Key Points
-
Responses to biologic therapies for rheumatoid arthritis (RA) decrease depending on the patient population: early RA, methotrexate-naive; established RA, methotrexate-experienced; or late RA, anti-TNF experienced
-
Within these populations, approved biologic agents that target different molecules have similar efficacy, possibly because they all ultimately inhibit a common pathway, namely proinflammatory cytokines such as TNF or IL-6
-
The best outcomes are achieved by timely adaptation or switching of therapies in accordance with disease activity, in a treat-to-target approach, with the aim of remission or at least low disease activity
-
Once a good outcome has been reached, reducing the dose or expanding the interval between doses is a feasible approach that enables maintenance of the outcome in most patients
-
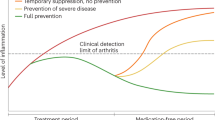
There exists a 'window of opportunity' soon after symptom onset to prevent the occurrence of damage, but treatment at this stage cannot reverse the disease in most patients
-
Reversal of disease might become possible by use of preventative therapies that interfere with the pre-arthritic process before the disease has manifested clinically
Abstract
Rheumatoid arthritis (RA) is considered a chronic disease that cannot be cured. Biologic agents have enabled good therapeutic successes; however, the response to biologic therapy depends on treatment history and, especially, disease duration. In general, the more drug-experienced the patients, the lower the response rates, although this limitation can be overcome by promptly adjusting or switching treatment in a treat-to-target approach. Another challenge is the question of how long therapy should be continued once the treatment target, which should be remission or at least a state of low disease activity, has been reached. The data available suggest that, in most patients with established disease, cessation of biologic therapy will be followed by disease flares, whereas a reduction of dose or an increase in the interval between doses enables maintenance of treatment success. Induction therapy very early in the disease course followed by withdrawal of the biologic agent might also be a feasible approach to attain sustained good outcomes, but currently available data are not strong enough to allow for such a conclusion to be reached. Taken together, this underscores the importance of research into the cause(s) of RA so that curative therapies can be developed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
[No authors listed] The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N. Engl. J. Med. 329, 977–986 (1993).
Warram, J. H., Manson, J. E. & Krolewski, A. S. Glycosylated hemoglobin and the risk of retinopathy in insulin-dependent diabetes mellitus. N. Engl. J. Med. 332, 1305–1306 (1995).
Garber, A. J. Treat-to-target trials: uses, interpretation and review of concepts. Diabetes Obes. Metab. 16, 193–205 (2014).
[No authors listed] Effects of treatment on morbidity in hypertension. II. Results in patients with diastolic blood pressure averaging 90 through 114 mm Hg. JAMA 213, 1143–1152 (1970).
Rachmani, R., Slavacheski, I., Berla, M., Frommer-Shapira, R. & Ravid, M. Treatment of high-risk patients with diabetes: motivation and teaching intervention: a randomized, prospective 8-year follow-up study. J. Am. Soc. Nephrol. 16 (Suppl. 1), S22–S26 (2005).
Patel, A. et al. Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): a randomised controlled trial. Lancet 370, 829–840 (2007).
Wilske, K. R. & Healey, L. A. Challenging the therapeutic pyramid: a new look at treatment strategies for rheumatoid arthritis. J. Rheumatol. 17 (Suppl. 25), 4–7 (1990).
Wright, V. & Amos, R. Do drugs change the course of rheumatoid arthritis? Br. Med. J. 280, 964–966 (1980).
Egsmose, C. et al. Patients with rheumatoid arthritis benefit from early 2nd line therapy: 5 year followup of a prospective double blind placebo controlled study. J. Rheumatol. 22, 2208–2213 (1995).
van der Heide, A. et al. The effectiveness of early treatment with “second-line” antirheumatic drugs. A randomized, controlled trial. Ann. Intern. Med. 124, 699–707 (1996).
Smolen, J. S. & Aletaha, D. Forget personalised medicine and focus on abating disease activity. Ann. Rheum. Dis. 72, 3–6 (2013).
Weinblatt, M. E. et al. Efficacy and safety of certolizumab pegol in a broad population of patients with active rheumatoid arthritis: results from the REALISTIC phase IIIb study. Rheumatology (Oxford) 51, 2204–2214 (2012).
Smolen, J. S. et al. Treating rheumatoid arthritis to target: recommendations of an international task force. Ann. Rheum. Dis. 69, 631–637 (2010).
Felson, D. T. et al. The American College of Rheumatology preliminary core set of disease activity measures for rheumatoid arthritis clinical trials. The Committee on Outcome Measures in Rheumatoid Arthritis Clinical Trials. Arthritis Rheum. 36, 729–740 (1993).
Aletaha, D. & Smolen, J. S. The definition and measurement of disease modification in inflammatory rheumatic diseases. Rheum. Dis. Clin. North Am. 32, 9–44 (2006).
van Ede, A. E. et al. Effect of folic or folinic acid supplementation on the toxicity and efficacy of methotrexate in rheumatoid arthritis: a forty-eight week, multicenter, randomized, double-blind, placebo-controlled study. Arthritis Rheum. 44, 1515–1524 (2001).
