Abstract
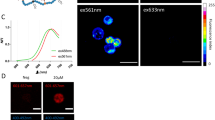
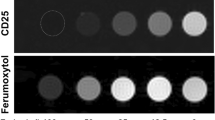
Observation of immune and stem cells in their native microenvironments requires the development of imaging agents to allow their in vivo tracking. We describe here the synthesis of magnetofluorescent nanoparticles for cell labeling in vitro and for multimodality imaging of administered cells in vivo. MION-47, a prototype monocrystalline iron oxide nanoparticle, was first converted to an intermediate bearing a fluorochrome and amine groups, then reacted with either HIV-Tat peptide or protamine to yield a nanoparticle with membrane-translocating properties. We describe how to assess optimal cell labeling with tests of cell phenotype and function. Synthesis of magnetofluorescent nanoparticles and cell-labeling optimization can be realized in 48 h, whereas nanoparticle uptakes and retention studies may generally take up to 120 h. Labeled cells can be detected by magnetic resonance imaging, fluorescence reflectance imaging, fluorescence-mediated tomography, confocal microscopy and flow cytometry, and can be purified based on their fluorescent or magnetic properties. The present protocol focuses on T-cell labeling but can be used for labeling a variety of circulating cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Lewin, M. et al. Tat peptide-derivatized magnetic nanoparticles allow in vivo tracking and recovery of progenitor cells. Nat. Biotechnol. 18, 410–414 (2000).
Kircher, M.F. et al. In vivo high resolution three-dimensional imaging of antigen-specific cytotoxic T-lymphocyte trafficking to tumors. Cancer Res. 63, 6838–6846 (2003).
Ntziachristos, V., Ripoll, J., Wang, L.V. & Weissleder, R. Looking and listening to light: the evolution of whole-body photonic imaging. Nat. Biotechnol. 23, 313–320 (2005).
Weissleder, R. Scaling down imaging: molecular mapping of cancer in mice. Nat. Rev. Cancer 2, 11–18 (2002).
Dudley, M.E. & Rosenberg, S.A. Adoptive-cell-transfer therapy for the treatment of patients with cancer. Nat. Rev. Cancer 3, 666–675 (2003).
Dudley, M.E. et al. Cancer regression and autoimmunity in patients after clonal repopulation with antitumor lymphocytes. Science 298, 850–854 (2002).
Yee, C., Riddell, S.R. & Greenberg, P.D. In vivo tracking of tumor-specific T cells. Curr. Opin. Immunol. 13, 141–146 (2001).
de Vries, I.J. et al. Magnetic resonance tracking of dendritic cells in melanoma patients for monitoring of cellular therapy. Nat. Biotechnol. 23, 1407–1413 (2005).
Frangioni, J.V. & Hajjar, R.J. In vivo tracking of stem cells for clinical trials in cardiovascular disease. Circulation 110, 3378–3383 (2004).
Vianello, F. et al. Murine B16 melanomas expressing high levels of the chemokine stromal-derived factor-1/CXCL12 induce tumor-specific T cell chemorepulsion and escape from immune control. J. Immunol. 176, 2902–2914 (2006).
Reynolds, F., Weissleder, R. & Josephson, L. Protamine as an efficient membrane-translocating peptide. Bioconjug. Chem. 16, 1240–1245 (2005).
Fawell, S. et al. Tat-mediated delivery of heterologous proteins into cells. Proc. Natl. Acad. Sci. USA 91, 664–668 (1994).
Nagahara, H. et al. Transduction of full-length TAT fusion proteins into mammalian cells: TAT-p27Kip1 induces cell migration. Nat. Med. 4, 1449–1452 (1998).
Schwarze, S.R., Ho, A., Vocero-Akbani, A. & Dowdy, S.F. In vivo protein transduction: delivery of a biologically active protein into the mouse. Science 285, 1569–1572 (1999).
Weissleder, R. & Ntziachristos, V. Shedding light onto live molecular targets. Nat. Med. 9, 123–128 (2003).
Weissleder, R., Kelly, K., Sun, E.Y., Shtatland, T. & Josephson, L. Cell-specific targeting of nanoparticles by multivalent attachment of small molecules. Nat. Biotechnol. 23, 1418–1423 (2005).
Arbab, A.S. et al. Efficient magnetic cell labeling with protamine sulfate complexed to ferumoxides for cellular MRI. Blood 104, 1217–1223 (2004).
Montet-Abou, K., Montet, X., Weissleder, R. & Josephson, L. Transfection agent induced nanoparticle cell loading. Mol. Imaging 4, 165–171 (2005).
Chen, M.L. et al. Regulatory T cells suppress tumor-specific CD8 T cell cytotoxicity through TGF-β signals in vivo. Proc. Natl. Acad. Sci. USA 102, 419–424 (2005).
Pittet, M.J. et al. Expansion and functional maturation of human tumor antigen-specific CD8+ T cells after vaccination with antigenic peptide. Clin. Cancer Res. 7, 796s–803s (2001).
Pittet, M.J. et al. Ex vivo characterization of allo-MHC-restricted T cells specific for a single MHC-peptide complex. J. Immunol. 176, 2330–2336 (2006).
Pittet, M.J. et al. Ex vivo IFN-γ secretion by circulating CD8 T lymphocytes: implications of a novel approach for T cell monitoring in infectious and malignant diseases. J. Immunol. 166, 7634–7640 (2001).
Kircher, M.F. et al. In vivo high resolution three-dimensional imaging of antigen-specific cytotoxic T-lymphocyte trafficking to tumors. Cancer Res. 63, 6838–6846 (2003).
Koehne, G. et al. Serial in vivo imaging of the targeted migration of human HSV-TK-transduced antigen-specific lymphocytes. Nat. Biotechnol. 21, 405–413 (2003).
Shachaf, C.M. et al. MYC inactivation uncovers pluripotent differentiation and tumour dormancy in hepatocellular cancer. Nature 431, 1112–1117 (2004).
Thorne, S.H., Negrin, R.S. & Contag, C.H. Synergistic antitumor effects of immune cell-viral biotherapy. Science 311, 1780–1784 (2006).
Shu, C.J. et al. Visualization of a primary anti-tumor immune response by positron emission tomography. Proc. Natl. Acad. Sci. USA 102, 17412–17417 (2005).
Murakami, T. et al. Immune evasion by murine melanoma mediated through CC chemokine receptor-10. J. Exp. Med. 198, 1337–1347 (2003).
Mempel, T.R., Henrickson, S.E. & Von Andrian, U.H. T-cell priming by dendritic cells in lymph nodes occurs in three distinct phases. Nature 427, 154–159 (2004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
R.W. is a founder of VisEn Medical.
Rights and permissions
About this article
Cite this article
Pittet, M., Swirski, F., Reynolds, F. et al. Labeling of immune cells for in vivo imaging using magnetofluorescent nanoparticles. Nat Protoc 1, 73–79 (2006). https://doi.org/10.1038/nprot.2006.11
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2006.11
This article is cited by
-
Covalent assembly of nanoparticles as a peptidase-degradable platform for molecular MRI
Nature Communications (2017)
-
Biomarkers in preclinical cancer imaging
European Journal of Nuclear Medicine and Molecular Imaging (2015)
-
Highly enhanced optical properties of indocyanine green/perfluorocarbon nanoemulsions for efficient lymph node mapping using near-infrared and magnetic resonance imaging
Nano Convergence (2014)
-
Imaging macrophages with nanoparticles
Nature Materials (2014)
-
In situ simultaneous monitoring of ATP and GTP using a graphene oxide nanosheet–based sensing platform in living cells
Nature Protocols (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.