Abstract
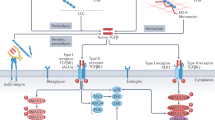
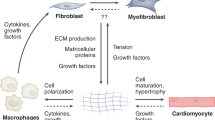
The syncytium of cardiomyocytes in the heart is tethered within a matrix composed principally of type I fibrillar collagen. The matrix has diverse mechanical functions that ensure the optimal contractile efficiency of this muscular pump. In the diseased heart, cardiomyocytes are lost to necrotic cell death, and phenotypically transformed fibroblast-like cells—termed 'myofibroblasts'—are activated to initiate a 'reparative' fibrosis. The structural integrity of the myocardium is preserved by this scar tissue, although at the expense of its remodelled architecture, which has increased tissue stiffness and propensity to arrhythmias. A persisting population of activated myofibroblasts turns this fibrous tissue into a living 'secretome' that generates angiotensin II and its type 1 receptor, and fibrogenic growth factors (such as transforming growth factor-β), all of which collectively act as a signal–transducer–effector signalling pathway to type I collagen synthesis and, therefore, fibrosis. Persistent myofibroblasts, and the resultant fibrous tissue they produce, cause progressive adverse myocardial remodelling, a pathological hallmark of the failing heart irrespective of its etiologic origin. Herein, we review relevant cellular, subcellular, and molecular mechanisms integral to cardiac fibrosis and consequent remodelling of atria and ventricles with a heterogeneity in cardiomyocyte size. Signalling pathways that antagonize collagen fibrillogenesis provide novel strategies for cardioprotection.
Key Points
-
The muscular parenchyma of the heart, a syncytium of cardiomyocytes, is tethered within a structural protein network primarily composed of type I fibrillar collagen
-
The matrix promotes transmission and coordination of forces generated within myofibres, prevents myofibre slippage while sustaining chamber geometry without deformation, and protects against myocardial rupture
-
When cardiomyocytes are lost to necrosis, fibroblast-like cells restore structural integrity of the myocardium and form a 'secretome' that exerts autocrine and paracrine actions to regulate collagen turnover
-
An adverse cell–cell interaction ensues between persistent myofibroblasts and cardiomyocytes, which negatively influences electrical behaviour of the myocardium, predisposing it to arrhythmias
-
Tendrils of myofibroblast-generated collagen can ensnare cardiomyocytes, resulting in reduced workload and, therefore, disuse atrophy of these cells
-
Key targets for downregulating matrix responses and, therefore, for cardioprotection lie in the myofibroblast secretome
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Weber, K. T. Cardiac interstitium in health and disease: the fibrillar collagen network. J. Am. Coll. Cardiol. 13, 1637–1652 (1989).
Laurent, G. J. Dynamic state of collagen: pathways of collagen degradation in vivo and their possible role in regulation of collagen mass. Am. J. Physiol. 252, C1–C9 (1987).
Schmitt-Gräff, A., Desmoulière, A. & Gabbiani, G. Heterogeneity of myofibroblast phenotypic features: an example of fibroblastic cell plasticity. Virchows Arch. 425, 3–24 (1994).
Gerling, I. C. et al. Aldosteronism: an immunostimulatory state precedes the proinflammatory/fibrogenic cardiac phenotype. Am. J. Physiol. Heart Circ. Physiol. 285, H813–H821 (2003).
Ahokas, R. A. et al. Aldosteronism and peripheral blood mononuclear cell activation. A neuroendocrine-immune interface. Circ. Res. 93, e124–e135 (2003).
Harrison, D. G., Cai, H., Landmesser, U. & Griendling, K. K. Interactions of angiotensin II with NAD(P)H oxidase, oxidant stress and cardiovascular disease. J. Renin Angiotensin Aldosterone Syst. 4, 51–61 (2003).
Harrison, D. G., Marvar, P. J. & Titze, J. M. Vascular inflammatory cells in hypertension. Front. Physiol. 3, 128 (2012).
Coen, M., Gabbiani, G. & Bochaton-Piallat, M. L. Myofibroblast-mediated adventitial remodeling: an underestimated player in arterial pathology. Arterioscler. Thromb. Vasc. Biol. 31, 2391–2396 (2011).
Rosker, C., Salvarani, N., Schmutz, S., Grand, T. & Rohr, S. Abolishing myofibroblast arrhythmogeneicity by pharmacological ablation of alpha-smooth muscle actin containing stress fibers. Circ. Res. 109, 1120–1131 (2011).
Yue, L., Xie, J. & Nattel, S. Molecular determinants of cardiac fibroblast electrical function and therapeutic implications for atrial fibrillation. Cardiovasc. Res. 89, 744–753 (2011).
