivotal roles during embryonic development and carcinoma progression. Members of the Snai1 family of zinc finger transcription factors are central mediators of EMT and induce EMT in part by directly repressing epithelial markers such as E-cadherin, a gatekeeper of the epithelial phenotype and a suppressor of tumor invasion. However, the molecular mechanism underlying Snai1-mediated transcriptional repression remains incompletely understood. Here we show that Snai1 physically interacts with and recruits the histone demethylase LSD1 (KDM1A) to epithelial gene promoters. LSD1 removes dimethylation of lysine 4 on histone H3 (H3K4m2), a covalent histone modification associated with active chromatin. Importantly, LSD1 is essential for Snai1-mediated transcriptional repression and for maintenance of the silenced state of Snai1 target genes in invasive cancer cells. In the absence of LSD1, Snai1 fails to repress E-cadherin. In cancer cells in which E-cadherin is silenced, depletion of LSD1 results in partial de-repression of epithelial genes and elevated H3K4m2 levels at the E-cadherin promoter. These results underline the critical role of LSD1 in Snai1-dependent transcriptional repression of epithelial markers and suggest that the LSD1 complex could be a potential therapeutic target for prevention of EMT-associated tumor invasion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ao A, Wang H, Kamarajugadda S, Lu J . (2008). Involvement of estrogen-related receptors in transcriptional response to hypoxia and growth of solid tumors. Proc Natl Acad Sci USA 105: 7821–7826.
Batlle E, Sancho E, Francí C, Domínguez D, Monfar M, Baulida J et al. (2000). The transcription factor snail is a repressor of E-cadherin gene expression in epithelial tumour cells. Nat Cell Biol 2: 84–89.
Bernstein BE, Mikkelsen TS, Xie X, Kamal M, Huebert DJ, Cuff J et al. (2006). A bivalent chromatin structure marks key developmental genes in embryonic stem cells. Cell 125: 315–326.
Cano A, Pérez-Moreno MA, Rodrigo I, Locascio A, Blanco MJ, del Barrio MG et al. (2000). The transcription factor snail controls epithelial-mesenchymal transitions by repressing E-cadherin expression. Nat Cell Biol 2: 76–83.
De Craene B, Gilbert B, Stove C, Bruyneel E, van Roy F, Berx G . (2005). The transcription factor Snail induces tumor cell invasion through modulation of the epithelial cell differentiation program. Cancer Res 65: 6237–6244.
Francí C, Takkunen M, Dave N, Alameda F, Gómez S, Rodríguez R et al. (2006). Expression of Snail protein in tumor-stroma interface. Oncogene 25: 5134–5144.
Grimes HL, Chan TO, Zweidler-McKay PA, Tong B, Tsichlis PN . (1996). The Gfi-1 proto-oncoprotein contains a novel transcriptional repressor domain, SNAG, and inhibits G1 arrest induced by interleukin-2 withdrawal. Mol Cell Biol 16: 6263–6272.
Hajra KM, Chen DY, Fearon ER . (2002). The SLUG zinc-finger protein represses E-cadherin in breast cancer. Cancer Res 62: 1613–1618.
Hay E . (1995). An overview of epithelio-mesenchymal transformation. Acta Anat 154: 8–20.
Herranz N, Pasini D, Díaz VM, Francí C, Gutierrez A, Dave N et al. (2008). Polycomb complex 2 is required for E-cadherin repression by the Snail1 transcription factor. Mol Cell Biol 28: 4772–4781.
Hou Z, Peng H, Ayyanathan K, Yan KP, Langer EM, Longmore GD et al. (2008). The LIM protein AJUBA recruits protein arginine methyltransferase 5 to mediate SNAIL-dependent transcriptional repression. Mol Cell Biol 28: 3198–3207.
Ikenouchi J, Matsuda M, Furuse M, Tsukita S . (2003). Regulation of tight junctions during the epithelium-mesenchyme transition: direct repression of the gene expression of claudins/occludin by Snail. J Cell Sci 116: 1959–1967.
Jenuwein T, Allis CD . (2001). Translating the histone code. Science 293: 1074–1080.
Klose RJ, Zhang Y . (2007). Regulation of histone methylation by demethylimination and demethylation. Nat Rev Mol Cell Biol 8: 307–318.
Kouzarides T . (2007). Chromatin modifications and their function. Cell 128: 693–705.
Lan F, Nottke AC, Shi Y . (2008). Mechanisms involved in the regulation of histone lysine demethylases. Curr Opin Cell Biol 20: 316–325.
Li B, Carey M, Workman JL . (2007). The role of chromatin during transcription. Cell 128: 707–719.
