Abstract
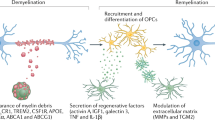
Microglia, the resident macrophages of the CNS, are exquisitely sensitive to brain injury and disease, altering their morphology and phenotype to adopt a so-called activated state in response to pathophysiological brain insults. Morphologically activated microglia, like other tissue macrophages, exist as many different phenotypes, depending on the nature of the tissue injury. Microglial responsiveness to injury suggests that these cells have the potential to act as diagnostic markers of disease onset or progression, and could contribute to the outcome of neurodegenerative diseases. The persistence of activated microglia long after acute injury and in chronic disease suggests that these cells have an innate immune memory of tissue injury and degeneration. Microglial phenotype is also modified by systemic infection or inflammation. Evidence from some preclinical models shows that systemic manipulations can ameliorate disease progression, although data from other models indicates that systemic inflammation exacerbates disease progression. Systemic inflammation is associated with a decline in function in patients with chronic neurodegenerative disease, both acutely and in the long term. The fact that diseases with a chronic systemic inflammatory component are risk factors for Alzheimer disease implies that crosstalk occurs between systemic inflammation and microglia in the CNS.
Key Points
-
The phenotype of microglia is tightly regulated within the normal healthy CNS
-
Microglia rapidly change their morphology and expression of diverse molecules in response to changes in homeostasis and pathological insults to the brain
-
Morphologically activated microglia display diverse phenotypes that critically depend on the sequence and duration of their exposure to various stimuli in different pathologies
-
Microglial morphology and changes in expression of a small number of markers are not simple guides to microglial phenotype and function
-
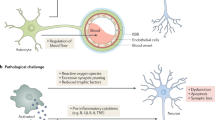
Microglial phenotype is modified by systemic infection and inflammation
-
Systemic inflammation influences the symptoms and progression of chronic neurodegenerative disease
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gordon, S. & Taylor, P. R. Monocyte and macrophage heterogeneity. Nat. Rev. Immunol. 5, 953–964 (2005).
Mosser, D. M. & Edwards, J. P. Exploring the full spectrum of macrophage activation. Nat. Rev. Immunol. 8, 958–969 (2008).
Ransohoff, R. M. & Perry, V. H. Microglial physiology: unique stimuli, specialized responses. Annu. Rev. Immunol. 27, 119–145 (2009).
Hanisch, U. K. & Kettenmann, H. Microglia: active sensor and versatile effector cells in the normal and pathologic brain. Nat. Neurosci. 10, 1387–1394 (2007).
Lawson, L. J., Perry, V. H., Dri, P. & Gordon, S. Heterogeneity in the distribution and morphology of microglia in the normal adult mouse brain. Neuroscience 39, 151–170 (1990).
de Haas, A. H., Boddeke, H. W. & Biber, K. Region-specific expression of immunoregulatory proteins on microglia in the healthy CNS. Glia 56, 888–894 (2008).
Nimmerjahn, A., Kirchhoff, F. & Helmchen, F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 308, 1314–1318 (2005).
Davalos, D. et al. ATP mediates rapid microglial response to local brain injury in vivo. Nat. Neurosci. 8, 752–758 (2005).
Lawson, L. J., Perry, V. H. & Gordon, S. Turnover of resident microglia in the normal adult mouse brain. Neuroscience 48, 405–415 (1992).
Ajami, B., Bennett, J. L., Krieger, C., Tetzlaff, W. & Rossi, F. M. Local self-renewal can sustain CNS microglia maintenance and function throughout adult life. Nat. Neurosci. 10, 1538–1543 (2007).
Mildner, A. et al. Microglia in the adult brain arise from Ly-6ChiCCR2+ monocytes only under defined host conditions. Nat. Neurosci. 10, 1544–1553 (2007).
Hickey, W. F. & Kimura, H. Perivascular microglial cells of the CNS are bone marrow-derived and present antigen in vivo. Science 239, 290–292 (1988).
Soulas, C. et al. Genetically modified CD34+ hematopoietic stem cells contribute to turnover of brain perivascular macrophages in long-term repopulated primates. Am. J. Pathol. 174, 1808–1817 (2009).
Barclay, A. N., Wright, G. J., Brooke, G. & Brown, M. H. CD200 and membrane protein interactions in the control of myeloid cells. Trends Immunol. 23, 285–290 (2002).
Hoek, R. M. et al. Down-regulation of the macrophage lineage through interaction with OX2 (CD200). Science 290, 1768–1771 (2000).