Jalil, J. E., Janicki, J. S., Pick, R., Abrahams, C. & Weber, K. T. Fibrosis-induced reduction of endomyocardium in the rat after isoproterenol treatment. Circ. Res. 65, 258–264 (1989).
Fidzian´ska, A., Bilin´ska, Z. T., Walczak, E., Witkowski, A. & Chojnowska, L. Autophagy in transition from hypertrophic cardiomyopathy to heart failure. J. Electron Microsc. (Tokyo) 59, 181–183 (2010).
Shang, F. & Taylor, A. Ubiquitin-proteasome pathway and cellular responses to oxidative stress. Free Radic. Biol. Med. 51, 5–16 (2011).
Rodriguez, J. E., Schisler, J. C., Patterson, C. & Willis, M. S. Seek and destroy: the ubiquitin—proteasome system in cardiac disease. Curr. Hypertens. Rep. 11, 396–405 (2009).
Cosper, P. F. & Leinwand, L. A. Cancer causes cardiac atrophy and autophagy in a sexually dimorphic manner. Cancer Res. 71, 1710–1720 (2011).
Beltrami, C. A. et al. Structural basis of end-stage failure in ischemic cardiomyopathy in humans. Circulation 89, 151–163 (1994).
Factor, S. M. et al. Pathologic fibrosis and matrix connective tissue in the subaortic myocardium of patients with hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 17, 1343–1351 (1991).
Waller, T. A., Hiser, W. L., Capehart, J. E. & Roberts, W. C. Comparison of clinical and morphologic cardiac findings in patients having cardiac transplantation for ischemic cardiomyopathy, idiopathic dilated cardiomyopathy, and dilated hypertrophic cardiomyopathy. Am. J. Cardiol. 81, 884–894 (1998).
Schaper, J., Lorenz-Meyer, S. & Suzuki, K. The role of apoptosis in dilated cardiomyopathy. Herz 24, 219–224 (1999).
de Leeuw, N. et al. Histopathologic findings in explanted heart tissue from patients with end-stage idiopathic dilated cardiomyopathy. Transpl. Int. 14, 299–306 (2001).
Yoshikane, H. et al. Collagen in dilated cardiomyopathy--scanning electron microscopic and immunohistochemical observations. Jpn Circ. J. 56, 899–910 (1992).
Marijianowski, M. M., Teeling, P., Mann, J. & Becker, A. E. Dilated cardiomyopathy is associated with an increase in the type I/type III collagen ratio: a quantitative assessment. J. Am. Coll. Cardiol. 25, 1263–1272 (1995).
Pearlman, E. S., Weber, K. T., Janicki, J. S., Pietra, G. G. & Fishman, A. P. Muscle fiber orientation and connective tissue content in the hypertrophied human heart. Lab. Invest. 46, 158–164 (1982).
Huysman, J. A. N., Vliegen, H. W., Van der Laarse, A. & Eulderink, F. Changes in nonmyocyte tissue composition associated with pressure overload of hypertrophic human hearts. Pathol. Res. Pract. 184, 577–581 (1989).
Rossi, M. A. Pathologic fibrosis and connective tissue matrix in left ventricular hypertrophy due to chronic arterial hypertension in humans. J. Hypertens. 16, 1031–1041 (1998).
Lopez, B., Gonzalez, A., Querejeta, R., Larman, M. & Diez, J. Alterations in the pattern of collagen deposition may contribute to the deterioration of systolic function in hypertensive patients with heart failure. J. Am. Coll. Cardiol. 48, 89–96 (2006).
Krayenbuehl, H. P. et al. Left ventricular myocardial structure in aortic valve disease before, intermediate, and late after aortic valve replacement. Circulation 79, 744–755 (1989).
Schwarz, F. et al. Myocardial structure and function in patients with aortic valve disease and their relation to postoperative results. Am. J. Cardiol. 41, 661–669 (1978).
Hein, S. et al. Progression from compensated hypertrophy to failure in the pressure-overloaded human heart: structural deterioration and compensatory mechanisms. Circulation 107, 984–991 (2003).
Brooks, W. W., Shen, S. S., Conrad, C. H., Goldstein, R. H. & Bing, O. H. Transition from compensated hypertrophy to systolic heart failure in the spontaneously hypertensive rat: Structure, function, and transcript analysis. Genomics 95, 84–92 (2010).
O'Hanlon, R. et al. Prognostic significance of myocardial fibrosis in hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 56, 867–874 (2010).
Green, J. J., Berger, J. S., Kramer, C. M. & Salerno, M. Prognostic value of late gadolinium enhancement in clinical outcomes for hypertrophic cardiomyopathy. JACC Cardiovasc. Imaging 5, 370–377 (2012).
