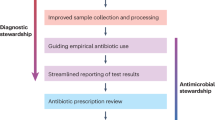
Key Points
-
Rates of infectious complications after transrectal prostate biopsies have increased over the past 15 years
-
The predominant risk factor for postbiopsy infection seems to be exposure to faecal fluoroquinolone-resistant bacteria
-
Every patient should undergo a preoperative assessment, including history of fluoroquinolone intake over the past 6 months, and a urine culture
-
Choice of empirical antibiotic prophylaxis should take regional resistance rates into consideration, as well as the pharmacokinetic and pharmacodynamic characteristics of each antibiotic
-
Novel strategies to reduce rates of infectious complications after transrectal prostate biopsies include targeted prophylaxis after sampling of the rectal flora, bowel preparation, and perineal prostate biopsy
Abstract
Over the years, prostate biopsy has become the gold-standard technique for diagnosing prostate carcinoma. Worldwide, several million prostate biopsies are performed every year, most commonly using the transrectal approach. Preoperative antibiotic prophylaxis with fluoroquinolones has been shown to be effective for reducing infection rates. However, in recent years, an increase in febrile infection rates after transrectal prostate biopsy (from 1% to 4%) has been reported in retrospective and prospective studies. The predominant risk factor for infection seems to be the presence of fluoroquinolone-resistant bacteria in faeces. Patients at risk of fluoroquinolone resistance should receive carefully selected antibiotics at sufficient concentrations to be effective. Targeted prophylaxis after rectal flora swabbing has been shown to be efficacious compared with empirical antibiotic prophylaxis. Several forms of bowel preparations are under investigation, although none have yet been shown to significantly reduce infection rates. Perineal prostate biopsy is currently being evaluated as a strategy for preventing the inoculation of rectal flora, but limited data support this approach at present.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Zani, E. L., Clark, O. A. & Rodrigues Netto, N. Jr. Antibiotic prophylaxis for transrectal prostate biopsy. Cochrane Database of Systematic Reviews, Issue 5. Art. No.: CD006576. http://dx.doi.org/10.1002/14651858.CD006576.pub2 (2011).
Carmignani, L. et al. Bacterial sepsis following prostatic biopsy. Int. Urol. Nephrol. 44, 1055–1063 (2012).
Liss, M. A. et al. Prevalence and significance of fluoroquinolone resistant Escherichia coli in patients undergoing transrectal ultrasound guided prostate needle biopsy. J. Urol. 185, 1283–1288 (2011).
Loeb, S., Carter, H. B., Berndt, S. I., Ricker, W. & Schaeffer, E. M. Complications after prostate biopsy: data from SEER-Medicare. J. Urol. 186, 1830–1834 (2011).
Loeb, S. et al. Infectious complications and hospital admissions after prostate biopsy in a European randomized trial. Eur. Urol. 61, 1110–1114 (2012).
Nam, R. K. et al. Increasing hospital admission rates for urological complications after transrectal ultrasound guided prostate biopsy. J. Urol. 183, 963–968 (2010).
Grabe, M. et al. Preoperative assessment of the patient and risk factors for infectious complications and tentative classification of surgical field contamination of urological procedures. World J. Urol. 30, 39–50 (2012).
Lindstedt, S., Lindstrom, U., Ljunggren, E., Wullt, B. & Grabe, M. Single-dose antibiotic prophylaxis in core prostate biopsy: Impact of timing and identification of risk factors. Eur. Urol. 50, 832–837 (2006).
Loeb, S. Infection after transrectal ultrasonography-guided prostate biopsy: increased relative risks after recent international travel or antibiotic use. BJU Int. 109, 1785–1786 (2012).
Loeb, S. et al. Systematic review of complications of prostate biopsy. Eur. Urol. 64, 876–892 (2013).
Simsir, A., Kismali, E., Mammadov, R., Gunaydin, G. & Cal, C. Is it possible to predict sepsis, the most serious complication in prostate biopsy? Urol. Int. 84, 395–399 (2010).
Wagenlehner, F. M. et al. Infective complications after prostate biopsy: outcome of the global prevalence study of infections in urology (GPIU) 2010 and 2011; a prospective multinational multicentre prostate biopsy study. Eur. Urol. 63, 521–527 (2013).