Walker, D. G., Dalsing-Hernandez, J. E., Campbell, N. A. & Lue, L. F. Decreased expression of CD200 and CD200 receptor in Alzheimer's disease: a potential mechanism leading to chronic inflammation. Exp. Neurol. 215, 5–19 (2009).
Lyons, A. et al. Fractalkine-induced activation of the phosphatidylinositol-3 kinase pathway attentuates microglial activation in vivo and in vitro. J. Neurochem. 110, 1547–1556 (2009).
van Beek, E. M., Cochrane, F., Barclay, A. N. & van den Berg, T. K. Signal regulatory proteins in the immune system. J. Immunol. 175, 7781–7787 (2005).
Streit, W. J. Microglial senescence: does the brain's immune system have an expiration date? Trends Neurosci. 29, 506–510 (2006).
Carare, R. O. et al. Solutes, but not cells, drain from the brain parenchyma along basement membranes of capillaries and arteries: significance for cerebral amyloid angiopathy and neuroimmunology. Neuropathol. Appl. Neurobiol. 34, 131–144 (2008).
Kreutzberg, G. W. Microglia: a sensor for pathological events in the CNS. Trends Neurosci. 19, 312–318 (1996).
Banati, R. B., Myers, R. & Kreutzberg, G. W. PK ('peripheral benzodiazepine')-binding sites in the CNS indicate early and discrete brain lesions: microautoradiographic detection of [3H]PK11195 binding to activated microglia. J. Neurocytol. 26, 77–82 (1997).
Banati, R. B. Visualising microglial activation in vivo. Glia 40, 206–217 (2002).
Edison, P. et al. Microglia, amyloid, and cognition in Alzheimer's disease: an [11C](R)PK11195-PET and [11C]PIB-PET study. Neurobiol. Dis. 32, 412–419 (2008).
Martinez, F. O., Sica, A., Mantovani, A. & Locati, M. Macrophage activation and polarization. Front Biosci. 13, 453–461 (2008).
Stout, R. D. et al. Macrophages sequentially change their functional phenotype in response to changes in microenvironmental influences. J. Immunol. 175, 342–349 (2005).
Stout, R. D., Watkins, S. K. & Suttles, J. Functional plasticity of macrophages: in situ reprogramming of tumor-associated macrophages. J. Leukoc. Biol. 86, 1105–1109 (2009).
Lassmann, H., Brück, W. & Lucchinetti, C. F. The immunopathology of multiple sclerosis: an overview. Brain Pathol. 17, 210–218 (2007).
Gold, R., Linington, C. & Lassmann, H. Understanding pathogenesis and therapy of multiple sclerosis via animal models: 70 years of merits and culprits in experimental autoimmune encephalomyelitis research. Brain 129, 1953–1971 (2006).
Bianchi, M. E. DAMPs, PAMPs and alarmins: all we need to know about danger. J. Leukoc. Biol. 81, 1–5 (2007).
Kono, H. & Rock, K. L. How dying cells alert the immune system to danger. Nat. Rev. Immunol. 8, 279–289 (2008).
Medzhitov, R. Recognition of microorganisms and activation of the immune response. Nature 449, 819–826 (2007).
Meylan, E., Tschopp, J. & Karin, M. Intracellular pattern recognition receptors in the host response. Nature 442, 39–44 (2006).
Salminen, A., Ojala, J., Kauppinen, A., Kaarniranta, K. & Suuronen, T. Inflammation in Alzheimer's disease: amyloid-β oligomers trigger innate immunity defence via pattern recognition receptors. Prog. Neurobiol. 87, 181–194 (2009).
Haynes, S. E. et al. The P2Y12 receptor regulates microglial activation by extracellular nucleotides. Nat. Neurosci. 9, 1512–1519 (2006).
Allan, S. M. & Rothwell, N. J. Inflammation in central nervous system injury. Philos. Trans. R. Soc. Lond. B Biol. Sci. 358, 1669–1677 (2003).
Wang, Q., Tang, X. N. & Yenari, M. A. The inflammatory response in stroke. J. Neuroimmunol. 184, 53–68 (2007).
Clausen, B. H. et al. Interleukin-1beta and tumor necrosis factor-alpha are expressed by different subsets of microglia and macrophages after ischemic stroke in mice. J. Neuroinflammation 5, 46 (2008).