Weber, K. T. et al. Collagen remodeling of the pressure-overloaded, hypertrophied nonhuman primate myocardium. Circ. Res. 62, 757–765 (1988).
Chapman, D., Weber, K. T. & Eghbali, M. Regulation of fibrillar collagen types I and III and basement membrane type IV collagen gene expression in pressure overloaded rat myocardium. Circ. Res. 67, 787–794 (1990).
Borg, T. K., Sullivan, T. & Ivy, J. Functional arrangement of connective tissue in striated muscle with emphasis on cardiac muscle. Scanning Electron Microsc. 4, 1775–1784 (1982).
Robinson, T. F., Cohen-Gould, L. & Factor, S. M. Skeletal framework of mammalian heart muscle. Arrangement of inter- and pericellular connective tissue structures. Lab. Invest. 49, 482–498 (1983).
Robinson, T. F., Factor, S. M. & Sonnenblick, E. H. The heart as a suction pump. Sci. Am. 254, 84–91 (1986).
Cooper, G. 4th. Cytoskeletal networks and the regulation of cardiac contractility: microtubules, hypertrophy, and cardiac dysfunction. Am. J. Physiol. Heart Circ. Physiol. 291, H1003–H1014 (2006).
Hoshijima, M. Mechanical stress-strain sensors embedded in cardiac cytoskeleton: Z disk, titin, and associated structures. Am. J. Physiol. Heart Circ. Physiol. 290, H1313–H1325 (2006).
Voelkel, T. & Linke, W. A. Conformation-regulated mechanosensory control via titin domains in cardiac muscle. Pflugers Arch. 462, 143–154 (2011).
Streeter, D. D., Spotnitz, H. M., Patel, D. P., Ross, J. & Sonnenblick, E. H. Fiber orientation in the canine left ventricle during diastole and systole. Circ. Res. 24, 339–347 (1969).
Robinson, T. F., Geraci, M. A., Sonnenblick, E. H. & Factor, S. M. Coiled perimysial fibers of papillary muscle in rat heart: morphology, distribution, and changes in configuration. Circ. Res. 63, 577–592 (1988).
Robinson, T. F., Cohen-Gould, L., Factor, S. M., Eghbali, M. & Blumenfeld, O. O. Structure and function of connective tissue in cardiac muscle: collagen types I and III in endomysial struts and pericellular fibers. Scanning Microsc. 2, 1005–1015 (1988).
Lerman, R. H. et al. Myocardial healing and repair after experimental infarction in the rabbit. Circ. Res. 53, 378–388 (1983).
Jugdutt, B. I. Left ventricular rupture threshold during the healing phase after myocardial infarction in the dog. Can. J. Physiol. Pharmacol. 65, 307–316 (1987).
Factor, S. M., Robinson, T. F., Dominitz, R. & Cho, S. H. Alterations of the myocardial skeletal framework in acute myocardial infarction with and without ventricular rupture. Am. J. Cardiovasc. Pathol. 1, 91–97 (1987).
Spinale, F. G. Matrix metalloproteinases: regulation and dysregulation in the failing heart. Circ. Res. 90, 520–530 (2002).
Zhao, M. J. et al. Profound structural alterations of the extracellular collagen matrix in postischemic dysfunctional (“stunned”) but viable myocardium. J. Am. Coll. Cardiol. 10, 1322–1334 (1987).
Rushmer, R. F. & Thal, N. The mechanics of ventricular contraction; a cinefluorographic study. Circulation 4, 219–228 (1951).
Fleckenstein, A., Frey, M. & Fleckenstein-Grun, G. Consequences of uncontrolled calcium entry and its prevention with calcium antagonists. Eur. Heart J. 4, 43–50 (1983).
Borkowski, B. J., Cheema, Y., Shahbaz, A. U., Bhattacharya, S. K. & Weber, K. T. Cation dyshomeostasis and cardiomyocyte necrosis. The Fleckenstein hypothesis revisited. Eur. Heart J. 32, 1846–1853 (2011).
Khan, M. U. et al. Mitochondria play a central role in nonischemic cardiomyocyte necrosis: common to acute and chronic stressor states. Pflügers Arch. 464, 123–131 (2012).
Garcia-Dorado, D., Ruiz-Meana, M., Inserte, J., Rodriguez-Sinovas, A. & Piper, H. M. Calcium-mediated cell death during myocardial reperfusion. Cardiovasc. Res. 94, 168–180 (2012).
Palmieri, G. M., Nutting, D. F., Bhattacharya, S. K., Bertorini, T. E. & Williams, J. C. Parathyroid ablation in dystrophic hamsters. Effects on Ca content and histology of heart, diaphragm, and rectus femoris. J. Clin. Invest. 68, 646–654 (1981).
