Abstract
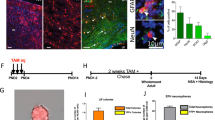
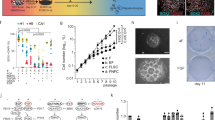
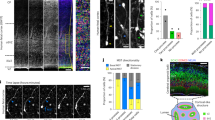
The olfactory neuroepithelium undergoes continual neurogenesis and, after extensive lesions, fully regenerates to maintain sensory function. The stem cell population underlying this regenerative capacity remains elusive. Here we show that mouse horizontal basal cells (HBCs) function as adult olfactory neuroepithelium neural stem cells and examine their distinct dynamics in olfactory neuroepithelium maintenance and regeneration. Fate-mapping analysis after olfactory neuroepithelium lesioning shows that HBCs are competent to regenerate both neuronal and non-neuronal olfactory neuroepithelium lineages. HBCs serve as a reservoir of long-lived progenitors that remain largely quiescent during normal neuronal turnover or even after acute, selective loss of mature neurons. Under these conditions, previously identified progenitors are largely responsible for tissue maintenance. Yet after extensive injuries that deplete resident neuronal precursors, HBCs transiently proliferate and their progeny fully reconstitute the neuroepithelium. Our data support a new model of adult neurogenesis in which distinct cell populations mediate normal neuronal turnover and neuronal replacement upon traumatic injury.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Gage, F.H. Mammalian neural stem cells. Science 287, 1433–1438 (2000).
Lie, D.C., Song, H., Colamarino, S.A., Ming, G.L. & Gage, F.H. Neurogenesis in the adult brain: new strategies for central nervous system diseases. Annu. Rev. Pharmacol. Toxicol. 44, 399–421 (2004).
Mitchell, B.D., Emsley, J.G., Magavi, S.S., Arlotta, P. & Macklis, J.D. Constitutive and induced neurogenesis in the adult mammalian brain: manipulation of endogenous precursors toward CNS repair. Dev. Neurosci. 26, 101–117 (2004).
Alvarez-Buylla, A., Garcia-Verdugo, J.M. & Tramontin, A.D. A unified hypothesis on the lineage of neural stem cells. Nat. Rev. Neurosci. 2, 287–293 (2001).
Schwob, J.E. Neural regeneration and the peripheral olfactory system. Anat. Rec. 269, 33–49 (2002).
Mackay-Sim, A. & Kittel, P. Cell dynamics in the adult mouse olfactory epithelium: a quantitative autoradiographic study. J. Neurosci. 11, 979–984 (1991).
Huard, J.M., Youngentob, S.L., Goldstein, B.J., Luskin, M.B. & Schwob, J.E. Adult olfactory epithelium contains multipotent progenitors that give rise to neurons and non-neural cells. J. Comp. Neurol. 400, 469–486 (1998).
Jang, W., Youngentob, S.L. & Schwob, J.E. Globose basal cells are required for reconstitution of olfactory epithelium after methyl bromide lesion. J. Comp. Neurol. 460, 123–140 (2003).
Carter, L.A., MacDonald, J.L. & Roskams, A.J. Olfactory horizontal basal cells demonstrate a conserved multipotent progenitor phenotype. J. Neurosci. 24, 5670–5683 (2004).
Caggiano, M., Kauer, J.S. & Hunter, D.D. Globose basal cells are neuronal progenitors in the olfactory epithelium: a lineage analysis using a replication-incompetent retrovirus. Neuron 13, 339–352 (1994).
Calof, A.L. et al. Progenitor cells of the olfactory receptor neuron lineage. Microsc. Res. Tech. 58, 176–188 (2002).
Chen, X., Fang, H. & Schwob, J.E. Multipotency of purified, transplanted globose basal cells in olfactory epithelium. J. Comp. Neurol. 469, 457–474 (2004).
Cau, E., Casarosa, S. & Guillemot, F. Mash1 and Ngn1 control distinct steps of determination and differentiation in the olfactory sensory neuron lineage. Development 129, 1871–1880 (2002).
Gordon, M.K., Mumm, J.S., Davis, R.A., Holcomb, J.D. & Calof, A.L. Dynamics of MASH1 expression in vitro and in vivo suggest a non–stem cell site of MASH1 action in the olfactory receptor neuron lineage. Mol. Cell. Neurosci. 6, 363–379 (1995).
Holbrook, E.H., Szumowski, K.E. & Schwob, J.E. An immunochemical, ultrastructural and developmental characterization of the horizontal basal cells of rat olfactory epithelium. J. Comp. Neurol. 363, 129–146 (1995).
Sicard, G., Feron, F., Andrieu, J.L., Holley, A. & Mackay-Sim, A. Generation of neurons from a nonneuronal precursor in adult olfactory epithelium in vitro. Ann. NY Acad. Sci. 855, 223–225 (1998).
Doetsch, F., Caille, I., Lim, D.A., Garcia-Verdugo, J.M. & Alvarez-Buylla, A. Subventricular zone astrocytes are neural stem cells in the adult mammalian brain. Cell 97, 703–716 (1999).
Palmer, T.D., Takahashi, J. & Gage, F.H. The adult rat hippocampus contains primordial neural stem cells. Mol. Cell. Neurosci. 8, 389–404 (1997).
Indra, A.K. et al. Temporally controlled site-specific mutagenesis in the basal layer of the epidermis: comparison of the recombinase activity of the tamoxifen-inducible Cre-ER(T) and Cre-ER(T2) recombinases. Nucleic Acids Res. 27, 4324–4327 (1999).
Ahn, S. & Joyner, A.L. In vivo analysis of quiescent adult neural stem cells responding to Sonic hedgehog. Nature 437, 894–897 (2005).