White, F., Nicoll, J. A. & Horsburgh, K. Alterations in ApoE and ApoJ in relation to degeneration and regeneration in a mouse model of entorhinal cortex lesion. Exp. Neurol. 169, 307–318 (2001).
Miklossy, J. & Van der Loos, H. The long-distance effects of brain lesions: visualization of myelinated pathways in the human brain using polarizing and fluorescence microscopy. J. Neuropathol. Exp. Neurol. 50, 1–15 (1991).
Palin, K., Cunningham, C., Forse, P., Perry, V. H. & Platt, N. Systemic inflammation switches the inflammatory cytokine profile in CNS Wallerian degeneration. Neurobiol. Dis. 30, 19–29 (2008).
Boven, L. A. et al. Myelin-laden macrophages are anti-inflammatory, consistent with foam cells in multiple sclerosis. Brain 129, 517–526 (2006).
Griffin, W. S. et al. Brain interleukin 1 and S-100 immunoreactivity are elevated in Down syndrome and Alzheimer disease. Proc. Natl Acad. Sci. USA 86, 7611–7615 (1989).
Etminan, M., Gill, S. & Samii, A. Effect of non-steroidal anti-inflammatory drugs on risk of Alzheimer's disease: systematic review and meta-analysis of observational studies. BMJ 327, 128 (2003).
Vlad, S. C., Miller, D. R., Kowall, N. W. & Felson, D. T. Protective effects of NSAIDs on the development of Alzheimer disease. Neurology 70, 1672–1677 (2008).
Weggen, S., Rogers, M. & Eriksen, J. NSAIDs: small molecules for prevention of Alzheimer's disease or precursors for future drug development? Trends Pharmacol. Sci. 28, 536–543 (2007).
McGeer, P. L. & McGeer, E. G. NSAIDs and Alzheimer disease: epidemiological, animal model and clinical studies. Neurobiol. Aging 28, 639–647 (2007).
Matsuo, K. et al. Transforming growth factor-β is involved in the pathogenesis of dialysis-related amyloidosis. Kidney Int. 57, 697–708 (2000).
Pepys, M. B. Science and serendipity. Clin. Med. 7, 562–578 (2007).
Kalaria, R. N. & Grahovac, I. Serum amyloid P immunoreactivity in hippocampal tangles, plaques and vessels: implications for leakage across the blood–brain barrier in Alzheimer's disease. Brain Res. 516, 349–353 (1990).
Tennent, G. A., Lovat, L, B. & Pepys, M. B. Serum amyloid P component prevents proteolysis of the amyloid fibrils of Alzheimer disease and systemic amyloidosis. Proc. Natl Acad. Sci. USA 92, 4299–4303 (1995).
Veerhuis, R. et al. Amyloid β plaque-associated proteins C1q and SAP enhance the A β1–42 peptide-induced cytokine secretion by adult human microglia in vitro. Acta Neuropathol. 105, 135–144 (2003).
Hirsch, E. C. & Hunot, S. Neuroinflammation in Parkinson's disease: a target for neuroprotection? Lancet Neurol. 8, 382–397 (2009).
Samii, A., Etminan, M., Wiens, M. O. & Jafari, S. NSAID use and the risk of Parkinson's disease: systematic review and meta-analysis of observational studies. Drugs Aging 26, 769–779 (2009).
Herrera, A. J., Castaño, A., Venero, J. L., Cano, J. & Machado, A. The single intranigral injection of LPS as a new model for studying the selective effects of inflammatory reactions on dopaminergic system. Neurobiol. Dis. 7, 429–447 (2000).
Troost, D., Van den Oord, J. J. & Vianney de Jong, J. M. Immunohistochemical characterization of the inflammatory infiltrate in amyotrophic lateral sclerosis. Neuropathol. Appl. Neurobiol. 16, 401–410 (1990).
Clement, A. M. et al. Wild-type nonneuronal cells extend survival of SOD1 mutant motor neurons in ALS mice. Science 302, 113–117 (2003).
Boillée, S. et al. Onset and progression in inherited ALS determined by motor neurons and microglia. Science 312, 1389–1392 (2006).
Akiyama, H. et al. Inflammation and Alzheimer's disease. Neurobiol. Aging 21, 383–421 (2000).
Friedlander, R. M. Apoptosis and caspases in neurodegenerative diseases. N. Engl. J. Med. 348, 1365–1375 (2003).
Savill, J., Dransfield, I., Gregory, C. & Haslett, C. A blast from the past: clearance of apoptotic cells regulates immune responses. Nat. Rev. Immunol. 2, 965–975 (2002).
