Abstract
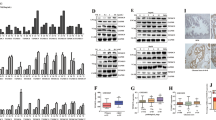
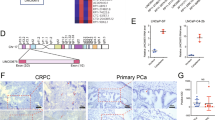
There are few successful therapies for castration-resistant prostate cancer (CRPC). Recently, CRPC has been thought to result from augmented androgen/androgen receptor (AR) signaling pathway, for most of which AR overexpression has been observed. In this study, Twist1, a member of basic helix-loop-helix transcription factors as well as AR was upregulated in response to hydrogen peroxide, and the response to which was abolished by an addition of N-acetyl-L-cysteine and Twist1 knockdown. In addition, castration-resistant LNCaP derivatives and hydrogen peroxide-resistant LNCaP derivatives exhibited a similar phenotype to each other. Then, both castration and AR knockdown increased intracellular reactive oxygen species level. Moreover, Twist1 was shown to regulate AR expression through binding to E-boxes in AR promoter region. Silencing of Twist1 suppressed cell growth of AR-expressing LNCaP cells as well as castration-resistant LNCaP derivatives by inducing cell-cycle arrest at G1 phase and cellular apoptosis. These findings indicated that castration-induced oxidative stress may promote AR overexpression through Twist1 overexpression, which could result in a gain of castration resistance. Modulation of castration-induced oxidative stress or Twist1/AR signaling might be a useful strategy for developing a novel therapeutics in prostate cancer, even in CRPC, which remains dependent on AR signaling by overexpressing AR.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Benz CC, Yau C . (2008). Ageing, oxidative stress and cancer: paradigms in parallax. Nat Rev Cancer 8: 875–879.
Best CJ, Gillespie JW, Yi Y, Chandramouli GV, Perlmutter MA, Gathright Y et al. (2005). Molecular alterations in primary prostate cancer after androgen ablation therapy. Clin Cancer Res 11: 6823–6834.
Billis A . (1996). Age and race distribution of high-grade prostatic intraepithelial neoplasia: an autopsy study in Brazil (South America). J Urol Pathol 5: 1–7.
Bostwick DG, Alexander EE, Singh R, Shan A, Qian J, Santella RM et al. (2000). Antioxidant enzyme expression and reactive oxygen species damage in prostatic intraepithelial neoplasia and cancer. Cancer 89: 123–134.
Castanon I, Setina SV, Kass J, Baylies MK . (2001). Dimerization partners determine the activity of the Twist bHLH protein during Drosophila mesoderm development. Development 128: 3145–3159.
Chen CD, Welsbie DS, Tran C, Baek SH, Chen R, Vessella R et al. (2004). Molecular determinants of resistance to antiandrogen therapy. Nat Med 10: 33–39.
Debes JD, Tindall DJ . (2002). The role of androgens and the androgen receptor in prostate cancer. Cancer Lett 187: 1–7.
Ghatak S, Ho SM . (1996). Age-related changes in the activities of antioxidant enzymes and lipid peroxidation status in ventral and dorsolateral prostate lobes of Noble rats. Biochem Biophys Res Commun 222: 362–367.
Gregory CW, Hamil KG, Kim D, Hall SH, Pretlow TG, Mohler JL et al. (1998). Androgen receptor expression in androgen-independent prostate cancer is associated with increased expression of androgen-regulated genes. Cancer Res 58: 5718–5724.
Gronberg H . (2003). Prostate cancer epidemiology. Lancet 361: 859–864.
Hakimian P, Blute Jr M, Kashanian J, Chan S, Silver D, Shabsigh R . (2008). Metabolic and cardiovascular effects of androgen deprivation therapy. BJU Int 102: 1509–1514.
Hoek K, Rimm DL, Williams KR, Zhao H, Ariyan S, Lin A et al. (2004). Expression profiling reveals novel pathways in the transformation of melanocytes to melanomas. Cancer Res 64: 5270–5282.
Hsing AW, Devesa SS . (2001). Trends and patterns of prostate cancer: what do they suggest? Epidemiol Rev 23: 3–13.
Ishikawa K, Takenaga K, Akimoto M, Koshikawa N, Yamaguchi A, Imanishi H et al. (2008). ROS-generating mitochondrial DNA mutations can regulate tumor cell metastasis. Science 320: 661–664.