Bleul, C.C. et al. Formation of a functional thymus initiated by a postnatal epithelial progenitor cell. Nature 441, 992–996 (2006).
Kawauchi, S. et al. Molecular signals regulating proliferation of stem and progenitor cells in mouse olfactory epithelium. Dev. Neurosci. 26, 166–180 (2004).
Wu, H.H. et al. Autoregulation of neurogenesis by GDF11. Neuron 37, 197–207 (2003).
Robinson, S.P., Langan-Fahey, S.M., Johnson, D.A. & Jordan, V.C. Metabolites, pharmacodynamics and pharmacokinetics of tamoxifen in rats and mice compared to the breast cancer patient. Drug Metab. Dispos. 19, 36–43 (1991).
Schwob, J.E., Youngentob, S.L. & Mezza, R.C. Reconstitution of the rat olfactory epithelium after methyl bromide–induced lesion. J. Comp. Neurol. 359, 15–37 (1995).
Miyawaki, A., Homma, H., Tamura, H., Matsui, M. & Mikoshiba, K. Zonal distribution of sulfotransferase for phenol in olfactory sustentacular cells. EMBO J. 15, 2050–2055 (1996).
Schwartz Levey, M., Chikaraishi, D.M. & Kauer, J.S. Characterization of potential precursor populations in the mouse olfactory epithelium using immunocytochemistry and autoradiography. J. Neurosci. 11, 3556–3564 (1991).
Beites, C.L., Kawauchi, S., Crocker, C.E. & Calof, A.L. Identification and molecular regulation of neural stem cells in the olfactory epithelium. Exp. Cell Res. 306, 309–316 (2005).
Manglapus, G.L., Youngentob, S.L. & Schwob, J.E. Expression patterns of basic helix-loop-helix transcription factors define subsets of olfactory progenitor cells. J. Comp. Neurol. 479, 216–233 (2004).
Bergman, U., Ostergren, A., Gustafson, A.L. & Brittebo, B. Differential effects of olfactory toxicants on olfactory regeneration. Arch. Toxicol. 76, 104–112 (2002).
Carr, V.M. & Farbman, A.I. Ablation of the olfactory bulb upregulates the rate of neurogenesis and induces precocious cell death in olfactory epithelium. Exp. Neurol. 115, 55–59 (1992).
Schwob, J.E., Huard, J.M., Luskin, M.B. & Youngentob, S.L. Retroviral lineage studies of the rat olfactory epithelium. Chem. Senses 19, 671–682 (1994).
Berton, T.R. et al. Tumor formation in mice with conditional inactivation of Brca1 in epithelial tissues. Oncogene 22, 5415–5426 (2003).
Merkle, F.T., Tramontin, A.D., Garcia-Verdugo, J.M. & Alvarez-Buylla, A. Radial glia give rise to adult neural stem cells in the subventricular zone. Proc. Natl. Acad. Sci. USA 101, 17528–17532 (2004).
Taylor, G., Lehrer, M.S., Jensen, P.J., Sun, T.T. & Lavker, R.M. Involvement of follicular stem cells in forming not only the follicle but also the epidermis. Cell 102, 451–461 (2000).
Ito, M. et al. Stem cells in the hair follicle bulge contribute to wound repair but not to homeostasis of the epidermis. Nat. Med. 11, 1351–1354 (2005).
Seri, B., Garcia-Verdugo, J.M., McEwen, B.S. & Alvarez-Buylla, A. Astrocytes give rise to new neurons in the adult mammalian hippocampus. J. Neurosci. 21, 7153–7160 (2001).
Lewcock, J.W. & Reed, R.R. A feedback mechanism regulates monoallelic odorant receptor expression. Proc. Natl. Acad. Sci. USA 101, 1069–1074 (2004).
Acknowledgements
We appreciate the helpful suggestions of J. Nathans, P. Chambon for providing the K5-CreERT2 mice, and D. Johnson for providing the K5-Cre mice. This work was supported by the Howard Hughes Medical Institute and grants from the US National Institutes of Health (R.R.R.).
Author information
Authors and Affiliations
Contributions
C.T.L. co-designed and conducted all of the experiments and co-wrote the manuscript. P.A.C. provided unpublished mouse strains and guided experimental design. R.R.R. supervised the project, designed experimental protocols and was responsible for writing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Rare incidences of LacZ+ non-HBC cells/clusters under normal condition. (PDF 665 kb)
Supplementary Fig. 2
Labeled HBCs express ICAM. (PDF 240 kb)
Supplementary Fig. 3
Proliferating basal cells. (PDF 395 kb)
Supplementary Fig. 4
Keratin 5 expression during postnatal OE development. (PDF 574 kb)
Rights and permissions
About this article
Cite this article
Leung, C., Coulombe, P. & Reed, R. Contribution of olfactory neural stem cells to tissue maintenance and regeneration. Nat Neurosci 10, 720–726 (2007). https://doi.org/10.1038/nn1882
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1882
This article is cited by
-
Type 2 and Non-type 2 Inflammation in the Upper Airways: Cellular and Molecular Alterations in Olfactory Neuroepithelium Cell Populations
Current Allergy and Asthma Reports (2024)
-
Basal stem cell progeny establish their apical surface in a junctional niche during turnover of an adult barrier epithelium
Nature Cell Biology (2023)
-
The potential therapeutic effect of platelet-rich plasma in the treatment of post-COVID-19 parosmia
The Egyptian Journal of Otolaryngology (2022)
-
Immature olfactory sensory neurons provide behaviourally relevant sensory input to the olfactory bulb
Nature Communications (2022)
-
A novel lineage of osteoprogenitor cells with dual epithelial and mesenchymal properties govern maxillofacial bone homeostasis and regeneration after MSFL
Cell Research (2022)