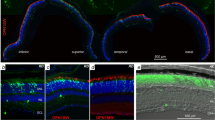
Abstract
Erythropoietin (Epo) is upregulated by hypoxia and provides protection against apoptosis of erythroid progenitors in bone marrow and brain neurons. Here we show in the adult mouse retina that acute hypoxia dose-dependently stimulates expression of Epo, fibroblast growth factor 2 and vascular endothelial growth factor via hypoxia-inducible factor-1α (HIF-1α) stabilization. Hypoxic preconditioning protects retinal morphology and function against light-induced apoptosis by interfering with caspase-1 activation, a downstream event in the intracellular death cascade. In contrast, induction of activator protein-1, an early event in the light-stressed retina, is not affected by hypoxia. The Epo receptor required for Epo signaling localizes to photoreceptor cells. The protective effect of hypoxic preconditioning is mimicked by systemically applied Epo that crosses the blood–retina barrier and prevents apoptosis even when given therapeutically after light insult. Application of Epo may, through the inhibition of apoptosis, be beneficial for the treatment of different forms of retinal disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Honig, L.S. & Rosenberg, R.N. Apoptosis and neurologic disease. Am. J. Med. 108, 317–330 (2000).
Nickells, R.W. Apoptosis of retinal ganglion cells in glaucoma: an update of the molecular pathways involved in cell death. Surv. Ophthalmol. 43, S151–161 (1999).
Adler, R., Curcio, C., Hicks, D., Price, D. & Wong, F. Cell death in age-related macular degeneration. Mol. Vis. 5, 31 (1999).
van Soest, S., Westerveld, A., de Jong, P.T., Bleeker-Wagemakers, E.M. & Bergen, A.A. Retinitis pigmentosa: Defined from a molecular point of view. Surv. Ophthalmol. 43, 321–334 (1999).
Cruickshanks, K.J., Klein, R., Klein, B.E. & Nondahl, D.M. Sunlight and the 5-year incidence of early age-related maculopathy: The Beaver Dam Eye Study. Arch. Ophthalmol. 119, 246–250 (2001).
Reme, C.E., Grimm, C., Hafezi, F., Marti, A. & Wenzel, A. Apoptotic cell death in retinal degenerations. Prog. Retin. Eye Res. 17, 443–464 (1998).
Grimm, C. et al. Protection of Rpe65-deficient mice identifies rhodopsin as a mediator of light-induced retinal degeneration. Nature Genet. 25, 63–66 (2000).
Wenzel, A., Reme, C.E., Williams, T.P., Hafezi, F. & Grimm, C. The Rpe65 Leu450Met variation increases retinal resistance against light-induced degeneration by slowing rhodopsin regeneration. J. Neurosci. 21, 53–58 (2001).
Saari, J.C. et al. Visual cycle impairment in cellular retinaldehyde binding protein (CRALBP) knockout mice results in delayed dark adaptation. Neuron 29, 739–748 (2001).
Wenzel, A. et al. Prevention of photoreceptor apoptosis by activation of the glucocorticoid receptor. Invest. Ophthalmol. Vis. Sci. 42, 1653–1659 (2001).
Grimm, C., Wenzel, A., Hafezi, F. & Reme, C.E. Gene expression in the mouse retina: The effect of damaging light. Mol. Vis. 6, 252–260 (2000).
Jelkmann, W., Erythropoietin: Structure, control of production, and function. Physiol. Rev. 72, 449–489 (1992).
Siren, A.L. et al. Erythropoietin prevents neuronal apoptosis after cerebral ischemia and metabolic stress. Proc. Natl. Acad. Sci. USA 98, 4044–4049 (2001).
Brines, M.L. et al. Erythropoietin crosses the blood–brain barrier to protect against experimental brain injury. Proc. Natl. Acad. Sci. USA 97, 10526–10531 (2000).
Zaman, K. et al. Protection from oxidative stress-induced apoptosis in cortical neuronal cultures by iron chelators is associated with enhanced DNA binding of hypoxia-inducible factor-1 and ATF-1/CREB and increased expression of glycolytic enzymes, p21(waf1/cip1), and erythropoietin. J. Neurosci. 19, 9821–9830 (1999).
Hofer, T., Wenger, H. & Gassmann, M. Oxygen sensing, HIF-1α stabilization and potential therapeutic strategies. Pflugers Arch. 443, 503–507 (2002).
Semenza, G.L. HIF-1, O(2), and the 3 phds. How animal cells signal hypoxia to the nucleus. Cell 107, 1–3 (2001).
Barbe, M.F., Tytell, M., Gower, D.J. & Welch, W.J. Hyperthermia protects against light damage in the rat retina. Science 241, 1817–1820 (1988).
Stroka, D.M. et al. HIF-1 is expressed in normoxic tissue and displays an organ-specific regulation under systemic hypoxia. FASEB J. 15, 2445–2453 (2001).
Fandrey, J. & Bunn, H.F. In vivo and in vitro regulation of erythropoietin mRNA: Measurement by competitive polymerase chain reaction. Blood 81, 617–623 (1993).
Steinberg, R.H. Monitoring communications between photoreceptors and pigment epithelial cells: Effects of “mild” systemic hypoxia. Invest. Ophthalmol. Vis. Sci. 28, 1888–1904 (1987).
Chilov, D., Hofer, T., Bauer, C., Wenger, R.H. & Gassmann, M. Hypoxia affects expression of circadian genes PER1 and CLOCK in mouse brain. FASEB J. 15, 2613–2622 (2001).
Coffer, P.J., Jin, J. & Woodgett, J.R. Protein kinase B (c-Akt): A multifunctional mediator of phosphatidylinositol 3-kinase activation. Biochem. J. 335, 1–13 (1998).