Visser, K. & van der Heijde, D. Optimal dosage and route of administration of methotrexate in rheumatoid arthritis: a systematic review of the literature. Ann. Rheum. Dis. 68, 1094–1099 (2009).
Gaujoux-Viala, C. et al. Efficacy of conventional synthetic disease-modifying antirheumatic drugs, glucocorticoids and tofacitinib: a systematic literature review informing the update of the EULAR recommendations for management of rheumatoid arthritis. Ann. Rheum. Dis. 73, 510–515 (2013).
Elliott, M. J. et al. Randomised double-blind comparison of chimeric monoclonal antibody to tumour necrosis factor α (cA2) versus placebo in rheumatoid arthritis. Lancet 344, 1105–1110 (1994).
Smolen, J. S., Van der Heijde, D., Machold, K. P., Aletaha, D. & Landewe, R. Proposal for a new nomenclature of disease-modifying antirheumatic drugs. Ann. Rheum. Dis. 73, 3–5 (2014).
Smolen, J. S. et al. Effect of interleukin-6 receptor inhibition with tocilizumab in patients with rheumatoid arthritis (OPTION study): a double-blind, placebo-controlled, randomised trial. Lancet 371, 987–997 (2008).
Tak, P. P. et al. Inhibition of joint damage and improved clinical outcomes with rituximab plus methotrexate in early active rheumatoid arthritis: the IMAGE trial. Ann. Rheum. Dis. 70, 39–46 (2011).
Westhovens, R. et al. Clinical efficacy and safety of abatacept in methotrexate-naive patients with early rheumatoid arthritis and poor prognostic factors. Ann. Rheum. Dis. 68, 1870–1877 (2009).
Burmester, G. R. et al. Tofacitinib (CP-690,550) in combination with methotrexate in patients with active rheumatoid arthritis with an inadequate response to tumour necrosis factor inhibitors: a randomised phase 3 trial. Lancet 381, 451–560 (2013).
Genovese, M. C. et al. Efficacy and safety of secukinumab in patients with rheumatoid arthritis: a phase II, dose-finding, double-blind, randomised, placebo controlled study. Ann. Rheum. Dis. 72, 863–869 (2013).
Smolen, J. S., Weinblatt, M. E., Sheng, S., Zhuang, Y. & Hsu, B. Sirukumab, a human anti-interleukin-6 monoclonal antibody: a randomised, 2-part (proof-of-concept and dose-finding), phase II study in patients with active rheumatoid arthritis despite methotrexate therapy. Ann. Rheum. Dis. 73, 1616–1625 (2014).
Toussirot, E. The IL23/TH17 pathway as a therapeutic target in chronic inflammatory diseases. Inflamm. Allergy Drug Targets 11, 159–168 (2012).
Cutolo, M. & Nadler, S. G. Advances in CTLA-4-Ig-mediated modulation of inflammatory cell and immune response activation in rheumatoid arthritis. Autoimmun. Rev. 12, 758–767 (2013).
Buch, M. et al. Updated consensus statement on the use of rituximab in patients with rheumatoid arthritis. Ann. Rheum. Dis. 70, 909–920 (2011).
van der Lubbe, P. A., Dijkmans, B. S., Markusse, H., Nassander, U. & Breedveld, F. C. A randomized, double-blind, placebo-controlled study of CD4 monoclonal antibody therapy in early rheumatoid arthritis. Arthritis Rheum. 38, 1097–1106 (1995).
Choy, E. H. et al. Repeat-cycle study of high-dose intravenous 4162W94 anti-CD4 humanized monoclonal antibody in rheumatoid arthritis. A randomized placebo-controlled trial. Rheumatology (Oxford) 41, 1142–1148 (2002).
Stohl, W. et al. Efficacy and safety of belimumab in patients with rheumatoid arthritis: a phase II, randomized, double-blind, placebo-controlled, dose-ranging study. J. Rheumatol. 40, 579–589 (2013).
Blüml, S., McKeever, K., Ettinger, R., Smolen, J. & Herbst, R. B-cell targeted therapeutics in clinical development. Arthritis Res. Ther. 15 (Suppl. 1), S4 (2013).
Bonelli, M. et al. Abatacept (CTLA-4IG) treatment reduces the migratory capacity of monocytes in patients with rheumatoid arthritis. Arthritis Rheum. 65, 599–607 (2013).
Genovese, M. C. et al. A 24-week, randomized, double-blind, placebo-controlled, parallel group study of the efficacy of oral SCIO-469, a p38 mitogen-activated protein kinase inhibitor, in patients with active rheumatoid arthritis. Arthritis Rheum. 38, 846–854 (2011).
Cohen, S. B. et al. Evaluation of the efficacy and safety of pamapimod, a p38 MAP kinase inhibitor, in a double-blind, methotrexate-controlled study of patients with active rheumatoid arthritis. Arthritis Rheum. 60, 335–344 (2009).
Smolen, J. S., Aletaha, D., Koeller, M., Weisman, M. & Emery, P. New therapies for the treatment of rheumatoid arthritis. Lancet 370, 1861–1874 (2007).