Jung, C., Martins, A. S., Niggli, E. & Shirokova, N. Dystrophic cardiomyopathy: amplification of cellular damage by Ca2+ signalling and reactive oxygen species-generating pathways. Cardiovasc. Res. 77, 766–773 (2008).
Matzinger, P. The danger model: a renewed sense of self. Science 296, 301–305 (2002).
Cleutjens, J. P. M., Kandala, J. C., Guarda, E., Guntaka, R. V. & Weber, K. T. Regulation of collagen degradation in the rat myocardium after infarction. J. Mol. Cell. Cardiol. 27, 1281–1292 (1995).
Sun, Y., Zhang, J., Zhang, J. Q. & Weber, K. T. Renin expression at sites of repair in the infarcted rat heart. J. Mol. Cell. Cardiol. 33, 995–1003 (2001).
Sun, Y., Cleutjens, J. P. M., Diaz-Arias, A. A. & Weber, K. T. Cardiac angiotensin converting enzyme and myocardial fibrosis in the rat. Cardiovasc. Res. 28, 1423–1432 (1994).
Sun, Y. & Weber, K. T. Cells expressing angiotensin II receptors in fibrous tissue of rat heart. Cardiovasc. Res. 31, 518–525 (1996).
Sun, Y., Zhang, J. Q., Zhang, J. & Ramires, F. J. A. Angiotensin II, transforming growth factor-β1 and repair in the infarcted heart. J. Mol. Cell. Cardiol. 30, 1559–1569 (1998).
Horiguchi, M., Ota, M. & Rifkin, D. B. Matrix control of transforming growth factor-β function. J. Biochem. 152, 321–329 (2012).
Filip, D. A., Radu, A. & Simionescu, M. Interstitial cells of the heart valves possess characteristics similar to smooth muscle cells. Circ. Res. 59, 310–320 (1986).
Katwa, L. C. et al. Valvular interstitial cells express angiotensinogen, cathepsin D, and generate angiotensin peptides. Int. J. Biochem. Cell Biol. 28, 807–821 (1996).
Bondi, C. D. et al. NAD(P)H oxidase mediates TGF-β1-induced activation of kidney myofibroblasts. J. Am. Soc. Nephrol. 21, 93–102 (2010).
Barker, T. H. et al. SPARC regulates extracellular matrix organization through its modulation of integrin-linked kinase activity. J. Biol. Chem. 280, 36483–36493 (2005).
Cleutjens, J. P. M., Verluyten, M. J. A., Smits, J. F. M. & Daemen, M. J. A. P. Collagen remodeling after myocardial infarction in the rat heart. Am. J. Pathol. 147, 325–338 (1995).
Chapman, D. & Eghbali, M. Expression of fibrillar types I and III and basement membrane collagen type IV genes in myocardium of tight skin mouse. Cardiovasc. Res. 24, 578–583 (1990).
Whittaker, P. Unravelling the mysteries of collagen and cicatrix after myocardial infarction. Cardiovasc. Res. 31, 19–27 (1996).
Fomovsky, G. M., Rouillard, A. D. & Holmes, J. W. Regional mechanics determine collagen fiber structure in healing myocardial infarcts. J. Mol. Cell. Cardiol. 52, 1083–1090 (2012).
Sun, Y., Kiani, M. F., Postlethwaite, A. E. & Weber, K. T. Infarct scar as living tissue. Basic Res. Cardiol. 97, 343–347 (2002).
van den Borne, S. W. et al. Myocardial remodeling after infarction: the role of myofibroblasts. Nat. Rev. Cardiol. 7, 30–37 (2010).
Sun, Y., Zhang, J. Q., Zhang, J. & Ramires, F. J. A. Angiotensin II, transforming growth factor-β1 and repair in the infarcted heart. J. Mol. Cell. Cardiol. 30, 1559–1569 (1998).
Sun, Y. & Weber, K. T. Angiotensin-converting enzyme and wound healing in diverse tissues of the rat. J. Lab. Clin. Med. 127, 94–101 (1996).
Sun, Y., Ramires, F. J. A., Zhou, G., Ganjam, V. K. & Weber, K. T. Fibrous tissue and angiotensin II. J. Mol. Cell. Cardiol. 29, 2001–2012 (1997).
Katwa, L. C. et al. Pouch tissue and angiotensin peptide generation. J. Mol. Cell. Cardiol. 30, 1401–1413 (1998).
Sun, Y. et al. Tissue angiotensin II in the regulation of inflammatory and fibrogenic components of repair in the rat heart. J. Lab. Clin. Med. 143, 41–51 (2004).
