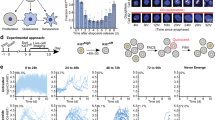
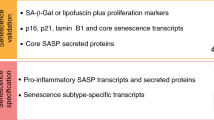
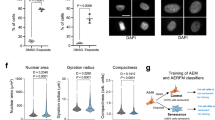
Abstract
Normal cells can permanently lose the ability to proliferate when challenged by potentially oncogenic stress, a process termed cellular senescence. Senescence-associated beta-galactosidase (SA-βgal) activity, detectable at pH 6.0, permits the identification of senescent cells in culture and mammalian tissues. Here we describe first a cytochemical protocol suitable for the histochemical detection of individual senescent cells both in culture and tissue biopsies. The second method is based on the alkalinization of lysosomes, followed by the use of 5-dodecanoylaminofluorescein di-β-D-galactopyranoside (C12FDG), a fluorogenic substrate for βgal activity. The cytochemical method takes about 30 min to execute, and several hours to a day to develop and score. The fluorescence methods take between 4 and 8 h to execute and can be scored in a single day. The cytochemical method is applicable to tissue sections and requires simple reagents and equipment. The fluorescence-based methods have the advantages of being more quantitative and sensitive.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Shay, J.W. & Wright, W.E. Hayflick, his limit, and cellular ageing. Nat. Rev. Mol. Cell Biol. 1, 72–76 (2000).
Campisi, J. & d'Adda di Fagagna, F. Cellular senescence: when bad things happen to good cells. Nat. Rev. Mol. Cell Biol. 8, 729–740 (2007).
Chretien, A. et al. Role of TGF-beta1-independent changes in protein neosynthesis, p38alphaMAPK, and cdc42 in hydrogen peroxide-induced senescence-like morphogenesis. Free Radic. Biol. Med. 44, 1732–1751 (2008).
Collado, M., Blasco, M.A. & Serrano, M. Cellular senescence in cancer and aging. Cell 130, 223–233 (2007).
Price, J.S. et al. The role of chondrocyte senescence in osteoarthritis. Aging Cell 1, 57–65 (2002).
Campisi, J. Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell 120, 513–522 (2005).
Erusalimsky, J.D. & Kurz, D.J. Cellular senescence in vivo: its relevance in ageing and cardiovascular disease. Exp. Gerontol. 40, 634–642 (2005).
Matthews, C. et al. Vascular smooth muscle cells undergo telomere-based senescence in human atherosclerosis: effects of telomerase and oxidative stress. Circ. Res. 99, 156–164 (2006).
Dimri, G.P. et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo . Proc. Natl. Acad. Sci. USA 92, 9363–9367 (1995).
Castro, P., Giri, D., Lamb, D. & Ittmann, M. Cellular senescence in the pathogenesis of benign prostatic hyperplasia. Prostate 55, 30–38 (2003).
Paradis, V. et al. Replicative senescence in normal liver, chronic hepatitis C, and hepatocellular carcinomas. Hum. Pathol. 32, 327–332 (2001).
van der Loo, B., Fenton, M.J. & Erusalimsky, J.D. Cytochemical detection of a senescence-associated beta-galactosidase in endothelial and smooth muscle cells from human and rabbit blood vessels. Exp. Cell Res. 241, 309–315 (1998).
Kurz, D.J., Decary, S., Hong, Y. & Erusalimsky, J.D. Senescence-associated (beta)-galactosidase reflects an increase in lysosomal mass during replicative ageing of human endothelial cells. J. Cell Sci. 113 (Pt 20): 3613–3622 (2000).
Brunk, U., Ericsson, J.L., Ponten, J. & Westermark, B. Residual bodies and 'aging' in cultured human glia cells. Effect of entrance into phase 3 and prolonged periods of confluence. Exp. Cell Res. 79, 1–14 (1973).
Cristofalo, V.J. et al. Replicative senescence: a critical review. Mech. Ageing Dev. 125, 827–848 (2004).
Robbins, E., Levine, E.M. & Eagle, H. Morphologic changes accompanying senescence of cultured human diploid cells. J. Exp. Med. 131, 1211–1222 (1970).
Yoshimori, T. et al. Bafilomycin A1, a specific inhibitor of vacuolar-type H(+)-ATPase, inhibits acidification and protein degradation in lysosomes of cultured cells. J. Biol. Chem. 266, 17707–17712 (1991).
Lee, B.Y. et al. Senescence-associated beta-galactosidase is lysosomal beta-galactosidase. Aging Cell 5, 187–195 (2006).
Yoshida, K. et al. Human beta-galactosidase gene mutations in GM1-gangliosidosis: a common mutation among Japanese adult/chronic cases. Am. J. Hum. Genet. 49, 435–442 (1991).
Suzuki, Y. & Oshima, A. A beta-galactosidase gene mutation identified in both Morquio B disease and infantile GM1 gangliosidosis. Hum. Genet. 91, 407 (1993).