Mierau, M. et al. Assessing remission in clinical practice. Rheumatology (Oxford) 46, 975–979 (2007).
Smolen, J. S. et al. Golimumab in patients with active rheumatoid arthritis after treatment with tumour necrosis factor α inhibitors (GO-AFTER study): a multicentre, randomised, double-blind, placebo-controlled, phase III trial. Ann. Rheum. Dis. 374, 210–221 (2009).
Cohen, S. B. et al. Rituximab for rheumatoid arthritis refractory to anti-tumor necrosis factor therapy: results of a multicenter, randomized, double-blind, placebo-controlled, phase III trial evaluating primary efficacy and safety at twenty-four weeks. Arthritis Rheum. 54, 2739–2806 (2006).
Emery, P. et al. IL-6 receptor inhibition with tocilizumab improves treatment outcomes in patients with rheumatoid arthritis refractory to anti-tumour necrosis factor biologicals: results from a 24-week multicentre randomised placebo-controlled trial. Ann. Rheum. Dis. 67, 1516–1523 (2008).
Genovese, M. C. et al. Abatacept for rheumatoid arthritis refractory to tumor necrosis factor α inhibition. N. Engl. J. Med. 353, 1114–1123 (2005).
Burmester, G. R. et al. Tocilizumab (TCZ) in combination and monotherapy versus methotrexate (MTX) in MTX-naive patients (pts) with early rheumatoid arthritis (RA): clinical and radiographic outcomes from a randomised, placebo-controlled trial [abstract OP0041]. Ann. Rheum. Dis. 72 (Suppl. 3), A63 (2013).
Kremer, J. M. et al. Effects of abatacept in patients with methotrexate-resistant active rheumatoid arthritis: a randomized trial. Ann. Intern. Med. 144, 865–876 (2006).
Westhovens, R. et al. Improved health-related quality of life for rheumatoid arthritis patients treated with abatacept who have inadequate response to anti-TNF therapy in a double-blind, placebo-controlled, multicentre randomized clinical trial. Rheumatology (Oxford) 45, 1238–1246 (2006).
Emery, P. et al. Golimumab, a human anti-tumor necrosis factor α monoclonal antibody, injected subcutaneously every four weeks in methotrexate-naive patients with active rheumatoid arthritis: twenty-four-week results of a phase III, multicenter, randomized, double-blind, placebo-controlled study of golimumab before methotrexate as first-line therapy for early-onset rheumatoid arthritis. Arthritis Rheum. 60, 2272–2283 (2009).
Keystone, E. C. et al. Golimumab, a human antibody to tumour necrosis factor α given by monthly subcutaneous injections, in active rheumatoid arthritis despite methotrexate therapy: the GO-FORWARD Study. Ann. Rheum. Dis. 68, 789–796 (2009).
Emery, P. et al. The efficacy and safety of rituximab in patients with active rheumatoid arthritis despite methotrexate treatment: results of a phase IIB randomized, double-blind, placebo-controlled, dose-ranging trial. Arthritis Rheum. 54, 1390–1400 (2006).
Breedveld, F. C. et al. The PREMIER study—a multicenter, randomized, double-blind clinical trial of combination therapy with adalimumab plus methotrexate versus methotrexate alone or adalimumab alone in patients with early, aggressive rheumatoid arthritis who had not had previous methotrexate treatment. Arthritis Rheum. 54, 26–37 (2006).
Keystone, E. C. et al. Radiographic, clinical, and functional outcomes of treatment with adalimumab (a human anti-tumor necrosis factor monoclonal antibody) in patients with active rheumatoid arthritis receiving concomitant methotrexate therapy: a randomized, placebo-controlled, 52-week trial. Arthritis Rheum. 50, 1400–1411 (2004).
Klareskog, L. et al. Therapeutic effect of the combination of etanercept and methotrexate compared with each treatment alone in patients with rheumatoid arthritis: double-blind randomised controlled trial. Lancet 363, 675–681 (2004).
Lipsky, P. E. et al. Infliximab and methotrexate in the treatment of rheumatoid arthritis. Anti-Tumor Necrosis Factor Trial in Rheumatoid Arthritis with Concomitant Therapy Study Group. N. Engl. J. Med. 343, 1594–1602 (2000).
Keystone, E. et al. Certolizumab pegol plus methotrexate is significantly more effective than placebo plus methotrexate in active rheumatoid arthritis: findings of a fifty-two-week, phase III, multicenter, randomized, double-blind, placebo-controlled, parallel-group study. Arthritis Rheum. 58, 3319–3329 (2008).
Smolen, J. S. et al. Efficacy and safety of certolizumab pegol plus methotrexate in active rheumatoid arthritis: the RAPID 2 study. Ann. Rheum. Dis. 68, 797–804 (2009).
Weinblatt, M. E. et al. A trial of etanercept, a recombinant tumor necrosis factor receptor:Fc fusion protein, in patients with rheumatoid arthritis receiving methotrexate. N. Engl. J. Med. 340, 253–259 (1999).