Sun, Y. & Weber, K. T. Angiotensin II and aldosterone receptor binding in rat heart and kidney: response to chronic angiotensin II or aldosterone administration. J. Lab. Clin. Med. 122, 404–411 (1993).
Katwa, L. C. et al. Cultured myofibroblasts generate angiotensin peptides de novo. J. Mol. Cell. Cardiol. 29, 1375–1386 (1997).
Campbell, S. E. & Katwa, L. C. Angiotensin II stimulated expression of transforming growth factor-beta 1 in cardiac fibroblasts and myofibroblasts. J. Mol. Cell. Cardiol. 29, 1947–1958 (1997).
Wang, Q. et al. Cooperative interaction of CTGF and TGF-β in animal models of fibrotic disease. Fibrogenesis Tissue Repair 4, 4 (2011).
Arnott, J. A. et al. Molecular requirements for induction of CTGF expression by TGF-β1 in primary osteoblasts. Bone 42, 871–885 (2008).
Wynn, T. A. Cellular and molecular mechanisms of fibrosis. J. Pathol. 214, 199–210 (2008).
Ye, H., Cai, P. C., Zhou, Q. & Ma, W. L. Transforming growth factor-β1 suppresses the up-regulation of matrix metalloproteinase-2 by lung fibroblasts in response to tumor necrosis factor-α. Wound Repair Regen. 19, 392–399 (2011).
Willems, I. E. M. G., Havenith, M. G., De Mey, J. G. R. & Daemen, M. J. A. P. The α-smooth muscle actin-positive cells in healing human myocardial scars. Am. J. Pathol. 145, 868–875 (1994).
Follonier Castella, L., Gabbiani, G., McCulloch, C. A. & Hinz, B. Regulation of myofibroblast activities: calcium pulls some strings behind the scene. Exp. Cell Res. 316, 2390–2401 (2010).
Cotran, R. S., Kumar, V. & Robbins, S. L. in Robbins Pathologic Basis of Disease (eds Cotran, R. S., Kumar, V. & Robbins, S. L.) 597–656 (W. B. Saunders, Philadelphia, 1989).
Choudhury, L. et al. Myocardial scarring in asymptomatic or mildly symptomatic patients with hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 40, 2156–2164 (2002).
Maron, B. J., Epstein, S. E. & Roberts, W. C. Hypertrophic cardiomyopathy and transmural myocardial infarction without significant atherosclerosis of the extramural coronary arteries. Am. J. Cardiol. 43, 1086–1102 (1979).
Finsterer, J. & Stöllberger, C. The heart in human dystrophinopathies. Cardiology 99, 1–19 (2003).
Swynghedauw, B. Molecular Cardiology for the Cardiologist (Kluwer, Boston, 1995).
Campbell, S. E., Rakusan, K. & Gerdes, A. M. Change in cardiac myocyte size distribution in aortic-constricted neonatal rats. Basic Res. Cardiol. 84, 247–258 (1989).
Pandya, K., Kim, H. S. & Smithies, O. Fibrosis, not cell size, delineates β-myosin heavy chain reexpression during cardiac hypertrophy and normal aging in vivo. Proc. Natl Acad. Sci. USA 103, 16864–16869 (2006).
López, J. E. et al. β-myosin heavy chain is induced by pressure overload in a minor subpopulation of smaller mouse cardiac myocytes. Circ. Res. 109, 629–638 (2011).
López, B., Querejeta, R., González, A., Larman, M. & Díez, J. Collagen cross-linking but not collagen amount associates with elevated filling pressures in hypertensive patients with stage C heart failure: potential role of lysyl oxidase. Hypertension 60, 677–683 (2012).
Diez, J., Lopez, B., Gonzalez, A. & Querejeta, R. Clinical aspects of hypertensive myocardial fibrosis. Curr. Opin. Cardiol. 16, 328–335 (2001).
Weber, K. T., Brilla, C. G. & Janicki, J. S. Myocardial fibrosis: functional significance and regulatory factors. Cardiovasc. Res. 27, 341–348 (1993).
Bing, O. H. L., Fanburg, B. L., Brooks, W. W. & Matsushita, S. The effect of the lathyrogen β-amino proprionitrile (BAPN) on the mechanical properties of experimentally hypertrophied rat cardiac muscle. Circ. Res. 43, 632–637 (1978).
Westermann, D. et al. Cardiac inflammation contributes to changes in the extracellular matrix in patients with heart failure and normal ejection fraction. Circ. Heart Fail. 4, 44–52 (2011).
Martos, R. et al. Diastolic heart failure: evidence of increased myocardial collagen turnover linked to diastolic dysfunction. Circulation 115, 888–895 (2007).
