Abstract
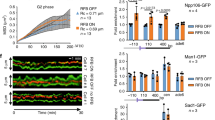
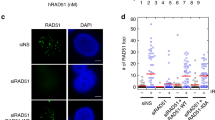
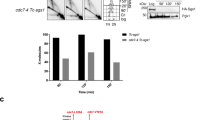
The role of Rad51 in an unperturbed cell cycle has been difficult to distinguish from its DNA repair function. Here, using EM to visualize replication intermediates assembled in Xenopus laevis egg extract, we show that Rad51 is required to prevent the accumulation of single-stranded DNA (ssDNA) gaps at replication forks and behind them. ssDNA gaps at forks arise from extended uncoupling of leading- and lagging-strand DNA synthesis. In contrast, ssDNA gaps behind forks, which are prevalent on damaged templates, result from Mre11-dependent degradation of newly synthesized DNA strands and are suppressed by inhibition of Mre11 nuclease activity. These findings reveal direct roles for Rad51 at replication forks, demonstrating that Rad51 protects newly synthesized DNA from Mre11-dependent degradation and promotes continuous DNA synthesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Branzei, D. & Foiani, M. Maintaining genome stability at the replication fork. Nat. Rev. Mol. Cell Biol. 11, 208–219 (2010).
Lambert, S., Froget, B. & Carr, A.M. Arrested replication fork processing: interplay between checkpoints and recombination. DNA Repair (Amst.) 6, 1042–1061 (2007).
Prakash, S., Johnson, R.E. & Prakash, L. Eukaryotic translesion synthesis DNA polymerases: specificity of structure and function. Annu. Rev. Biochem. 74, 317–353 (2005).
Moldovan, G.L., Pfander, B. & Jentsch, S. PCNA, the maestro of the replication fork. Cell 129, 665–679 (2007).
West, S.C. Molecular views of recombination proteins and their control. Nat. Rev. Mol. Cell Biol. 4, 435–445 (2003).
Tsuzuki, T. et al. Targeted disruption of the Rad51 gene leads to lethality in embryonic mice. Proc. Natl. Acad. Sci. USA 93, 6236–6240 (1996).
Sonoda, E. et al. Rad51-deficient vertebrate cells accumulate chromosomal breaks prior to cell death. EMBO J. 17, 598–608 (1998).
Aguilera, A. & Gomez-Gonzalez, B. Genome instability: a mechanistic view of its causes and consequences. Nat. Rev. Genet. 9, 204–217 (2008).
Lambert, S., Watson, A., Sheedy, D.M., Martin, B. & Carr, A.M. Gross chromosomal rearrangements and elevated recombination at an inducible site-specific replication fork barrier. Cell 121, 689–702 (2005).
Alabert, C., Bianco, J.N. & Pasero, P. Differential regulation of homologous recombination at DNA breaks and replication forks by the Mrc1 branch of the S-phase checkpoint. EMBO J. 28, 1131–1141 (2009).
Lehmann, A.R. & Fuchs, R.P. Gaps and forks in DNA replication: Rediscovering old models. DNA Repair (Amst.) 5, 1495–1498 (2006).
Lopes, M., Foiani, M. & Sogo, J.M. Multiple mechanisms control chromosome integrity after replication fork uncoupling and restart at irreparable UV lesions. Mol. Cell 21, 15–27 (2006).
Heller, R.C. & Marians, K.J. Replisome assembly and the direct restart of stalled replication forks. Nat. Rev. Mol. Cell Biol. 7, 932–943 (2006).
Petermann, E. & Caldecott, K.W. Evidence that the ATR/Chk1 pathway maintains normal replication fork progression during unperturbed S phase. Cell Cycle 5, 2203–2209 (2006).
Moriel-Carretero, M. & Aguilera, A. A postincision-deficient TFIIH causes replication fork breakage and uncovers alternative Rad51- or Pol32-mediated restart mechanisms. Mol. Cell 37, 690–701 (2010).
Hekmat-Nejad, M., You, Z., Yee, M.C., Newport, J.W. & Cimprich, K.A. X. laevis ATR is a replication-dependent chromatin-binding protein required for the DNA replication checkpoint. Curr. Biol. 10, 1565–1573 (2000).