St Clair, E. W. et al. Combination of infliximab and methotrexate therapy for early rheumatoid arthritis: a randomized, controlled trial. Arthritis Rheum. 50, 3432–3443 (2004).
Smolen, J. S. et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: update. Ann. Rheum. Dis. 73, 492–509 (2013).
Aletaha, D., Funovits, J. & Smolen, J. S. The importance of reporting disease activity states in clinical trials of rheumatoid arthritis. Arthritis Rheum. 58, 2622–2631 (2008).
Schiff, M., Zhou, X., Kelly, S., Le Bars, M. & Genovese, M. Efficacy of abatacept in RA patients with an inadequate response to anti-TNF therapy regardless of reason for failure, or type or number of prior anti-TNF therapy used [abstract FRI0160]. Ann. Rheum. Dis. 67 (Suppl. II), 337 (2008).
Kremer, J. M. et al. Efficacy of rituximab in active RA patients with an inadequate response to one or more TNF inhibitors [abstract FRI0133]. Ann. Rheum. Dis. 65 (Suppl. II), 326. (2006).
de Jong, P. H. et al. Randomised comparison of initial triple DMARD therapy with methotrexate monotherapy in combination with low-dose glucocorticoid bridging therapy; 1-year data of the tREACH trial. Ann Rheum. Dis. 73, 1331–1339 (2014).
Genovese, M. C. et al. A phase II randomized study of subcutaneous ixekizumab, an anti-IL-17 monoclonal antibody, in rheumatoid arthritis patients who were naive to biologic agents or had an inadequate response to tumor necrosis factor inhibitors. Arthritis Rheumatol. 66, 1693–1704 (2014).
Ritchlin, C. et al. Efficacy and safety of the anti-IL-12/23 p40 monoclonal antibody, ustekinumab, in patients with active psoriatic arthritis despite conventional non-biological and biological anti-tumour necrosis factor therapy: 6-month and 1-year results of the phase 3, multicentre, double-blind, placebo-controlled, randomised PSUMMIT 2 trial. Ann. Rheum. Dis. 73, 990–999 (2014).
Brennan, F. M., Chantry, D., Jackson, A., Maini, R. & Feldmann, M. Inhibitory effect of TNF-α antibodies on synovial cell interleukin-1 production in rheumatoid arthritis. Lancet 2, 244–247 (1989).
Scheinecker, C., Redlich, K. & Smolen, J. S. Cytokines as therapeutic targets: advances and limitations. Immunity 28, 440–444 (2008).
van den Brande, J. M. et al. Infliximab but not etanercept induces apoptosis in lamina propria T-lymphocytes from patients with Crohn's disease. Gastroenterology 124, 1774–1785 (2003).
Sieper, J., Porter-Brown, B., Thompson, L., Harari, O. & Dougados, M. Assessment of short-term symptomatic efficacy of tocilizumab in ankylosing spondylitis: results of randomised, placebo-controlled trials. Ann. Rheum. Dis. 73, 95–100 (2014).
Ito, H. et al. A pilot randomized trial of a human anti-interleukin-6 receptor monoclonal antibody in active Crohn's disease. Gastroenterology 126, 989–996 (2004).
Papp, K. A. et al. Efficacy and safety of secukinumab in the treatment of moderate-to-severe plaque psoriasis: a randomized, double-blind, placebo-controlled phase II dose-ranging study. Br. J. Dermatol. 168, 412–421 (2013).
Mease, P. et al. Secukinumab, a human anti-interleukin-17A monoclonal antibody, improves active psoriatic arthritis and inhibits radiographic progression: efficacy and safety data from a phase 3 randomized, multicenter, double-blind, placebo-controlled study. Arthritis Rheum. 66 (Suppl.), S423–S424 (2014).
McInnes, I. B. et al. Efficacy and safety of secukinumab, a fully human anti-interleukin-17A monoclonal antibody, in patients with moderate-to-severe psoriatic arthritis: a 24-week, randomised, double-blind, placebo-controlled, phase II proof-of-concept trial. Ann. Rheum. Dis. 73, 349–356 (2014).
Baeten, D. et al. Anti-interleukin-17A monoclonal antibody secukinumab in treatment of ankylosing spondylitis: a randomised, double-blind, placebo-controlled trial. Lancet 382, 1705–1713 (2013).
Sieper, J. et al. Secukinumab, a monoclonal antibody to interleukin-17A, significantly improves signs and symptoms of active ankylosing spondylitis: results of a phase 3, randomized, placebo-controlled trial with subcutaneous loading and maintenance dosing. Arthritis Rheum. 66 (Suppl.), S232 (2014).
Hueber, W. et al. Secukinumab, a human anti-IL-17A monoclonal antibody, for moderate to severe Crohn's disease: unexpected results of a randomised, double-blind placebo-controlled trial. Gut 61, 1693–1700 (2012).
Griffiths, C. E. et al. Comparison of ustekinumab and etanercept for moderate-to-severe psoriasis. N. Engl. J. Med. 362, 118–128 (2010).
McInnes, I. B. et al. Efficacy and safety of ustekinumab in patients with active psoriatic arthritis: 1 year results of the phase 3, multicentre, double-blind, placebo-controlled PSUMMIT 1 trial. Lancet 382, 780–789 (2013).