Scheff, S. W. & Price, D. A. Synaptic pathology in Alzheimer's disease: a review of ultrastructural studies. Neurobiol. Aging 24, 1029–1046 (2003).
Games, D., Buttini, M., Kobayashi, D., Schenk, D. & Seubert, P. Mice as models: transgenic approaches and Alzheimer's disease. J. Alzheimers Dis. 9, 133–149 (2006).
Moore, D. J. & Dawson, T. M. Value of genetic models in understanding the cause and mechanisms of Parkinson's disease. Curr. Neurol. Neurosci. Rep. 8, 288–296 (2008).
Brandt, R., Hundelt, M. & Shahani, N. Tau alteration and neuronal degeneration in tauopathies: mechanisms and models. Biochim. Biophys. Acta 1739, 331–354 (2005).
Kokjohn, T. A. & Roher, A. E. Amyloid precursor protein transgenic mouse models and Alzheimer's disease: understanding the paradigms, limitations, and contributions. Alzheimers Dement. 5, 340–347 (2009).
Radde, R., Duma, C., Goedert, M. & Jucker, M. The value of incomplete mouse models of Alzheimer's disease. Eur. J. Nucl. Med. Mol. Imaging 35, S70–S74 (2008).
Perry, V. H., Cunningham, C. & Holmes, C. Systemic infections and inflammation affect chronic neurodegeneration. Nat. Rev. Immunol. 7, 161–167 (2007).
Cunningham, C., Wilcockson, D. C., Campion, S., Lunnon, K. & Perry, V. H. Central and systemic endotoxin challenges exacerbate the local inflammatory response and increase neuronal death during chronic neurodegeneration. J. Neurosci. 25, 9275–9284 (2005).
Moran, L. B. & Graeber, M. B. The facial nerve axotomy model. Brain Res. Brain Res. Rev. 44, 154–178 (2004).
Wake, H., Moorhouse, A. J., Jinno, S., Kohsaka, S. & Nabekura, J. Resting microglia directly monitor the functional state of synapses in vivo and determine the fate of ischemic terminals. J. Neurosci. 29, 3974–3980 (2009).
Siskova, Z., Page, A., O'Connor, V. & Perry, V. H. Degenerating synaptic boutons in prion disease. Microglia activation without synaptic stripping. Am. J. Pathol. 175, 1610–1621 (2009).
Morgan, D., Gordon, M. N., Tan, J., Wilcock, D. & Rojiani, A. M. Dynamic complexity of the microglial activation response in transgenic models of amyloid deposition: implications for Alzheimer therapeutics. J. Neuropathol. Exp. Neurol. 64, 743–753 (2005).
Simard, A. R., Soulet, D., Gowing, G., Julien, J. P. & Rivest, S. Bone marrow-derived microglia play a critical role in restricting senile plaque formation in Alzheimer's disease. Neuron 49, 489–502 (2006).
Boissonneault, V. Powerful beneficial effects of macrophage colony-stimulating factor on β-amyloid deposition and cognitive impairment in Alzheimer's disease. Brain 132, 1078–1092 (2009).
Hamilton, J. A. Colony-stimulating factors in inflammation and autoimmunity. Nat. Rev. Immunol. 8, 533–544 (2008).
Ransohoff, R. M. Microgliosis: the questions shape the answers. Nat. Neurosci. 10, 1507–1509 (2007).
Jucker, M. & Heppner, F. L. Cerebral and peripheral amyloid phagocytes—an old liaison with a new twist. Neuron 59, 8–10 (2008).
Grathwohl, S. A. et al. Formation and maintenance of Alzheimer's disease β-amyloid plaques in the absence of microglia. Nat. Neurosci. 12, 1361–1363 (2009).
Schenk, D. et al. Immunization with amyloid-β attenuates Alzheimer-disease-like pathology in the PDAPP mouse. Nature 400, 173–177 (1999).
Janus, C. Vaccines for Alzheimer's disease: how close are we? CNS Drugs 17, 457–474 (2003).
Cunningham, C. et al. Systemic inflammation induces acute behavioral and cognitive changes and accelerates neurodegenerative disease. Biol. Psychiatry 15, 304–312 (2009).
Nguyen, M. D., D'Aigle, T., Gowing, G., Julien, J. P. & Rivest, S. Exacerbation of motor neuron disease by chronic stimulation of innate immunity in a mouse model of amyotrophic lateral sclerosis. J. Neurosci. 24, 1340–1349 (2004).