Kwok WK, Ling MT, Lee TW, Lau TC, Zhou C, Zhang X et al. (2005). Up-regulation of TWIST in prostate cancer and its implication as a therapeutic target. Cancer Res 65: 5153–5162.
Landies SH, Murray T, Bolden S, Wingo PA . (1999). Cancer statistics. CA Cancer J Clin 49: 8–31.
Lee DK, Chang C . (2003). Expression and degradation of androgen receptor: mechanism and clinical implication. J Clin Endocrinol Metab 88: 4043–4054.
Linja MJ, Savinainen KJ, Saramaki OR, Tammela TL, Vessella RL, Visakorpi T . (2001). Amplification and overexpression of androgen receptor gene in hormone-refractory prostate cancer. Cancer Res 61: 3550–3555.
Lu T, Finkel T . (2008). Free radicals and senescence. Exp Cell Res 314: 1918–1922.
Maestro R, Dei Tos AP, Hamamori Y, Krasnokutsky S, Sartorelli V, Kedes L et al. (1999). Twist is a potential oncogene that inhibits apoptosis. Genes Dev 13: 2207–2217.
Maynard S, Schurman SH, Harboe C, de Souza-Pinto NC, Bohr VA . (2009). Base excision repair of oxidative DNA damage and association with cancer and aging. Carcinogenesis 30: 2–10.
Mitsiades N, Correa D, Gross CP, Hurria A, Slovin SF . (2008). Cognitive effects of hormonal therapy in older adults. Semin Oncol 35: 569–581.
Oberley TD, Zhong W, Szweda LI, Oberley LW . (2000). Localization of antioxidant enzymes and oxidative damage products in normal and malignant prostate epithelium. Prostate 44: 144–155.
Olson EN, Klein WH . (1994). bHLH factors in muscle development: dead lines and commitments, what to leave in and what to leave out. Genes Dev 8: 1–8.
Pang ST, Dillner K, Wu X, Pousette A, Norstedt G, Flores-Morales A . (2002). Gene expression profiling of androgen deficiency predicts a pathway of prostate apoptosis that involves genes related to oxidative stress. Endocrinology 143: 4897–4906.
Patel BJ, Pantuck AJ, Zisman A, Tsui KH, Paik SH, Caliliw R et al. (2000). CL1-GFP: an androgen independent metastatic tumor model for prostate cancer. J Urol 164: 1420–1425.
Pathak S, Singh R, Verschoyle RD, Greaves P, Farmer PB, Steward WP et al. (2008). Androgen manipulation alters oxidative DNA adduct levels in androgen-sensitive prostate cancer cells grown in vitro and in vivo. Cancer Lett 261: 74–83.
Pinthus JH, Bryskin I, Trachtenberg J, Lu JP, Singh G, Fridman E et al. (2007). Androgen induces adaptation to oxidative stress in prostate cancer: implications for treatment with radiation therapy. Neoplasia 9: 68–80.
Reddy GP, Barrack ER, Dou QP, Menon M, Pelley R, Sarkar FH et al. (2006). Regulatory processes affecting androgen receptor expression, stability, and function: potential targets to treat hormone-refractory prostate cancer. J Cell Biochem 98: 1408–1423.
Ripple MO, Henry WF, Rago RP, Wilding G . (1997). Prooxidant-antioxidant shift induced by androgen treatment of human prostate carcinoma cells. J Natl Cancer Inst 89: 40–48.
Rosivatz E, Becker I, Specht K, Fricke E, Luber B, Busch R et al. (2002). Differential expression of the epithelial-mesenchymal transition regulators snail, SIP1, and twist in gastric cancer. Am J Pathol 161: 1881–1891.
Sakr WA, Grignon DJ, Haas GP, Heilbrun LK, Pontes JE, Crissman JD . (1996). Age and racial distribution of prostatic intraepithelial neoplasia. Eur Urol 30: 138–144.
Scher HI, Sawyers CL . (2006). Biology of progressive, castration-resistant prostate cancer: directed therapies targeting the androgen-receptor signaling axis. J Clin Oncol 23: 8253–8261.
Shiota M, Izumi H, Onitsuka T, Miyamoto N, Kashiwagi E, Kidani A et al. (2008a). Twist promotes tumor cell growth through YB-1 expression. Cancer Res 68: 98–105.
Shiota M, Izumi H, Onitsuka T, Miyamoto N, Kashiwagi E, Kidani A et al. (2008b). Twist and p53 reciprocally regulate target genes via direct interaction. Oncogene 27: 5543–5553.