Sandborn, W. J. et al. Ustekinumab induction and maintenance therapy in refractory Crohn's disease. N. Engl. J. Med. 367, 1519–1528 (2012).
Smolen, J. S. et al. Radiographic changes in rheumatoid arthritis patients attaining different disease activity states with methotrexate monotherapy and infliximab plus methotrexate: the impacts of remission and TNF-blockade. Ann. Rheum. Dis. 68, 823–827 (2009).
Radner, H., Smolen, J. S. & Aletaha, D. Remission in rheumatoid arthritis: benefit over low disease activity in patient reported outcomes and costs. Arthritis Res. Ther. 16, R56 (2014).
Aletaha, D. et al. Remission and active disease in rheumatoid arthritis: defining criteria for disease activity states. Arthritis Rheum. 52, 2625–2636 (2005).
Van der Heijde, D. et al. Comparison of different definitions to classify remission and sustained remission: 1 year TEMPO results. Ann. Rheum. Dis. 64, 1582–1587 (2005).
Bakker, M. F., Jacobs, J. W., Verstappen, S. M. & Bijlsma, J. W. Tight control in the treatment of rheumatoid arthritis: efficacy and feasibility. Ann. Rheum. Dis. 66 (Suppl. 3), iii56–iii60 (2007).
Smolen, J. S. & Aletaha, D. The assessment of disease activity in rheumatoid arthritis. Clin. Exp. Rheumatol. 28 (Suppl. 59), S18–S27 (2010).
Aletaha, D. & Smolen, J. S. Joint damage in rheumatoid arthritis progresses in remission according to the Disease Activity Score in 28 joints and is driven by residual swollen joints. Arthritis Rheum. 63, 3702–3711 (2011).
Kavanaugh, A. et al. Clinical, functional and radiographic consequences of achieving stable low disease activity and remission with adalimumab plus methotrexate or methotrexate alone in early rheumatoid arthritis: 26-week results from the randomised, controlled OPTIMA study. Ann. Rheum. Dis. 72, 64–71 (2013).
Felson, D. T. et al. American College of Rheumatology/European League Against Rheumatism provisional definition of remission in rheumatoid arthritis for clinical trials. Ann. Rheum. Dis. 70, 404–413 (2011).
Balsa, A., de Miguel, E., Castillo, C., Peiteado, D. & Martin-Mola, E. Superiority of SDAI over DAS-28 in assessment of remission in rheumatoid arthritis patients using power Doppler ultrasonography as a gold standard. Rheumatology (Oxford) 49, 683–690 (2010).
Sakellariou, G., Scire, C. A., Verstappen, S. M., Montecucco, C. & Caporali, R. In patients with early rheumatoid arthritis, the new ACR/EULAR definition of remission identifies patients with persistent absence of functional disability and suppression of ultrasonographic synovitis. Ann. Rheum. Dis. 72, 245–249 (2013).
Emery, P. et al. Comparison of methotrexate monotherapy with a combination of methotrexate and etanercept in active, early, moderate to severe rheumatoid arthritis (COMET): a randomised, double-blind, parallel treatment trial. Lancet 372, 375–382 (2008).
Food and Drug Administration. Guidance for industry—rheumatoid arthritis: developing drug products for treatment. Draft guidance May 2013. http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM354468.pdf (2013).
van der Heijde, D. M. et al. Prognostic factors for radiographic damage and physical disability in early rheumatoid arthritis. A prospective follow-up study of 147 patients. Br. J. Rheumatol. 31, 519–525 (1992).
Scott, D. L. et al. Progression of radiological changes in rheumatoid arthritis. Ann. Rheum. Dis. 43, 8–17 (1984).
Fries, J. F. et al. Assessment of radiologic progression in rheumatoid arthritis. A randomized, controlled trial. Arthritis Rheum. 29, 1–9 (1986).
Sigler, J. W. et al. Gold salts in the treatment of rheumatoid arthritis. A double-blind study. Ann. Intern. Med. 80, 21–26 (1974).
van der Heijde, D. M., van Riel, P. L., Nuver-Zwart, I. H. & van de Putte, L. B. Sulphasalazine versus hydroxychloroquine in rheumatoid arthritis: 3-year follow-up. Lancet 335, 539 (1990).
Dawes, P. T. et al. Prediction of progressive joint damage in patients with rheumatoid arthritis receiving gold or D-penicillamine therapy. Ann. Rheum. Dis. 45, 945–949 (1986).
Drossaers-Bakker, K. W. et al. Long-term course and outcome of functional capacity inrheumatoid arthritis: the effect of disease activity and radiologic damage over time. Arthritis Rheum. 42, 1854–1860 (1999).
van der Heijde, D., Landewe R, van Vollenhoven, R., Fatenejad, S. & Klareskog, L. Level of radiographic damage and radiographic progression are determinants of physical function: a longitudinal analysis of the TEMPO trial. Ann. Rheum. Dis. 67, 1267–1270 (2008).