Loeb, S., Carter, H. B., Berndt, S. I., Ricker, W. & Schaeffer, E. M. Is repeat prostate biopsy associated with a greater risk of hospitalization? Data from SEER-Medicare. J. Urol. 189, 867–870 (2013).
Nam, R. K. et al. Increasing hospital admission rates for urological complications after transrectal ultrasound guided prostate biopsy. J. Urol. 189, S12–S18 (2013).
Mouraviev, V. et al. The feasibility of multiparametric magnetic resonance imaging for targeted biopsy using novel navigation systems to detect early stage prostate cancer: the preliminary experience. J. Endourol. 27, 820–825 (2013).
Pepe, P. & Aragona, F. Morbidity after transperineal prostate biopsy in 3,000 patients undergoing 12 vs 18 vs more than 24 needle cores. Urology 81, 1142–1146 (2013).
Batura, D. & Gopal Rao, G. The national burden of infections after prostate biopsy in England and Wales: a wake-up call for better prevention. J. Antimicrob. Chemother. 68, 247–249 (2013).
Heidenreich, A. et al. EAU Guidelines on Prostate Cancer. Part II: Treatment of Advanced, Relapsing, and Castration-Resistant Prostate Cancer. Eur. Urol. http://dx.doi.org/10.1016/j.eururo.2013.11.002
Wirth, M. P. et al. Interdisziplinäre Leitlinie der Qualität S3 zur Früherkennung, Diagnose und Therapie der verschiedenen Stadien des Prostatakarzinoms: AWMF-Register-Nummer (043–022OL) Version 2.0 – 1. Aktualisierung 2011 [online], (2011).
Scardino, P. T. The responsible use of antibiotics for an elevated PSA level. Nat. Clin. Pract. Urol. 4, 1 (2007).
Ballentine Carter, H. et al. Early detection of prostate cancer: AUA guideline. J. Urol. 190, 419–426 (2013).
Carignan, A. et al. Increasing risk of infectious complications after transrectal ultrasound-guided prostate biopsies: time to reassess antimicrobial prophylaxis? Eur. Urol. 62, 453–459 (2012).
Williamson, D. A. et al. Escherichia coli bloodstream infection after transrectal ultrasound-guided prostate biopsy: implications of fluoroquinolone-resistant sequence type 131 as a major causative pathogen. Clin. Infect. Dis. 54, 1406–1412 (2012).
Steensels, D. et al. Fluoroquinolone-resistant E. coli in intestinal flora of patients undergoing transrectal ultrasound-guided prostate biopsy--should we reassess our practices for antibiotic prophylaxis? Clin. Microbiol. Infect. 18, 575–581 (2012).
Patel, U. et al. Infection after transrectal ultrasonography-guided prostate biopsy: increased relative risks after recent international travel or antibiotic use. BJU Int. 109, 1781–1785 (2011).
Williamson, D. A., Masters, J., Freeman, J. & Roberts, S. Travel-associated extended-spectrum β-lactamase-producing Escherichia coli bloodstream infection following transrectal ultrasound-guided prostate biopsy. BJU Int. 109, E21–E22 (2012).
Zaytoun, O. M. et al. Emergence of fluoroquinolone-resistant Escherichia coli as cause of postprostate biopsy infection: implications for prophylaxis and treatment. Urology 77, 1035–1041 (2011).
Grabe, M. et al. Guidelines on urological infections: European Association of Urology Guidelines 224–241 (European Association of Urology, Arnhem, 2013).
Gonzalez, C. M. et al. AUA/SUNA white paper on the incidence, prevention and treatment of complications related to prostate needle biopsy. AUA [online], (2013).
Laupland, K. B., Gregson, D. B., Church, D. L., Ross, T. & Pitout, J. D. Incidence, risk factors and outcomes of Escherichia coli bloodstream infections in a large Canadian region. Clin. Microbiol. Infect. 14, 1041–1047 (2008).
Puig, J. et al. Transrectal ultrasound-guided prostate biopsy: is antibiotic prophylaxis necessary? Eur. Radiol. 16, 939–943 (2006).
Bootsma, A. M., Laguna Pes, M. P., Geerlings, S. E. & Goossens, A. Antibiotic prophylaxis in urologic procedures: a systematic review. Eur. Urol. 54, 1270–1286 (2008).
Wagenlehner, F. M. et al. [Antibiotic prophylaxis in urology]. Urologe A 50, 1469–1480 (2011).