Trenz, K., Smith, E., Smith, S. & Costanzo, V. ATM and ATR promote Mre11 dependent restart of collapsed replication forks and prevent accumulation of DNA breaks. EMBO J. 25, 1764–1774 (2006).
Costanzo, V. et al. Mre11 protein complex prevents double-strand break accumulation during chromosomal DNA replication. Mol. Cell 8, 137–147 (2001).
Trenz, K., Errico, A. & Costanzo, V. Plx1 is required for chromosomal DNA replication under stressful conditions. EMBO J. 27, 876–885 (2008).
McGarry, T.J. & Kirschner, M.W. Geminin, an inhibitor of DNA replication, is degraded during mitosis. Cell 93, 1043–1053 (1998).
Hashimoto, Y. & Takisawa, H. X. laevisCut5 is essential for a CDK-dependent process in the initiation of DNA replication. EMBO J. 22, 2526–2535 (2003).
Thorslund, T. & West, S.C. BRCA2: a universal recombinase regulator. Oncogene 26, 7720–7730 (2007).
Carreira, A. et al. The BRC repeats of BRCA2 modulate the DNA-binding selectivity of RAD51. Cell 136, 1032–1043 (2009).
Leach, C.A. & Michael, W.M. Ubiquitin/SUMO modification of PCNA promotes replication fork progression in X. laevis egg extracts. J. Cell Biol. 171, 947–954 (2005).
Stokes, M.P. & Michael, W.M. DNA damage-induced replication arrest in X. laevis egg extracts. J. Cell Biol. 163, 245–255 (2003).
Su, X., Bernal, J.A. & Venkitaraman, A.R. Cell-cycle coordination between DNA replication and recombination revealed by a vertebrate N-end rule degron-Rad51. Nat. Struct. Mol. Biol. 15, 1049–1058 (2008).
Fukui, T. et al. Distinct roles of DNA polymerases delta and epsilon at the replication fork in X. laevis egg extracts. Genes Cells 9, 179–191 (2004).
Tsujikawa, L., Weinfield, M. & Reha-Krantz, L.J. Differences in replication of a DNA template containing an ethyl phosphotriester by T4 DNA polymerase and Escherichia coli DNA polymerase I. Nucleic Acids Res. 31, 4965–4972 (2003).
Nagaraju, G. & Scully, R. Minding the gap: the underground functions of BRCA1 and BRCA2 at stalled replication forks. DNA Repair (Amst.) 6, 1018–1031 (2007).
Lopes, M. Electron microscopy methods for studying in vivo DNA replication intermediates. Methods Mol. Biol. 521, 605–631 (2009).
Sogo, J.M., Lopes, M. & Foiani, M. Fork reversal and ssDNA accumulation at stalled replication forks owing to checkpoint defects. Science 297, 599–602 (2002).
Symington, L.S. Role of RAD52 epistasis group genes in homologous recombination and double-strand break repair. Microbiol. Mol. Biol. Rev. 66, 630–670 (2002).
Petrini, J.H., Bressan, D.A. & Yao, M.S. The RAD52 epistasis group in mammalian double strand break repair. Semin. Immunol. 9, 181–188 (1997).
Krogh, B.O. & Symington, L.S. Recombination proteins in yeast. Annu. Rev. Genet. 38, 233–271 (2004).
Dupré, A. et al. A forward chemical genetic screen reveals an inhibitor of the Mre11–Rad50–Nbs1 complex. Nat. Chem. Biol. 4, 119–125 (2008).
Petrini, J.H. S-phase functions of the Mre11 complex. Cold Spring Harb. Symp. Quant. Biol. 65, 405–411 (2000).
Tashiro, S. et al. S phase specific formation of the human Rad51 protein nuclear foci in lymphocytes. Oncogene 12, 2165–2170 (1996).
Tarsounas, M., Davies, D. & West, S.C. BRCA2-dependent and independent formation of RAD51 nuclear foci. Oncogene 22, 1115–1123 (2003).
McIlwraith, M.J. et al. Human DNA polymerase eta promotes DNA synthesis from strand invasion intermediates of homologous recombination. Mol. Cell 20, 783–792 (2005).