Godoy, M. C., Tarelli, R., Ferrari, C. C., Sarchi, M. I. & Pitossi, F. J. Central and systemic IL-1 exacerbates neurodegeneration and motor symptoms in a model of Parkinson's disease. Brain 131, 1880–1894 (2008).
Dantzer, R., O'Connor, J. C., Freund, G. G., Johnson, R. W. & Kelley, K. W. From inflammation to sickness and depression: when the immune system subjugates the brain. Nat. Rev. Neurosci. 9, 46–56 (2008).
Nicoll, J. A. et al. Neuropathology of human Alzheimer disease after immunization with amyloid-β peptide: a case report. Nat. Med. 9, 448–452 (2003).
Holmes, C. et al. Long-term effects of Aβ42 immunisation in Alzheimer's disease: follow-up of a randomised, placebo-controlled phase I trial. Lancet 372, 216–223 (2008).
Orgogozo, J. M. et al. Subacute meningoencephalitis in a subset of patients with AD after Aβ42 immunization. Neurology 61, 46–54 (2003).
Boche, D. et al. Consequence of Aβ immunization on the vasculature of human Alzheimer's disease brain. Brain 131, 299–310 (2008).
Fong, T. G., Tulebaev, S. R. & Inouye, S. K. Delirium in elderly adults: diagnosis, prevention and treatment. Nat. Rev. Neurol. 5, 210–220 (2009).
Fick, D. M., Agostini, J. V. & Inouye, S. K. Delirium superimposed on dementia: a systematic review. J. Am. Geriatr. Soc. 50, 1723–1732 (2002).
Fong, T. G. et al. Delirium accelerates cognitive decline in Alzheimer disease. Neurology 72, 1570–1575 (2009).
Holmes, C. et al. Systemic inflammation and disease progression in Alzheimer disease. Neurology 73, 768–774 (2009).
Luchsinger, J. A. Adiposity, hyperinsulinemia, diabetes and Alzheimer's disease: an epidemiological perspective. Eur. J. Pharmacol. 585, 119–129 (2008).
Qiu, C., De Ronchi, D. & Fratiglioni, L. The epidemiology of the dementias: an update. Curr. Opin. Psychiatry 20, 380–385 (2007).
Kodl, C. T. & Seaquist, E. R. Cognitive dysfunction and diabetes mellitus. Endocr. Rev. 29, 494–511 (2008).
Cechetto, D. F., Hachinski, V. & Whitehead, S. N. Vascular risk factors and Alzheimer's disease. Expert. Rev. Neurother. 8, 743–750 (2008).
Kamer, A. R. et al. Alzheimer's disease and peripheral infections: the possible contribution from periodontal infections, model and hypothesis. J. Alzheimers Dis. 13, 437–449 (2008).
Harold, D. et al. Genome-wide association study identifies variants at CLU and PICALM associated with Alzheimer's disease. Nat. Genet. 41, 1088–1093 (2009).
Lambert, J. C. et al. Genome-wide association study identifies variants at CLU and CR1 associated with Alzheimer's disease. Nat. Genet. 41, 1094–1099 (2009).
Acknowledgements
Work in the authors' laboratories is supported by the Medical Research Council, the Alzheimer's Research Trust, the Alzheimer's Society, and the Wellcome Trust. We are grateful to Prof. Peter Beverley for discussions on innate immune memory.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Perry, V., Nicoll, J. & Holmes, C. Microglia in neurodegenerative disease. Nat Rev Neurol 6, 193–201 (2010). https://doi.org/10.1038/nrneurol.2010.17
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2010.17
This article is cited by
-
Nanotechnology for microglial targeting and inhibition of neuroinflammation underlying Alzheimer’s pathology
Translational Neurodegeneration (2024)
-
Susceptibility to acute cognitive dysfunction in aged mice is underpinned by reduced white matter integrity and microgliosis
Communications Biology (2024)
-
Aerobic Glycolysis Induced by mTOR/HIF-1α Promotes Early Brain Injury After Subarachnoid Hemorrhage via Activating M1 Microglia
Translational Stroke Research (2024)
-
Hippocampal Microglia Activation Induced by Acute Pancreatic Injury in Rats
Digestive Diseases and Sciences (2024)
-
Rapamycin Alleviates Neuronal Injury and Modulates Microglial Activation After Cerebral Ischemia
Molecular Neurobiology (2024)