Debacq-Chainiaux, F. et al. Screening of senescence-associated genes with specific DNA array reveals the role of IGFBP-3 in premature senescence of human diploid fibroblasts. Free Radic. Biol. Med. 44, 1817–1832 (2008).
Debacq-Chainiaux, F. et al. Repeated exposure of human skin fibroblasts to UVB at subcytotoxic level triggers premature senescence through the TGF-beta1 signaling pathway. J. Cell Sci. 118, 743–758 (2005).
Dumont, P. et al. Induction of replicative senescence biomarkers by sublethal oxidative stresses in normal human fibroblast. Free Radic. Biol. Med. 28, 361–373 (2000).
Frippiat, C. et al. Subcytotoxic H2O2 stress triggers a release of transforming growth factor-beta 1, which induces biomarkers of cellular senescence of human diploid fibroblasts. J. Biol. Chem. 276, 2531–2537 (2001).
Bertrand-Vallery, V. et al. Repeated exposures to UVB induce differentiation rather than senescence of human keratinocytes lacking p16(INK-4A). Biogerontology epub ahead of print, 10.1007/s10522-009-9238-y (25 June 2009).
Lewis, D.A., Yi, Q., Travers, J.B. & Spandau, D.F. UVB-induced senescence in human keratinocytes requires a functional insulin-like growth factor-1 receptor and p53. Mol. Biol. Cell 19, 1346–1353 (2008).
Masters, J.R. & Stacey, G.N. Changing medium and passaging cell lines. Nat. Protoc. 2, 2276–2284 (2007).
Chen, Q. & Ames, B.N. Senescence-like growth arrest induced by hydrogen peroxide in human diploid fibroblast F65 cells. Proc. Natl. Acad. Sci. USA 91, 4130–4134 (1994).
Dumont, P. et al. Growth kinetics rather than stress accelerate telomere shortening in cultures of human diploid fibroblasts in oxidative stress-induced premature senescence. FEBS Lett. 502, 109–112 (2001).
Toussaint, O. et al. Stress-induced premature senescence or stress-induced senescence-like phenotype: one in vivo reality, two possible definitions? Scientific World Journal 2, 230–247 (2002).
von Zglinicki, T. Oxidative stress shortens telomeres. Trends Biochem. Sci. 27, 339–344 (2002).
Tsolou, A. et al. ssDNA fragments induce cell senescence by telomere uncapping. Exp. Gerontol. 43, 892–899 (2008).
Coppe, J.P. et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 6, 2853–2868 (2008).
Rodier, F. et al. Persistent DNA damage signaling triggers senescence-associated inflammatory cytokine secretion. Nat. Cell Biol. 11, 973–979 (2009).
Patschan, S. et al. Mapping mechanisms and charting the time course of premature cell senescence and apoptosis: lysosomal dysfunction and ganglioside accumulation in endothelial cells. Am. J. Physiol. Renal Physiol. 294, F100–F109 (2008).
Acknowledgements
O.T. and F.D.-C. are respectively Senior Research Associate and Research Associate of the FNRS, Belgium. We acknowledge the Région Wallonne for the First-DEI 'Cosmet-X' project, the Network II Senegene project and the Pole of Excellence Nanotoxico project. We thank the European Commission for the Integrated Projects Proteomage (LSHM-CT-2005-518230) and GeHA (LSHM-CT-2004-503270); for the Coordination & Support Actions Link-Age and WhyWeAge (LSHM-CT-2005-513866, HEALTH-F4-2008-200970); and the Marie Curie Matiss Project (MTKI-CT-2006–042768). Also supported by grants from the US National Institutes of Health (AG09909, AG017242, CA0126540, AG032117 and AG025708) and the British Heart Foundation.
Author information
Authors and Affiliations
Contributions
F.D.-C., O.T., J.D.E. and J.C. carried out and analyzed experiments. F.D.-C., O.T., J.D.E. and J.C. wrote the paper.
Corresponding author
Rights and permissions
About this article
Cite this article
Debacq-Chainiaux, F., Erusalimsky, J., Campisi, J. et al. Protocols to detect senescence-associated beta-galactosidase (SA-βgal) activity, a biomarker of senescent cells in culture and in vivo. Nat Protoc 4, 1798–1806 (2009). https://doi.org/10.1038/nprot.2009.191
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2009.191
This article is cited by
-
Morphology-based deep learning enables accurate detection of senescence in mesenchymal stem cell cultures
BMC Biology (2024)
-
The synergism of cytosolic acidosis and reduced NAD+/NADH ratio is responsible for lactic acidosis-induced vascular smooth muscle cell impairment in sepsis
Journal of Biomedical Science (2024)
-
Cholesterol biosynthetic pathway induces cellular senescence through ERRα
npj Aging (2024)
-
Proteomics of mouse brain endothelium uncovers dysregulation of vesicular transport pathways during aging
Nature Aging (2024)
-
Palmitic acid in type 2 diabetes mellitus promotes atherosclerotic plaque vulnerability via macrophage Dll4 signaling
Nature Communications (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.