Gabbiani, G., Ryan, G. B. & Majno, G. Presence of modified fibroblasts in granulation tissue and their possible role in wound contraction. Experientia 27, 549–550 (1971).
Lorell, B. H. Diastolic dysfunction in pressure-overload hypertrophy and its modification by angiotensin II: current concepts. Basic Res. Cardiol. 87 (Suppl. 2), 163–172 (1992).
Friedrich, S. P. et al. Intracardiac angiotensin-converting enzyme inhibition improves diastolic function in patients with left ventricular hypertrophy due to aortic stenosis. Circulation 90, 2761–2771 (1994).
Karagueuzian, H. S. Targeting cardiac fibrosis: a new frontier in antiarrhythmic therapy? Am. J. Cardiovasc. Dis. 1, 101–109 (2011).
Hothi, S. S. et al. Epac activation, altered calcium homeostasis and ventricular arrhythmogenesis in the murine heart. Pflugers Arch. 457, 253–270 (2008).
Rohr, S. Myofibroblasts in diseased hearts: new players in cardiac arrhythmias? Heart Rhythm 6, 848–856 (2009).
Polyakova, V., Miyagawa, S., Szalay, Z., Risteli, J. & Kostin, S. Atrial extracellular matrix remodelling in patients with atrial fibrillation. J. Cell. Mol. Med. 12, 189–208 (2008).
Kallergis, E. M. et al. Extracellular matrix alterations in patients with paroxysmal and persistent atrial fibrillation: biochemical assessment of collagen type-I turnover. J. Am. Coll. Cardiol. 52, 211–215 (2008).
Weber, K. T., Janicki, J. S. & Fishman, A. P. Aerobic limit of the heart perfused at constant pressure. Am. J. Physiol. 238, H118–H125 (1980).
Brilla, C. G., Janicki, J. S. & Weber, K. T. Cardioreparative effects of lisinopril in rats with genetic hypertension and left ventricular hypertrophy. Circulation 83, 1771–1779 (1991).
Youn, H. J. et al. Relation between flow reserve capacity of penetrating intramyocardial coronary arteries and myocardial fibrosis in hypertension: study using transthoracic Doppler echocardiography. J. Am. Soc. Echocardiogr. 19, 373–378 (2006).
Warnes, C. A., Maron, B. J. & Roberts, W. C. Massive cardiac ventricular scarring in first-degree relatives with hypertrophic cardiomyopathy. Am. J. Cardiol. 54, 1377–1379 (1984).
Maron, B. J., Wolfson, J. K., Epstein, S. E. & Roberts, W. C. Intramural (“small vessel”) coronary artery disease in hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 8, 545–557 (1986).
Olivotto, I. et al. Microvascular function is selectively impaired in patients with hypertrophic cardiomyopathy and sarcomere myofilament gene mutations. J. Am. Coll. Cardiol. 58, 839–48 (2011).
Weber, K. T., Jalil, J. E., Janicki, J. S. & Pick, R. Myocardial collagen remodeling in pressure overload hypertrophy: a case for interstitial heart disease. Am. J. Hypertens. 2, 931–940 (1989).
Collier, P., Ledwidge, M. & McDonald, K. Diagnostics and therapeutic interventions in myocardial interstitial disease, a previously neglected pathology. QJM 105, 721–724 (2012).
Griffiths, E. J. Mitochondria and heart disease. Adv. Exp. Med. Biol. 942, 249–267 (2012).
Dai, D. F. et al. Mitochondrial targeted antioxidant peptide ameliorates hypertensive cardiomyopathy. J. Am. Coll. Cardiol. 58, 73–82 (2011).
Shahbaz, A. U. et al. Mitochondria-targeted cardioprotection in aldosteronism. J. Cardiovasc. Pharmacol. 57, 37–43 (2011).
Cheema, Y. et al. Mitochondriocentric pathway to cardiomyocyte necrosis in aldosteronism: cardioprotective responses to carvedilol and nebivolol. J. Cardiovasc. Pharmacol. 58, 80–86 (2011).
Yoshida, A. et al. H2 mediates cardioprotection via involvements of KATP channels and permeability transition pores of mitochondria in dogs. Cardiovasc. Drugs Ther. 26, 217–226 (2012).
Aurora, A. B. et al. MicroRNA-214 protects the mouse heart from ischemic injury by controlling Ca2+ overload and cell death. J. Clin. Invest. 122, 1222–1232 (2012).
Bauersachs, J. Regulation of myocardial fibrosis by MicroRNAs. J. Cardiovasc. Pharmacol. 56, 454–459 (2010).
Thum, T. & Lorenzen, J. M. Cardiac fibrosis revisited by microRNA therapeutics. Circulation 126, 800–802 (2012).