Smolen, J. S., Aletaha, D., Grisar, J. C., Stamm, T. A. & Sharp, J. T. Estimation of a numerical value for joint damage-related physical disability in rheumatoid arthritis clinical trials. Ann. Rheum. Dis. 69, 1058–1064 (2010).
Aletaha, D., Smolen, J. & Ward, M. M. Measuring function in rheumatoid arthritis: identifying reversible and irreversible components. Arthritis Rheum. 54, 2784–2792 (2006).
Aletaha, D., Funovits, J., Keystone, E. C. & Smolen, J. S. Disease activity early in the course of treatment predicts response to therapy after one year in rheumatoid arthritis patients. Arthritis Rheum. 56, 3226–3235 (2007).
Provan, S. A. et al. Remission is the goal for cardiovascular risk management in patients with rheumatoid arthritis: a cross-sectional comparative study. Ann. Rheum. Dis. 70, 812–817 (2011).
Linde, L., Sorensen, J., Ostergaard, M., Horslev-Petersen, K. & Hetland, M. L. Does clinical remission lead to normalization of EQ-5D in patients with rheumatoid arthritis and is selection of remission criteria important? J. Rheumatol. 37, 285–290 (2010).
Thiele, K. et al. Performance of the 2011 ACR/EULAR preliminary remission criteria compared with DAS28 remission in unselected patients with rheumatoid arthritis. Ann. Rheum. Dis. 72, 1194–1199 (2013).
Ridker, P. M. et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N. Engl. J. Med. 359, 2195–2207 (2008).
Schoels, M. et al. Evidence for treating rheumatoid arthritis to target: results of a systematic literature search. Ann. Rheum. Dis. 69, 638–643 (2010).
Grigor, C. et al. Effect of a treatment strategy of tight control for rheumatoid arthritis (the TICORA study): a single-blind randomised controlled trial. Lancet 364, 263–269 (2004).
De Wit, M., Smolen, J. S., Gossec, L. & van der Heijde, D. Treating rheumatoid arthritis to target: the patient version of the international recommendations. Ann. Rheum. Dis. 70, 891–895 (2011).
Smolen, J. S. et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs. Ann. Rheum. Dis. 69, 964–975 (2010).
Singh, J. A. et al. 2012 update of the 2008 American College of Rheumatology recommendations for the use of disease-modifying antirheumatic drugs and biologic agents in the treatment of rheumatoid arthritis. Arthritis Care Res (Hoboken) 64, 625–639 (2012).
Bakker, M. F. et al. Performance of a multi-biomarker score measuring rheumatoid arthritis disease activity in the CAMERA tight control study. Ann. Rheum. Dis. 71, 1692–1697 (2012).
Vermeer, M. et al. Adherence to a treat-to-target strategy in early rheumatoid arthritis: results of the DREAM remission induction cohort. Arthritis Res. Ther. 14, R254 (2012).
Smolen, J. S. et al. Adjustment of therapy in rheumatoid arthritis on the basis of achievement of stable low disease activity with adalimumab plus methotrexate or methotrexate alone: the randomised controlled OPTIMA trial. Lancet 383, 321–332 (2014).
Porter, D., Dale, J. & Sattar, N. How low to aim in rheumatoid arthritis? Learning from other disciplines. Ann. Rheum. Dis. 73, 480–482 (2014).
Solomon, D. H. et al. Review: treat to target in rheumatoid arthritis: fact, fiction, or hypothesis? Arthritis Rheumatol. 66, 775–782 (2014).
Nell, V. et al. Benefit of very early referral and very early therapy with disease-modifying anti-rheumatic drugs in patients with early rheumatoid arthritis. Rheumatology (Oxford) 43, 906–914 (2004).
McCarty, D. J. Suppress rheumatoid inflammation early and leave the pyramid to the Egyptians. J. Rheumatol. 17, 1117–1118 (1990).
Binder, N. B. et al. Tumor necrosis factor-inhibiting therapy preferentially targets bone destruction but not synovial inflammation in a tumor necrosis factor-driven model of rheumatoid arthritis. Arthritis Rheum. 65, 608–617 (2013).
Lard, L. R. et al. Early versus delayed treatment in patients with recent-onset rheumatoid arthritis: comparison of two cohorts who received different treatment strategies. Am. J. Med. 111, 446–451 (2001).
Nam, J. L. et al. Remission induction comparing infliximab and high-dose intravenous steroid, followed by treat-to-target: a double-blind, randomised, controlled trial in new-onset, treatment-naive, rheumatoid arthritis (the IDEA study). Ann. Rheum. Dis. 73, 75–85 (2014).
ten Wolde, S. et al. Randomised placebo-controlled study of stopping second-line drugs in rheumatoid arthritis. Lancet 347, 347–352 (1996).
Kremer, J. M., Rynes, R. I. & Bartholomew, L. E. Severe flare of rheumatoid arthritis after discontinuation of long-term methotrexate therapy. Double-blind study. Am. J. Med. 82, 781–786 (1987).