Lim S, Janzer A, Becker A, Zimmer A, Schüle R, Buettner R et al. (2010). Lysine-specific demethylase 1 (LSD1) is highly expressed in ER-negative breast cancers and a biomarker predicting aggressive biology. Carcinogenesis 31: 512–520.
Nieto MA . (2002). The Snail superfamily of zinc-finger transcription factors. Nat Rev Mol Cell Biol 3: 155–166.
Peinado H, Ballestar E, Esteller M, Cano A . (2004). Snail mediates E-cadherin repression by the recruitment of the Sin3A/histone deacetylase 1 (HDAC1)/HDAC2 complex. Mol Cell Biol 24: 306–319.
Peinado H, Olmeda D, Cano A . (2007). Snail, Zeb and bHLH factors in tumor progression: an alliance against the epithelial phenotype? Nat Rev Cancer 7: 415–428.
Perl AK, Wilgenbus P, Dahl U, Semb H, Christofori G . (1998). A causal role for E-cadherin in the transition from adenoma to carcinoma. Nature 392: 190–193.
Polyak K, Weinberg RA . (2009). Transitions between epithelial and mesenchymal states: acquisition of malignant and stem cell traits. Nat Rev Cancer 9: 265–273.
Saleque S, Kim J, Rooke HM, Orkin SH . (2007). Epigenetic regulation of hematopoietic differentiation by Gfi-1 and Gfi-1b is mediated by the cofactors CoREST and LSD1. Mol Cell 27: 562–572.
Schulte JH, Lim S, Schramm A, Friedrichs N, Koster J, Versteeg R et al. (2009). Lysine-specific demethylase 1 is strongly expressed in poorly differentiated neuroblastoma: implications for therapy. Cancer Res 69: 2065–2071.
Shi Y . (2007). Histone lysine demethylases: emerging roles in development, physiology and disease. Nat Rev Genet 8: 829–833.
Shi Y, Lan F, Matson C, Mulligan P, Whetstine JR, Cole PA et al. (2004). Histone demethylation mediated by the nuclear amine oxidase homolog LSD1. Cell 119: 941–953.
Thiery JP . (2002). Epithelial–mesenchymal transitions in tumor progression. Nature Rev Cancer 2: 442–454.
Thiery JP, Acloque H, Huang RY, Nieto MA . (2009). Epithelial-mesenchymal transitions in development and disease. Cell 139: 871–890.
Thiery JP, Sleeman JP . (2006). Complex networks orchestrate epithelial-mesenchymal transitions. Nat Rev Mol Cell Biol 7: 131–142.
Wang J, Scully K, Zhu X, Cai L, Zhang J, Prefontaine GG et al. (2007). Opposing LSD1 complexes function in developmental gene activation and repression programmes. Nature 446: 882–887.
Wang J, Hevi S, Kurash JK, Lei H, Gay F, Bajko J et al. (2009). The lysine demethylase LSD1 (KDM1) is required for maintenance of global DNA methylation. Nat Genet 41: 125–129.
Yang J, Weinberg RA . (2008). Epithelial-mesenchymal transition: at the crossroads of development and tumor metastasis. Dev Cell 14: 818–829.
Acknowledgements
We appreciate technical assistance from Linbao Ai and Heiman Wang. We are grateful to Mien-Chie Hung (MD Anderson Cancer Center, Texas, USA), M Angela Nieto (Instituto de Neurociencias CSIC-UMH, Spain), Suming Huang, Yi Qiu and Lizi Wu for kindly providing reagents. We thank Mike Kilberg and Jorg Bungert for critical reading of the paper. This study was supported by grants to JL from Stop! Children's Cancer, Florida Bankhead-Coley Cancer Research Program (09BN-12-23092), The National Cancer Institute (R01CA137021) and to AP from the University Scholars Program at University of Florida, Gainesville, FL, USA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Lin, T., Ponn, A., Hu, X. et al. Requirement of the histone demethylase LSD1 in Snai1-mediated transcriptional repression during epithelial-mesenchymal transition. Oncogene 29, 4896–4904 (2010). https://doi.org/10.1038/onc.2010.234
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.234
Keywords
This article is cited by
-
Ubiquitin-specific protease 28: the decipherment of its dual roles in cancer development
Experimental Hematology & Oncology (2023)
-
CHST2-mediated sulfation of MECA79 antigens is critical for breast cancer cell migration and metastasis
Cell Death & Disease (2023)
-
Epigenetic regulation of hybrid epithelial-mesenchymal cell states in cancer
Oncogene (2023)
-
Roles of lysine-specific demethylase 1 (LSD1) in homeostasis and diseases
Journal of Biomedical Science (2021)
-
FBXL10 promotes EMT and metastasis of breast cancer cells via regulating the acetylation and transcriptional activity of SNAI1
Cell Death Discovery (2021)