Miura, Y., Miura, O., Ihle, J.N. & Aoki, N. Activation of the mitogen-activated protein kinase pathway by the erythropoietin receptor. J. Biol. Chem. 269, 29962–29969 (1994).
Ozaki, H. et al. Hypoxia inducible factor-1α is increased in ischemic retina: Temporal and spatial correlation with VEGF expression. Invest. Ophthalmol. Vis. Sci. 40, 182–189 (1999).
Siren, A.L. & Ehrenreich, H. Erythropoietin—a novel concept for neuroprotection. Eur. Arch. Psychiatry Clin. Neurosci. 251, 179–184 (2001).
Robinson, G.S. et al. Nonvascular role for VEGF: VEGFR-1, 2 activity is critical for neural retinal development. FASEB J. 15, 1215–1217 (2001).
Faktorovich, E.G., Steinberg, R.H., Yasumura, D., Matthes, M.T. & LaVail, M.M. Photoreceptor degeneration in inherited retinal dystrophy delayed by basic fibroblast growth factor. Nature 347, 83–86 (1990).
Faktorovich, E.G., Steinberg, R.H., Yasumura, D., Matthes, M.T. & LaVail, M.M. Basic fibroblast growth factor and local injury protect photoreceptors from light damage in the rat. J. Neurosci. 12, 3554–3567 (1992).
Yamada, H. et al. Fibroblast growth factor-2 decreases hyperoxia-induced photoreceptor cell death in mice. Am. J. Pathol. 159, 1113–1120 (2001).
LaVail, M.W. et al. Protection of mouse photoreceptors by survival factors in retinal degenerations. Invest. Ophthalmol. Vis. Sci. 39, 592–602 (1998).
Spencer, B., Agarwala, S., Gentry, L. & Brandt, C.R. HSV-1 vector-delivered FGF2 to the retina is neuroprotective but does not preserve functional responses. Mol. Ther. 3, 746–756 (2001).
Wenzel, A. et al. c-fos controls the “private pathway” of light-induced apoptosis of retinal photoreceptors. J. Neurosci. 20, 81–88 (2000).
Chattopadhyay, A., Choudhury, T.D., Bandyopadhyay, D. & Datta, A.G. Protective effect of erythropoietin on the oxidative damage of erythrocyte membrane by hydroxyl radical. Biochem. Pharmacol. 59, 419–425 (2000).
Keller, C., Grimm, C., Wenzel, A., Hafezi, F. & Reme, C. Protective effect of halothane anesthesia on retinal light damage: Inhibition of metabolic rhodopsin regeneration. Invest. Ophthalmol. Vis. Sci. 42, 476–480 (2001).
Ostroy, S.E., Gaitatzes, C.G. & Friedmann, A.L. Hypoxia inhibits rhodopsin regeneration in the excised mouse eye. Invest. Ophthalmol. Vis. Sci. 34, 447–452 (1993).
Bowers, F. et al. Effects of oxygen and bFGF on the vulnerability of photoreceptors to light damage. Invest. Ophthalmol. Vis. Sci. 42, 804–815 (2001).
Lip, P.L., Blann, A.D., Hope-Ross, M., Gibson, J.M. & Lip, G.Y. Age-related macular degeneration is associated with increased vascular endothelial growth factor, hemorheology and endothelial dysfunction. Ophthalmology 108, 705–710 (2001).
Pierce, E.A. et al. Mutations in a gene encoding a new oxygen-regulated photoreceptor protein cause dominant retinitis pigmentosa. Nature Genet. 22, 248–254 (1999).
Seeliger, M.W. et al. New views on RPE65 deficiency: The rod system is the source of vision in a mouse model of Leber congenital amaurosis. Nature Genet. 29, 70–74 (2001).
Camenisch, G. et al. General applicability of chicken egg yolk antibodies: the performance of IgY immunoglobulins raised against the hypoxia-inducible factor 1α. Faseb J. 13, 81–88 (1999).
Kueng-Hitz, N. et al. The retina of c-fos−/− mice: Electrophysiologic, morphologic and biochemical aspects. Invest. Ophthalmol. Vis. Sci. 41, 909–916 (2000).
Fry, J. Biological data analysis: A practical approach (IRL Press, Oxford, 1993).
Toutenberg, H. Experimental Design and Model Choice. (Physica, Berlin, 1995).
Acknowledgements
We thank D. Greuter, G. Hoegger, C. Imsand and S. Keller for technical assistance; I. Desbaillets for discussion; F. Valeri for statistical analysis; U. Busse for administrative help; and T. Seiler and K. Landau for continuous support of our work. This study was supported by the Swiss National Science Foundation, Theodore Ott Foundation Switzerland, Hartmann-Müller Foundation Switzerland, Velux Foundation, Switzerland and the German Research Council.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Grimm, C., Wenzel, A., Groszer, M. et al. HIF-1-induced erythropoietin in the hypoxic retina protects against light-induced retinal degeneration. Nat Med 8, 718–724 (2002). https://doi.org/10.1038/nm723
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm723
This article is cited by
-
Erythropoietin regulates signaling pathways associated with neuroprotective events
Experimental Brain Research (2022)
-
Erythropoietin promotes hippocampal mitochondrial function and enhances cognition in mice
Communications Biology (2021)
-
Transcriptomic analysis of the mouse retina after acute and chronic normobaric and hypobaric hypoxia
Scientific Reports (2021)
-
Erythropoietin exposure of isolated pancreatic islets accelerates their revascularization after transplantation
Acta Diabetologica (2021)
-
Hif1a and Hif2a can be safely inactivated in cone photoreceptors
Scientific Reports (2019)