Ahern, M. J., Hall, N. D., Case, K. & Maddison, P. J. D-penicillamine withdrawal in rheumatoid arthritis. Ann. Rheum. Dis. 43, 213–217 (1984).
ten Wolde, S., Hermans, J., Breedveld, F. C. & Dijkmans, B. A. Effect of resumption of second line drugs in patients with rheumatoid arthritis that flared up after treatment discontinuation. Ann. Rheum. Dis. 56, 235–239 (1997).
O'Mahony, R., Richards, A., Deighton, C. & Scott, D. Withdrawal of DMARDs in patients with rheumatoid arthritis: a systematic review and meta-analysis. Ann. Rheum. Dis. 69, 1823–1826 (2010).
Burmester, G. R. et al. Efficacy and safety of ascending methotrexate dose in combination with adalimumab: the randomised CONCERTO trial. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2013-204769.
Maini, R. N. et al. Therapeutic efficacy of multiple intravenous infusions of anti-tumor necrosis factor α monoclonal antibody combined with low-dose weekly methotrexate in rheumatoid arthritis. Arthritis Rheum. 41, 1552–1563 (1998).
Quinn, M. A. et al. Very early treatment with infliximab in addition to methotrexate in early, poor-prognosis rheumatoid arthritis reduces magnetic resonance imaging evidence of synovitis and damage, with sustained benefit after infliximab withdrawal: results from a twelve-month randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 52, 27–35 (2005).
Raza, K. The Michael Mason prize: early rheumatoid arthritis—the window narrows. Rheumatology (Oxford) 49, 406–410 (2010).
Tanaka, Y. et al. Discontinuation of infliximab after attaining low disease activity in patients with rheumatoid arthritis: RRR (Remission induction by Remicade in RA) study. Ann. Rheum. Dis. 69, 1286–1291 (2010).
Tanaka, Y. et al. Discontinuation of adalimumab after achieving remission in patients with established rheumatoid arthritis: 1-year outcome of the HONOR study. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2013-204016.
Saleem, B. et al. Patients with RA in remission on TNF blockers: when and in whom can TNF blocker therapy be stopped? Ann. Rheum. Dis. 69, 1636–1642 (2010).
Brocq, O. et al. Effect of discontinuing TNFα antagonist therapy in patients with remission of rheumatoid arthritis. Joint Bone Spine 76, 350–355 (2009).
Smolen, J. S. et al. Maintenance, reduction, or withdrawal of etanercept after treatment with etanercept and methotrexate in patients with moderate rheumatoid arthritis (PRESERVE): a randomised controlled trial. Lancet 381, 918–929 (2013).
Smolen, J. S., Szumski, A., Koenig, A. S. & Jones, T. V. Predictors of loss of clinical remission with etanercept 50 mg, 25 mg or placebo combined with methotrexate in moderate rheumatoid arthritis patients: results of the PRESERVE trial [abstract #FRI0164]. Ann. Rheum. Dis. 71 (Suppl 3), 367 (2012).
Smolen, J. S. et al. Certolizumab pegol in rheumatoid arthritis patients with low to moderate activity: the CERTAIN double-blind, randomised, placebo-controlled trial. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2013-204632.
Huizinga, T. W. J. et al. Clinical and radiographic outcomes at 2 years and the effect of tocilizumab discontinuation following sustained remission in the second and third year of the ACT-RAY study. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2014-205752.
Smolen, J. S. & Aletaha, D. Interleukin-6 receptor inhibition with tocilizumab and attainment of disease remission in rheumatoid arthritis: the role of acute-phase reactants. Arthritis Rheum. 63, 43–52 (2011).
Fautrel, B. et al. Targeting the lowest efficacious dose for rheumatoid arthritis patients in remission: clinical and structural impact of a stepdown strategy trial based on progressive spacing of TNF-blocker injections (STRASS trial) [abstract #OP0066]. Ann. Rheum Dis. 72 (Suppl. 3), 72 (2013).
Wilske, K. R. Approaches to the management of rheumatoid arthritis: rationale for early combination therapy. Br. J. Rheumatol. 32 (Suppl. 1), 24–27 (1993).
Dawes, P. T., Symmons, D. P. Short-term effects of antirheumatic drugs. Baillieres Clin. Rheumatol. 6, 117–140 (1992).
Allaart, C. F., Lems, W. F. & Huizinga, T. W. The BeSt way of withdrawing biologic agents. Clin. Exp. Rheumatol. 31 (4 Suppl. 78), S14–S18 (2013).
Detert, J. et al. Induction therapy with adalimumab plus methotrexate for 24 weeks followed by methotrexate monotherapy up to week 48 versus methotrexate therapy alone for DMARD-naive patients with early rheumatoid arthritis: HIT HARD, an investigator-initiated study. Ann. Rheum. Dis. 72, 844–850 (2013).
Emery, P. et al. Sustained remission with etanercept tapering in early rheumatoid arthritis. N. Engl. J. Med. 371, 1781–1792 (2014).
Machold, K. P. et al. The Stop Arthritis Very Early (SAVE) trial, an international multicentre, randomised, double-blind, placebo-controlled trial on glucocorticoids in very early arthritis. Ann. Rheum. Dis. 69, 495–502 (2010).