European Centre for Disease Prevention and Control (ECDC). Proportion of aminopenicillins resistant (R+I) enterococcus faecalis isolates in participating countries in 2012. ECDC [online], (2011).
Al-Hasan, M. N., Lahr, B. D., Eckel-Passow, J. E. & Baddour, L. M. Antimicrobial resistance trends of Escherichia coli bloodstream isolates: a population-based study, 1998–2007. J. Antimicrob. Chemother. 64, 169–174 (2009).
Tandogdu, Z. et al. Resistance patterns of nosocomial urinary tract infections in urology departments: 8-year results of the global prevalence of infections in urology study. World J. Urol. http://dx.doi.org/10.1007/s00345-013-1154-8
European Centre for Disease Prevention and Control (ECDC). ECDC Risk assessment on the spread of carbapenemase-producing Enterobacteriaceae (CPE) through patient transfer between healthcare facilities, with special emphasis on cross-border transfer. ECDC [online], (2011).
Qi, C. et al. Characterization of ciprofloxacin-resistant escherichia coli isolates among men undergoing evaluation for transrectal ultrasound-guided prostate biopsy: prevalence, clonality, and mechanisms of antimicrobial resistance. J. Urol. 190, 2026–2032 (2013).
Roach, M. B., Figueroa, T. E., McBride, D., George, W. J. & Neal, D. E. Jr. Ciprofloxacin versus gentamicin in prophylaxis against bacteraemia in transrectal prostate needle biopsy. Urology 38, 84–87 (1991).
Kataoka, N. [Concentration of antimicrobial agents in male genital organs]. Hinyokika Kiyo 29, 1219–1230 (1983).
Wagenlehner, F. M., Weidner, W., Sorgel, F. & Naber, K. G. The role of antibiotics in chronic bacterial prostatitis. Int. J. Antimicrob. Agents 26, 1–7 (2005).
Barza, M. & Cuchural, G. The penetration of antibiotics into the prostate in chronic bacterial prostatitis. Eur. J. Clin. Microbiol. 3, 503–505 (1984).
Naber, K. G. & Sorgel, F. Antibiotic therapy—rationale and evidence for optimal drug concentrations in prostatic and seminal fluid and in prostatic tissue. Andrologia 35, 331–335 (2003).
Goto, T. et al. Diffusion of piperacillin, cefotiam, minocycline, amikacin and ofloxacin into the prostate. Int. J. Urol. 5, 243–246 (1998).
Naber, K. G. et al. Concentrations of cefpodoxime in plasma, ejaculate and in prostatic fluid and adenoma tissue. Infection 19, 30–35 (1991).
Nishikawa, G. et al. Prostatic penetration of meropenem in humans, and dosage considerations for prostatitis based on a site-specific pharmacokinetic/pharmacodynamic evaluation. Int. J. Antimicrob. Agents 41, 267–271 (2013).
Nakamura, K. et al. Determination of doripenem penetration into human prostate tissue and assessment of dosing regimens for prostatitis based on site-specific pharmacokinetic-pharmacodynamic evaluation. J. Chemother. 24, 32–37 (2012).
Wright, W. L., Larking, P. & Lovell-Smith, C. J. Concentrations of trimethoprim and sulphamethoxazole in the human prostate gland after intramuscular injection. Br. J. Urol. 54, 550–551 (1982).
Naber, K. G., Roscher, K., Botto, H. & Schaefer, V. Oral levofloxacin 500 mg once daily in the treatment of chronic bacterial prostatitis. Int. J. Antimicrob. Agents 32, 145–153 (2008).
Ongun, S., Aslan, G. & Avkan-Oguz, V. The effectiveness of single-dose fosfomycin as antimicrobial prophylaxis for patients undergoing transrectal ultrasound-guided biopsy of the prostate. Urol. Int. 89, 439–444 (2012).
Wagenlehner, F., Stower-Hoffmann, J., Schneider-Brachert, W., Naber, K. G. & Lehn, N. Influence of a prophylactic single dose of ciprofloxacin on the level of resistance of Escherichia coli to fluoroquinolones in urology. Int. J. Antimicrob. Agents 15, 207–211 (2000).
Taylor, A. K. et al. Targeted antimicrobial prophylaxis using rectal swab cultures in men undergoing transrectal ultrasound guided prostate biopsy is associated with reduced incidence of postoperative infectious complications and cost of care. J. Urol. 187, 1275–1279 (2012).