Gieling, R. G., Burt, A. D. & Mann, D. A. Fibrosis and cirrhosis reversibility - molecular mechanisms. Clin. Liver Dis. 12, 915–937 (2008).
Elsharkawy, A. M., Oakley, F. & Mann, D. A. The role and regulation of hepatic stellate cell apoptosis in reversal of liver fibrosis. Apoptosis 10, 927–939 (2005).
Muddu, A. K., Guha, I. N., Elsharkawy, A. M. & Mann, D. A. Resolving fibrosis in the diseased liver: translating the scientific promise to the clinic. Int. J. Biochem. Cell Biol. 39, 695–714 (2007).
Smits, J. F. M., van Krimpen, C., Schoemaker, R. G., Cleutjens, J. P. M. & Daemen, M. J. A. P. Angiotensin II receptor blockade after myocardial infarction in rats: effects on hemodynamics, myocardial DNA synthesis, and interstitial collagen content. J. Cardiovasc. Pharmacol. 20, 772–778 (1992).
Tsutsui, H. et al. Angiotensin II type 1 receptor blocker attenuates myocardial remodeling and preserves diastolic function in diabetic heart. Hypertens. Res. 30, 439–449 (2007).
Matsusaka, H. et al. Angiotensin II type 1 receptor blocker attenuates exacerbated left ventricular remodeling and failure in diabetes-associated myocardial infarction. J. Cardiovasc. Pharmacol. 48, 95–102 (2006).
Iwamoto, M. et al. Connective tissue growth factor induction in a pressure-overloaded heart ameliorated by the angiotensin II type 1 receptor blocker olmesartan. Hypertens. Res. 33, 1305–1311 (2010).
Hoch, N. E. et al. Regulation of T-cell function by endogenously produced angiotensin II. Am. J. Physiol. Regul. Integr. Comp. Physiol. 296, R208–R216 (2009).
Touyz, R. M. Intracellular mechanisms involved in vascular remodelling of resistance arteries in hypertension: role of angiotensin II. Exp. Physiol. 90, 449–55 (2005).
Fujiwara, Y. et al. Inhibition of experimental abdominal aortic aneurysm in a rat model by the angiotensin receptor blocker valsartan. Int. J. Mol. Med. 22, 703–708 (2008).
Habashi, J. P. et al. Losartan, an AT1 antagonist, prevents aortic aneurysm in a mouse model of Marfan syndrome. Science 312, 117–121 (2006).
Moltzer, E. et al. Impaired vascular contractility and aortic wall degeneration in fibulin-4 deficient mice: effect of angiotensin II type 1 (AT1) receptor blockade. PLoS ONE 6, e23411 (2011).
Aksu, S. et al. Over-expression of angiotensin-converting enzyme (CD 143) on leukemic blasts as a clue for the activated local bone marrow RAS in AML. Leuk. Lymphoma 47, 891–896 (2006).
Haznedaroglu, I. C. & Beyazit, Y. Pathobiological aspects of the local bone marrow renin-angiotensin system: a review. J. Renin Angiotensin. Aldosterone Syst. 11, 205–213 (2010).
Garcia, G. E. ANG II receptor antagonists as modulators of macrophages polarization. Am. J. Physiol. Renal Physiol. 298, F868–F869 (2010).
Rehman, A. et al. Angiotensin type 2 receptor agonist compound 21 reduces vascular injury and myocardial fibrosis in stroke-prone spontaneously hypertensive rats. Hypertension 59, 291–299 (2012).
Keidar, S., Kaplan, M. & Gamliel-Lazarovich, A. ACE2 of the heart: from angiotensin I to angiotensin (1–7). Cardiovasc. Res. 73, 463–469 (2007).
Zisman, L. S. et al. Increased angiotensin-(1–7)-forming activity in failing human heart ventricles: evidence for upregulation of the angiotensin-converting enzyme Homologue ACE2. Circulation 108, 1707–1712 (2003).
Burrell, L. M. et al. Myocardial infarction increases ACE2 expression in rat and humans. Eur. Heart J. 26, 369–375; discussion 322–324 (2005).
Takeda, Y. et al. Effects of aldosterone and angiotensin II receptor blockade on cardiac angiotensinogen and angiotensin-converting enzyme 2 expression in Dahl salt-sensitive hypertensive rats. Am. J. Hypertens. 20, 1119–1124 (2007).
Sukumaran, V. et al. Cardioprotective effects of telmisartan against heart failure in rats induced by experimental autoimmune myocarditis through the modulation of angiotensin-converting enzyme-2/angiotensin 1–7/mas receptor axis. Int. J. Biol. Sci. 7, 1077–1092 (2011).