Schlacher, K., Cox, M.M., Woodgate, R. & Goodman, M.F. RecA acts in trans to allow replication of damaged DNA by DNA polymerase V. Nature 442, 883–887 (2006).
Reuven, N.B., Arad, G., Stasiak, A.Z., Stasiak, A. & Livneh, Z. Lesion bypass by the Escherichia coli DNA polymerase V requires assembly of a RecA nucleoprotein filament. J. Biol. Chem. 276, 5511–5517 (2001).
Compton, S.A., Ozgur, S. & Griffith, J.D. Ring-shaped Rad51 paralog protein complexes bind Holliday junctions and replication forks as visualized by electron microscopy. J. Biol. Chem. 285, 13349–13356 (2010).
Sung, P. & Klein, H. Mechanism of homologous recombination: mediators and helicases take on regulatory functions. Nat. Rev. Mol. Cell Biol. 7, 739–750 (2006).
Barber, L.J. et al. RTEL1 maintains genomic stability by suppressing homologous recombination. Cell 135, 261–271 (2008).
Lisby, M. & Rothstein, R. Choreography of recombination proteins during the DNA damage response. DNA Repair (Amst.) 8, 1068–1076 (2009).
Gibson, F.P., Leach, D.R. & Lloyd, R.G. Identification of sbcD mutations as cosuppressors of recBC that allow propagation of DNA palindromes in Escherichia coli K-12. J. Bacteriol. 174, 1222–1228 (1992).
Eykelenboom, J.K., Blackwood, J.K., Okely, E. & Leach, D.R. SbcCD causes a double-strand break at a DNA palindrome in the Escherichia coli chromosome. Mol. Cell 29, 644–651 (2008).
Acknowledgements
We thank S. West, S. Boulton and members of the genome stability lab for their insightful comments, H. Mahbubani, J. Kirk for technical support with X. laevis and the Center for Microscopy and Image Analysis of the University of Zurich for technical assistance with electron microscopy. This work was funded by Cancer Research UK. V.C. is also supported by the European Research Council start-up grant (206281), the Lister Institute of Preventive Medicine and the European Molecular Biology Organization (EMBO) Young Investigator Program. M.L. and A.R.C. are supported by the Swiss National Science Foundation grant (PP00A-114922). A.R.C. was also supported by an EMBO short-term fellowship. Recombinant human Rad51 proteins and the cDNA fragment encoding human BRC4 (residues 1511–1579 of BRCA2) cloned into pDONR221 were provided by F. Esashi (Oxford University). The pET28-based expression vectors of wild-type and mutant (K164R) X. laevis PCNA and the pET21-based expression vector of human p27 were provided by H. Ulrich (Cancer Research UK) and T. Hunt (Cancer Research UK), respectively. Mirin was provided by J. Gautier (Columbia University). Antibodies to Mcm2, Cdc45, Psf2 and Pol ɛ p60 subunit were provided by H. Takisawa (Osaka University); antibodies to Pol δ p125 subunit and Pol η were provided by S. Waga (Japan Women's University) and M. Akiyama (Nara Institute of Technology).
Author information
Authors and Affiliations
Contributions
Y.H. and A.R.C. carried out experiments; M.L. and V.C. conceived experiments and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 (PDF 5173 kb)
Rights and permissions
About this article
Cite this article
Hashimoto, Y., Ray Chaudhuri, A., Lopes, M. et al. Rad51 protects nascent DNA from Mre11-dependent degradation and promotes continuous DNA synthesis. Nat Struct Mol Biol 17, 1305–1311 (2010). https://doi.org/10.1038/nsmb.1927
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1927
This article is cited by
-
FLIP(C1orf112)-FIGNL1 complex regulates RAD51 chromatin association to promote viability after replication stress
Nature Communications (2024)
-
Structural and functional insights into the epigenetic regulator MRG15
Acta Pharmacologica Sinica (2024)
-
Polymerase iota (POLI) confers radioresistance of esophageal squamous cell carcinoma by regulating RAD51 stability and facilitating homologous recombination
Cell Death Discovery (2023)
-
Multi-step processing of replication stress-derived nascent strand DNA gaps by MRE11 and EXO1 nucleases
Nature Communications (2023)
-
Replication fork uncoupling causes nascent strand degradation and fork reversal
Nature Structural & Molecular Biology (2023)