Jimenez-Boj, M. E. et al. Predicting rheumatoid arthritis by autoantibody testing (preRA): preliminary results of a community-based investigation [abstract FRI0085]. Ann. Rheum. Dis. 71 (Suppl. 3), 338 (2012).
Cohen, S. et al. Treatment of rheumatoid arthritis with anakinra, a recombinant human interleukin-1 receptor antagonist, in combination with methotrexate: results of a twenty-four-week, multicenter, randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 46, 614–624 (2002).
Van Nies, J. A. et al. What is the evidence for the presence of a therapeutic window of opportunity in rheumatoid arthritis? A systematic literature review. Ann. Rheum. Dis. 73, 861–870 (2014).
Raza, K., Saber, T. P., Kvien, T. K., Tak, P. P. & Gerlag, D. M. Timing the therapeutic window of opportunity in early rheumatoid arthritis: proposal for definitions of disease duration in clinical trials. Ann. Rheum. Dis. 71, 1921–1923 (2012).
Van Aken, J., Heimans, L., Gillet-van, D. H., Visser, K., Ronday, H. K., Speyer, I. et al. Five-year outcomes of probable rheumatoid arthritis treated with methotrexate or placebo during the first year (the PROMPT study). Ann. Rheum. Dis. 73, 396–400 (2014).
Raza, K. et al. Early rheumatoid arthritis is characterized by a distinct and transient synovial fluid cytokine profile of T cell and stromal cell origin. Arthritis Res. Ther. 7, R784–R795 (2005).
Tak, P. P. Analyzing synovial tissue samples. What can we learn about early rheumatoid arthritis, the heterogeneity of the disease, and the effects of treatment? J. Rheumatol. 32 (Suppl. 72), 25–26 (2005).
Tak, P. P. et al. Analysis of the synovial cell infiltrate in early rheumatoid synovial tissue in relation to local disease activity. Arthritis Rheum. 40, 217–225 (1997).
Gerlag, D. M. et al. EULAR recommendations for terminology and research in individuals at risk of rheumatoid arthritis: report from the Study Group for Risk Factors for Rheumatoid Arthritis. Ann. Rheum. Dis. 71, 638–641 (2012).
Aho, K., Heliovaara, M., Maatela, J., Tuomi, T. & Palusuo, T. Rheumatoid factors antedating clinical rheumatoid arthritis. J. Rheumatol. 18, 1282–1284 (1991).
Aho, K., von Essen, R., Kurki, P., Palusuo, T. & Heliovaara, M. Antikeratin antibody and antiperinuclear factor as markers for subclinical rheumatoid disease process. J. Rheumatol. 20, 1278–1281 (1993).
Nielen, M. M. et al. Specific autoantibodies precede the symptoms of rheumatoid arthritis: a study of serial measurements in blood donors. Arthritis Rheum. 50, 380–386 (2004).
Kraan, M. C. et al. Asymptomatic synovitis precedes clinically manifest arthritis. Arthritis Rheum. 41, 1481–1488 (1998).
Hayer, S. et al. Tenosynovitis and osteoclast formation as the initial preclinical changes in a murine model of inflammatory arthritis. Arthritis Rheum. 56, 79–88 (2007).
Smolen, J. S. et al. Pre-arthritis: a concept whose time has come. Future Rheumatol. 1, 1–4 (2006).
Author information
Authors and Affiliations
Contributions
Both authors contributed to researching data for the article, discussing the content, writing and review/editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
J.S.S. has received research support from Abbvie, BMS, MSD, Pfizer, Roche and UCB, and has acted as a consultant and is a member of the speakers' bureau for Abbvie, Amgen, BMS, Celgene, Glaxo, Infinity, Janssen, Lilly, Medimmune, MSD, Novo-Nordisk, Pfizer, Roche, Samsung, Sanofi, Sandoz and UCB. D.A. has received research support from BMS, MSD and Roche, and has acted as a consultant and is a member of the speakers' bureau for Abbvie, BMS, Janssen, Medac, MSD, Pfizer and Roche.
Rights and permissions
About this article
Cite this article
Smolen, J., Aletaha, D. Rheumatoid arthritis therapy reappraisal: strategies, opportunities and challenges. Nat Rev Rheumatol 11, 276–289 (2015). https://doi.org/10.1038/nrrheum.2015.8
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2015.8
This article is cited by
-
Signaling pathways in rheumatoid arthritis: implications for targeted therapy
Signal Transduction and Targeted Therapy (2023)
-
Matrix metalloproteinases in arthritis: towards precision medicine
Nature Reviews Rheumatology (2023)
-
Fluorinated polyamidoamine dendrimer-mediated miR-23b delivery for the treatment of experimental rheumatoid arthritis in rats
Nature Communications (2023)
-
A review of the pleiotropic actions of the IFN-inducible CXC chemokine receptor 3 ligands in the synovial microenvironment
Cellular and Molecular Life Sciences (2023)
-
Comprehensive phytochemical characterization of Persea americana Mill. fruit via UPLC/HR-ESI–MS/MS and anti-arthritic evaluation using adjuvant-induced arthritis model
Inflammopharmacology (2023)