Kassiri, Z. et al. Loss of angiotensin-converting enzyme 2 accelerates maladaptive left ventricular remodeling in response to myocardial infarction. Circ. Heart Fail. 2, 446–455 (2009).
Zhao, Y. X. et al. ACE2 overexpression ameliorates left ventricular remodeling and dysfunction in a rat model of myocardial infarction. Hum. Gene Ther. 21, 1545–1554 (2010).
Dong, B. et al. Angiotensin-converting enzyme-2 overexpression improves left ventricular remodeling and function in a rat model of diabetic cardiomyopathy. J. Am. Coll. Cardiol. 59, 739–747 (2012).
Trask, A. J. et al. Inhibition of angiotensin-converting enzyme 2 exacerbates cardiac hypertrophy and fibrosis in Ren-2 hypertensive rats. Am. J. Hypertens. 23, 687–693 (2010).
Liu, X., Hu, H. & Yin, J. Q. Therapeutic strategies against TGF-β signaling pathway in hepatic fibrosis. Liver Int. 26, 8–22 (2006).
Yang, F., Chung, A. C., Huang, X. R. & Lan, H. Y. Angiotensin II induces connective tissue growth factor and collagen I expression via transforming growth factor-β-dependent and -independent Smad pathways: the role of Smad3. Hypertension 54, 877–884 (2009).
Lan, H. Y. Transforming growth factor-β/Smad signalling in diabetic nephropathy. Clin. Exp. Pharmacol. Physiol. 39, 731–738 (2012).
Hinz, B. et al. Recent developments in myofibroblast biology: paradigms for connective tissue remodeling. Am. J. Pathol. 180, 1340–1355 (2012).
Hawinkels, L. J. & Ten Dijke, P. Exploring anti-TGF-β therapies in cancer and fibrosis. Growth Factors 29, 140–152 (2011).
Bauersachs, J. Regulation of myocardial fibrosis by MicroRNAs. J. Cardiovasc. Pharmacol. 56, 454–459 (2010).
Guntaka, R. V., Varma, B. R. & Weber, K. T. Triplex-forming oligonucleotides as modulators of gene expression. Int. J. Biochem. Cell Biol. 35, 22–31 (2003).
Koilan, S. et al. Prevention of liver fibrosis by triple helix-forming oligodeoxyribonucleotides targeted to the promoter region of type I collagen gene. Oligonucleotides 20, 231–237 (2010).
Brilla, C. G., Funck, R. C. & Rupp, H. Lisinopril-mediated regression of myocardial fibrosis in patients with hypertensive heart disease. Circulation 102, 1388–1393 (2000).
Varo, N. et al. Chronic AT1 blockade stimulates extracellular collagen type I degradation and reverses myocardial fibrosis in spontaneously hypertensive rats. Hypertension 35, 1197–1202 (2000).
Díez, J. et al. Losartan-dependent regression of myocardial fibrosis is associated with reduction of left ventricular chamber stiffness in hypertensive patients. Circulation 105, 2512–2517 (2002).
Acknowledgements
This work was supported, in part, by NIH grants R01-HL73043 and R01-HL90867 (K. T. Weber); R01-HL77668 and R01-HL96503 (Y. Sun). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH.
Author information
Authors and Affiliations
Contributions
The authors are part of a multidisciplinary investigative team. K. T. Weber and S. K. Bhattacharya wrote the manuscript. All authors researched data for the article, substantially contributed to discussion of content, and reviewed/edited the manuscript before submission.
Corresponding author
Rights and permissions
About this article
Cite this article
Weber, K., Sun, Y., Bhattacharya, S. et al. Myofibroblast-mediated mechanisms of pathological remodelling of the heart. Nat Rev Cardiol 10, 15–26 (2013). https://doi.org/10.1038/nrcardio.2012.158
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2012.158
This article is cited by
-
Morroniside improves AngII-induced cardiac fibroblast proliferation, migration, and extracellular matrix deposition by blocking p38/JNK signaling pathway through the downregulation of KLF5
Naunyn-Schmiedeberg's Archives of Pharmacology (2024)
-
COVID-19 HEART unveiling as atrial fibrillation: pathophysiology, management and future directions for research
The Egyptian Heart Journal (2023)
-
IRX2 regulates angiotensin II-induced cardiac fibrosis by transcriptionally activating EGR1 in male mice
Nature Communications (2023)
-
Targeting necroptosis in fibrosis
Molecular Biology Reports (2023)
-
DLK1 overexpression improves sepsis-induced cardiac dysfunction and fibrosis in mice through the TGF-β1/Smad3 signaling pathway and MMPs
Journal of Molecular Histology (2023)