Abstract
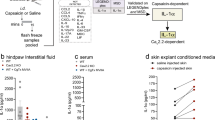
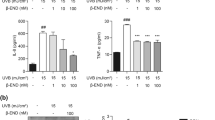
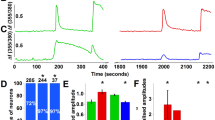
Endothelin-1 (ET-1) is a newly described pain mediator that is involved in the pathogenesis of pain states ranging from trauma to cancer. ET-1 is synthesized by keratinocytes in normal skin and is locally released after cutaneous injury. While it is able to trigger pain through its actions on endothelin-A (ETA) receptors of local nociceptors, it can coincidentally produce analgesia through endothelin-B (ETB) receptors. Here we map a new endogenous analgesic circuit, in which ETB receptor activation induces the release of β-endorphin from keratinocytes and the activation of G-protein-coupled inwardly rectifying potassium channels (GIRKs, also named Kir-3) linked to opioid receptors on nociceptors. These results indicate the existence of an intrinsic feedback mechanism to control peripheral pain in skin, and establish keratinocytes as an ETB receptor–operated opioid pool.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
McQuay, H. & Moore, A. Introduction. in An Evidence-Based Resource For Pain Relief. (eds. McQuay, H & Moore, A.) 23 (Oxford University Press, Oxford, 1998).
Scholz, J. & Woolf, C.J. Can we conquer pain? Nat. Neurosci. 5 (suppl. 1), 1062– 1067 (2002).
Julius, D. & Basbaum, A.I. Molecular mechanisms of nociception. Nature 413, 203– 210 (2001).
Hickey, K.A., Rubanyi, G.M., Paul, R.J. & Highsmith, R.F. Characterization of a coronary vasoconstrictor produced by cultured endothelial cells. Am. J. Physiol. 248, C550– C556 (1985).
Yanagisawa, M. et al. A novel potent vasoconstrictor peptide produced by vascular endothelial cells. Nature 332, 411– 415 (1988).
Rubanyi, G.M. & Polokoff, A. Endothelins: molecular biology, biochemistry, pharmacology, physiology, and pathophysiology. Pharmacol. Rev. 48, 325– 415 (1994).
Ahn, G.Y. et al. The expression of endothelin-1 and its binding sites in mouse skin increased after ultraviolet B irradiation or local injection of tumor necrosis factor alpha. J. Dermatol. 25, 78– 84 (1998).
Luscher, T.F. & Barton, M. Endothelins and endothelin receptor antagonists: therapeutic considerations for a novel class of cardiovascular drugs. Circulation 102, 2434– 2440 (2000).
Nelson, J. et al. Identification of endothelin-1 in the pathophysiology of metastatic adenocarcinoma of the prostate. Nat. Med. 1, 944– 949 (1995).
Ferreira, S.H., Romitelli, M. & de Nucci, G. Endothelin-1 participation in overt and inflammatory pain. J. Cardiovasc. Pharmacol. 13 (suppl. 5), S220– S222 (1989).
Davar, G., Hans, G., Fareed, M.U., Sinnott, C. & Strichartz, G. Behavioral signs of acute pain produced by application of endothelin-1 to rat sciatic nerve. NeuroReport 9, 2279– 2283 (1998).
Piovezan, A.P., D'Orleans-Juste, P., Souza, G.E. & Rae, G.A. Endothelin-1-induced ET(A) receptor-mediated nociception, hyperalgesia and oedema in the mouse hind-paw: modulation by simultaneous ET(B) receptor activation. Br. J. Pharmacol. 129, 961– 968 (2000).
Jarvis, M.F. et al. ABT-627, an endothelin ET(A) receptor-selective antagonist, attenuates tactile allodynia in a diabetic rat model of neuropathic pain. Eur. J. Pharmacol. 388, 29– 35 (2000).
Fareed, M.U., Hans, G., Atanda, A., Strichartz, G. & Davar, G. Pharmacological characterization of acute pain behavior produced by application of endothelin-1 to rat sciatic nerve. J. Pain 1, 46– 53 (2000).
Gokin, A.P. et al. Local injection of endothelin-1 produces pain-like behavior and excitation of nociceptors in rats. J. Neurosci. 21, 5358– 5366 (2001).
Pomonis, J.D., Rogers, S.D., Peters, C.M., Ghilardi, J.R. & Mantyh, P.W. Expression and localization of endothelin receptors: implications for the involvement of peripheral glia in nociception. J. Neurosci. 21, 999– 1006 (2001).