Jeon, S. S., Woo, S. H., Hyun, J. H., Choi, H. Y. & Chai, S. E. Bisacodyl rectal preparation can decrease infectious complications of transrectal ultrasound-guided prostate biopsy. Urology 62, 461–466 (2003).
Abughosh, Z. et al. A prospective randomized trial of povidone-iodine prophylactic cleansing of the rectum before transrectal ultrasound guided prostate biopsy. J. Urol. 189, 1326–1331 (2012).
Issa, M. M. et al. Formalin disinfection of biopsy needle minimizes the risk of sepsis following prostate biopsy. J. Urol. 190, 1769–1775 (2013).
Hara, R. et al. Optimal approach for prostate cancer detection as initial biopsy: prospective randomized study comparing transperineal versus transrectal systematic 12-core biopsy. Urology 71, 191–195 (2008).
Miller, J., Perumalla, C. & Heap, G. Complications of transrectal versus transperineal prostate biopsy. ANZ J. Surg. 75, 48–50 (2005).
Shen, P. F. et al. The results of transperineal versus transrectal prostate biopsy: a systematic review and meta-analysis. Asian J. Androl. 14, 310–315 (2012).
Dellinger, R. P. et al. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit. Care Med. 41, 580–637 (2013).
Elhanan, G., Sarhat, M. & Raz, R. Empiric antibiotic treatment and the misuse of culture results and antibiotic sensitivities in patients with community-acquired bacteraemia due to urinary tract infection. J. Infect. 35, 283–288 (1997).
Dellinger, R. P. et al. Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock, 2012. Intensive Care Med. 39, 165–228 (2013).
Wagenlehner, F. M. et al. Diagnosis and management for urosepsis. Int. J. Urol. 20, 963–970 (2013).
Perletti, G., Marras, E., Wagenlehner, F. M. & Magri, V. Antimicrobial therapy for chronic bacterial prostatitis. Cochrane Database of Systematic Reviews, Issue 8. Art. No.: CD009071. http://dx.doi.org/10.1002/14651858.CD009071.pub2 (2013).
Author information
Authors and Affiliations
Contributions
F. M. E. Wagenlehner researched, wrote, edited, and discussed this article with colleagues. A. Pilatz researched the literature and reviewed the manuscript before submission. P. Waliszewski, W. Weidner, and T. E. Bjerklund Johansen reviewed the article and made substantial contributions towards discussions of content.
Corresponding author
Ethics declarations
Competing interests
F. M. E. Wagenlehner has served as a paid consultant for Astellas, AstraZeneca, Bionorica, Cernelle, Cubist, OM-Pharma, Lilly Pharma, Pierre Fabre, and Rosen-Pharma. He has received lecture honoraria from AstraZeneca, Bionorica, OM-Pharma, Pierre Fabre, Rosen Pharma, and Serag Wiessner. He has been paid for performing clinical trials on behalf of Astellas, AstraZeneca, Calixa, Cerexa, Cernelle, Cubist, GSK, Merlion, OM-Pharma, Janssen-Cilag, Johnson & Johnson, Lilly Pharma, Pharmacia, Pierre-Fabre, Rosen Pharma, Sanofi-Aventis, Strathmann, and Zambon. The other authors declare no competing interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Wagenlehner, F., Pilatz, A., Waliszewski, P. et al. Reducing infection rates after prostate biopsy. Nat Rev Urol 11, 80–86 (2014). https://doi.org/10.1038/nrurol.2013.322
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2013.322
This article is cited by
-
Antimicrobial prophylaxis protocol based on rectal swab culture before prostate biopsy to prevent infectious complications: a prospective randomized comparative study
International Urology and Nephrology (2024)
-
Antibiotikaprophylaxe bei transrektaler Prostatabiopsie
Der Urologe (2022)
-
Zeitgemäße Antibiotikaprophylaxe bei der transrektalen Prostatabiopsie
Uro-News (2020)
-
Antibiotic resistance, hospitalizations, and mortality related to prostate biopsy: first report from the Norwegian Patient Registry
World Journal of Urology (2020)
-
Fiducial markers implantation for prostate image-guided radiotherapy: a report on the transperineal approach
La radiologia medica (2019)