Shiota M, Izumi H, Tanimoto A, Takahashi M, Miyamoto N, Kashiwagi E et al. (2009). Programmed cell death protein 4 down-regulates Y-box binding protein-1 expression via a direct interaction with Twist1 to suppress cancer cell growth. Cancer Res 69: 3148–3156.
Stasinopoulos IA, Mironchik Y, Raman A, Wildes F, Winnard P, Raman V . (2005). HOXA5-Twist interaction alters p53 homeostasis in breast cancer cells. J Biol Chem 280: 2294–2299.
Tamm NN, Gao Y, Leung YK, Ho SM . (2003). Androgenic regulation of oxidative stress in the rat prostate: involvement of NAD(P)H oxidases and antioxidant defense machinery during prostatic involution and regrowth. Am J Pathol 163: 2513–2522.
Taplin ME, Rajeshkumar B, Halabi S, Werner CP, Woda BA, Picus J et al. (2003). Androgen receptor mutations in androgen-independent prostate cancer: Cancer and Leukemia Group B Study 9663. J Clin Oncol 21: 2673–2678.
Tso CL, McBride WH, Sun J, Patel B, Tsui KH, Paik SH et al. (2000). Androgen deprivation induces selective outgrowth of aggressive hormone-refractory prostate cancer clones expressing distinct cellular and molecular properties not present in parental androgen-dependent cancer cells. Cancer J 6: 220–233.
Valsesia-Wittmann S, Magdeleine M, Dupasquier S, Garin E, Jallas AC, Combaret V et al. (2004). Oncogenic cooperation between H-Twist and N-Myc overrides failsafe programs in cancer cells. Cancer Cell 6: 625–630.
van Doorn R, Dijkman R, Vermeer MH, Out-Luiting JJ, van der Raaij-Helmer EM, Willemze R et al. (2004). Aberrant expression of the tyrosine kinase receptor EphA4 and the transcription factor twist in Sezary syndrome identified by gene expression analysis. Cancer Res 64: 5578–5586.
Yang J, Mani SA, Donaher JL, Ramaswamy S, Itzykson RA, Come C et al. (2004). Twist, a master regulator of morphogenesis, plays an essential role in tumor metastasis. Cell 117: 927–939.
Yang MH, Wu MZ, Chiou SH, Chen PM, Chang SY, Liu CJ et al. (2008). Direct regulation of TWIST by HIF-1α promotes metastasis. Nat Cell Biol 10: 295–305.
Zegarra-Moro OL, Schmidt LJ, Huang H, Tindall DJ . (2002). Disruption of androgen function inhibits proliferation of androgen-refractory prostate cancer cells. Cancer Res 62: 1008–1013.
Acknowledgements
This work was supported in part by Health Sciences Research Grants for Clinical Research for evidence-based medicine and grants-in-aid for Cancer Research (016), from the Ministry of Health, Labor and Welfare, Japan, Young Researcher Promotion Grant from the Japanese Urological Association, Japan. We are grateful to Dr Chawnshang Chang for providing the pCMV-AR plasmid. We would like to thank Dr Dongchon Kang (Kyushu University, Fukuoka, Japan) for helping with quantitative real-time PCR and flow cytometry, and Noriko Hakoda, Hitomi Matoba and Seiko Kamori for their technical assistances.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shiota, M., Yokomizo, A., Tada, Y. et al. Castration resistance of prostate cancer cells caused by castration-induced oxidative stress through Twist1 and androgen receptor overexpression. Oncogene 29, 237–250 (2010). https://doi.org/10.1038/onc.2009.322
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.322
Keywords
This article is cited by
-
PPFIA4 promotes castration-resistant prostate cancer by enhancing mitochondrial metabolism through MTHFD2
Journal of Experimental & Clinical Cancer Research (2022)
-
GSTM2 is a key molecular determinant of resistance to SG-ARIs
Oncogene (2022)
-
ISL1 promotes enzalutamide resistance in castration-resistant prostate cancer (CRPC) through epithelial to mesenchymal transition (EMT)
Scientific Reports (2021)
-
Independence of HIF1a and androgen signaling pathways in prostate cancer
BMC Cancer (2020)
-
TXNDC9 regulates oxidative stress-induced androgen receptor signaling to promote prostate cancer progression
Oncogene (2020)