De-Melo, J.D., Tonussi, C.R., D'Orleans-Juste, P. & Rae, G.A. Articular nociception induced by endothelin-1, carrageenan and LPS in naive and previously inflamed knee-joints in the rat: inhibition by endothelin receptor antagonists. Pain 77, 261– 269 (1998).
Wacnik, P.W. et al. Functional interactions between tumor and peripheral nerve: morphology, algogen identification, and behavioral characterization of a new murine model of cancer pain. J. Neurosci. 21, 9355– 9366 (2001).
Carducci, M.A. et al. Atrasentan, an endothelin-receptor antagonist for refractory adenocarcinomas: safety and pharmacokinetics. J. Clin. Oncol. 8, 2171– 2180 (2002).
Khodorova, A., Fareed, M.U., Gokin, A., Strichartz, G.R. & Davar, G. Local injection of a selective endothelin-b receptor agonist inhibits endothelin-1 induced pain-like behavior and excitation of nociceptors in a naloxone-sensitive manner. J. Neurosci. 22, 7788– 7796 (2002).
Fields, H.L., Emson, P.C., Leigh, B.K., Gilbert, R.F. & Iversen, L.L. Multiple opiate receptor sites on primary afferent fibers. Nature 284, 351– 353 (1980).
Stein, C. Peripheral mechanisms of opioid analgesia. Anesth. Analg. 76, 182– 191 (1993).
Machelska, H., Cabot, P.J., Mousa, S.A., Zhang, Q. & Stein, C. Pain control in inflammation governed by selectins. Nat. Med. 4, 1425– 1428 (1998).
Akil, H., Mayer, D.J. & Liebeskind, J.C. Comparison in the rat between analgesia induced by stimulation of periacqueducal gray matter and morphine analgesia. C. R. Acad. Sci. Hebd. Seances Acad. Sci. D 274, 3603– 3605 (1972).
Hughes, J. et al. Identification of two related pentapeptides from the brain with potent opiate agonist activity. Nature 258, 577– 580 (1975).
Akil, H., Mayer, D.J. & Liebeskind, J.C. Antagonism of stimulation-produced analgesia by naloxone, a narcotic antagonist. Science 191, 961– 962 (1976).
Fields, H.L., Heinricher, M.M. & Mason, P. Neurotransmitters in nociceptive modulatory circuits. Ann. Rev. Neurosci. 14, 219– 245 (1991).
Harris, J.A. Descending antinociceptive mechanisms in the brainstem: their role in the animal's defensive system. J. Physiol. (Paris) 90, 15– 25 (1996).
Gutstein, H.B., Mansour, A., Watson, S.J., Akil, H. & Fields, H.L. Mu and κ-opioid receptors in periaqueductal gray and rostral ventromedial medulla. NeuroReport 9, 1777– 1781 (1998).
Stein, C. The control of pain in peripheral tissue by opioids. N. Engl. J. Med. 332, 168– 590 (1995).
Calignano, A., La Rana, G., Giuffrida, A. & Piomelli, D. Control of pain initiation by endogenous cannabinoids. Nature 394, 277– 281 (1998).
Wettstein, G. & Grouhel, A. Opioid antagonist profile of SC nor-binaltorphimine in the formalin paw assay. Pharmacol. Biochem. Behav. 53, 411– 416 (1996).
Wintzen, M., Yaar, M., Burbach, J.P. & Gilchrest, B.A. Proopiomelanocortin gene product regulation in keratinocytes. J. Invest. Dermatol. 106, 673– 678 (1996).
Zanello, S.B, Jackson, D.M. & Holick, M.F. An immunocytochemical approach to the study of beta-endorphin production in human keratinocytes using confocal microscopy. Ann. NY Acad. Sci. 885, 85– 99 (1999).
Hargreaves, K.M., Flores, C.M., Dionne, R.A. & Mueller, G.P. The role of pituitary beta-endorphin in mediating corticotropin-releasing factor-induced antinociception. Am. J. Physiol. 258, E235– E242 (1990).
Mark, M.D. & Herlitze, S. G-protein mediated gating of inward-rectifier K+ channels. Eur. J. Biochem. 267, 5830– 5836 (2000).
Rogalski, S.L., Cyr, C. & Chavkin, C. Activation of the endothelin receptor inhibits the G protein-coupled inwardly rectifying potassium channel by a phospholipase A2-mediated mechanism. J. Neurochem. 72, 1409– 1416 (1999).
Ikeda, K., Kobayashi, T., Kumanishi, T., Niki, H. & Yano, R. Involvement of G-protein-activated inwardly rectifying K (GIRK) channels in opioid-induced analgesia. Neurosci. Res. 38, 113– 116 (2000).
Blanchet, C. & Luscher, C. Desensitization of mu-opioid receptor-evoked potassium currents: initiation at the receptor, expression at the effector. Proc. Natl. Acad. Sci. USA 99, 4674– 4679 (2002).
Torrecilla, M. et al. G-protein-gated potassium channels containing Kir3.2 and Kir3.3 subunits mediate the acute inhibitory effects of opioids on locus ceruleus neurons. J. Neurosci. 22, 432– 834 (2002).
Mitrovic, I., et al. Contribution of GIRK2-mediated postsynaptic signaling to opiate and alpha 2-adrenergic analgesia and analgesic sex differences. Proc. Natl. Acad. Sci. USA 100, 271– 276 (2003).
Blednov, Y.A., Stoffel, M., Alva, H. & Harris, R.A. A pervasive mechanism for analgesia: Activation of GIRK2 channels. Proc. Natl. Acad. Sci. USA 100, 77– 82 (2003).
Zhou, Q.L., Strichartz, G. & Davar, G. Endothelin-1 activates ET(A) receptors to increase intracellular calcium in model sensory neurons. NeuroReport 12, 3853– 3857 (2001).
Zhou, Z., Davar, G. & Strichartz, G. Endothelin-A receptor dependent enhanced activation gating of tetrodotoxin-resistant sodium currents in rat sensory neurons: a possible mechanism of ET-1 induced pain, J. Neurosci 22, 6325– 6330 (2002).
Slominski, A., Wortsman, J., Luger, T., Paus, R. & Solomon, S. Corticotropin releasing hormone and proopiomelanocortin involvement in the cutaneous response to stress. Physiol. Rev. 80, 979– 1020 (2000).
Peier, A.M. et al. A heat-sensitive TRP channel expressed in keratinocytes. Science 296, 2046– 2049 (2002).
Zimmermann, M. Ethical guidelines for investigations of experimental pain in conscious animals. Pain 16, 109– 110 (1983).
Signorini, S., Liao, Y.J., Duncan, S.A., Jan, L.Y. & Stoffel, M. Normal cerebellar development but susceptibility to seizures in mice lacking G-protein-coupled, inwardly rectifying K+ channel GIRK2. Proc. Natl. Acad. Sci. USA 94, 923– 927 (1997).
Broadbear, J.H. et al. Methocinnamox is a potent, long-lasting, and selective antagonist of morphine-mediated antinociception in the mouse: comparison with clocinnamox, β-funaltrexamine, and β-chlornaltrexamine. J. Pharmacol. Exp. Ther. 294, 933– 940 (2000).
Acknowledgements
The authors thank D.E. Clapham for discussion on GIRKs and for providing GIRK knockout mice, C. Stein, F. Porreca and J.-G. Gu for help with preparation of the manuscript, and J. Bell for graphical assistance. Supported by the United States Public Health Service (National Cancer Institute), B. Navarro was supported by Kaplan fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Khodorova, A., Navarro, B., Jouaville, L. et al. Endothelin-B receptor activation triggers an endogenous analgesic cascade at sites of peripheral injury. Nat Med 9, 1055–1061 (2003). https://doi.org/10.1038/nm885
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm885
This article is cited by
-
Sovateltide (ILR-1620) Improves Motor Function and Reduces Hyperalgesia in a Rat Model of Spinal Cord Injury
Neurocritical Care (2024)
-
Role of central endothelin-1 in hyperalgesia, anhedonia, and hypolocomotion induced by endotoxin in male rats
Experimental Brain Research (2021)
-
Mouse connective tissue mast cell proteases tryptase and carboxypeptidase A3 play protective roles in itch induced by endothelin-1
Journal of Neuroinflammation (2020)
-
Lifting the veil on the keratinocyte contribution to cutaneous nociception
Protein & Cell (2020)
-
Involvement of Endothelin Receptors in Peripheral Sensory Neuropathy Induced by Oxaliplatin in Mice
Neurotoxicity Research (2019)