Abstract
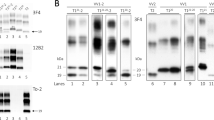
The hallmark of prion diseases is the presence of an aberrant isoform of the prion protein (PrPres) that is insoluble in nondenaturing detergents and resistant to proteases. We investigated the allelic origin of PrPres in brains of subjects heterozygous for the D178N mutation linked to fatal familial insomnia (FFI) and a subtype of Creutzfeldt-Jakob disease (CJD178), as well as for insertional mutations associated with another CJD subtype. We found that in FFI and CJD178 subjects, only mutant PrP was detergent-insoluble and protease-resistant. Therefore, PrPres derives exclusively from the mutant allele carrying the D178N mutation. In contrast, in the CJD subtype harboring insertional mutations, wild-type PrP was also detergent-insoluble and likely to be protease-resistant. Our findings indicate that the participation of the wild-type PrP in the formation of PrPres depends on the type of mutations, providing an insight into the molecular mechanisms underlying the phenotypic heterogeneity in familial prion diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
DeArmond, S.J. & Prusiner, S.B. Etiology and pathogenesis of prion disease. Am. J. Pathol. 146, 785–811 (1995).
Parchi, P. & Gambetti, P. Human prion diseases. Curr. Opin. Neural. 8, 286–293 (1995).
Goldfarb, L.G. et al. Fatal familial insomnia and familial Creutzfeldt-Jakob disease: Disease phenotype determined by a DNA polymorphism. Science 258, 806–808 (1992).
Mckinley, M.P., Bolton, D.C. & Prusiner, S.B. A protease-resistant protein is a structural component of the scrapie prion. Cell 35, 57–62 (1983).
Meyer, R.K. et al. Separation and properties of Cellular and scrapie prion proteins. Proc. Natl. Acad. Sci. USA 83, 2310–2314 (1986).
Oesch, B. et al. A Cellular gene encodes scrapie PrP 27–30 protein. Cell 40, 735–746 (1985).
Basler, K. et al. Scrapie and Cellular PrP isoforms are encoded by the same chromosomal gene. Cell 46, 417–428 (1986).
Stahl, N. et al. Structural analysis of the scrapie prion protein using mass spectrometry and amino acid sequencing. Biochemistry 32, 1991–2002 (1993).
Caughey, B.W. et al. Secondary structure analysis of the scrapie-associated protein PrP 27–30 in water by infrared spectroscopy. Biochemistry. 30, 7672–7680 (1991).
Pan, K.-M. et al. Conversion of α-helices into β-sheets features in the formation of the scrapie prion proteins. Proc Natl. Acad. Sci. USA 90, 10962–10966 (1993).
Safar, J., Roller, P.P., Gajdusek, D.C. & Gibbs, C.J. Conformational transitions, dissociation, and unfolding of scrapie amyloid (prion) protein. J. Biol. Chem. 268, 20276–20284 (1993).
Kocisko, D.A. et al. Cell-free formation of protease-resistant prion protein. Nature 370, 471–474 (1994).
Kocisko, D.A. et al. Species specificity in the Cell-free conversion of prion protein to protease-resistant forms: A model for the scrapie species barrier. Proc. Natl. Acad. Sci. USA 92, 3923–3927 (1995).
Kitamoto, T., Lizuka, R. & Tateishi, J. An amber mutation of prion protein in Gerstmann-Straussler syndrome with mutant PrP plaques. Biochem. Biophys. Res. Commun. 192, 525–531 (1993).
Tagliavini, F. et al. Amyloid fibrils in Gerstmann-Straussler-Scheinker disease (Indiana and Swedish kindreds) express only PrP peptides encoded by the mutant allele. Cell 79, 695–703 (1994).
Gabizon, R. et al. Insoluble wild-type and protease-resistant mutant prion protein in brains of patients with inherited prion diseases. Nature Med. 2, 59–64. (1996).
Bosque, P.J., Vnencak-Jones, C.L., Johnson, M.D., Whitlock, J.A. & McLean, M.J. A PrP gene codon 178 base substitution and a 24-bp interstitial deletion in familial Creutzfeldt-lakob disease. Neurology 42, 1864–1870 (1992).
Reder, A.T. et al. Clinical and genetic studies of fatal familial insomnia. Neurology 45, 1068–1075 (1995).
Palmer, M.S. et al. Deletions in the prion protein gene are not associated with CJD. Hum. Mol. Genet. 2, 541–544 (1993).
Owen, F. et al. An in-frame insertion in the prion protein gene in familial Creutzfeldt-Jakob disease. Mol. Brain Res. 7, 273–276 (1990).
Goldfarb, L.G. et al. Transmissible familial Creutzfeldt-Jakob disease associated with five, seven, and eight extra octapeptide coding repeats in the PRNP gene. Proc. Natl. Acad. Sci. USA 88, 10926–10930 (1991).
Capellari, S. et al. Familial prion disease with a novel 144-bp insertion in the prion protein gene in a Basque family. Neurology 49, 133–141 (1997).
Chen, S.G. et al. Truncated forms of the human prion protein in normal and in prion diseases. J. Biol. Chem. 270, 19173–19180 (1995).
Bolton, D.C., Bendheim, P.E., Marmorstein, A.D. & Potempska, A. Isolation and structure studies of the intact scrapie agent protein. Arch. Biochem. Biophys. 258, 579–590 (1987).
Monari, L. et al. Fatal familial insomnia and familial Creutzfeldt-Jakob disease: Different prion proteins determined by a DNA polymorphism. Proc. Natl. Acad. Sci. USA, 91, 2839–2842 (1994).
Parchi, P. et al. Molecular basis of phenotypic variability in sporadic Creutzfeldt-Jakob disease. Ann. Neural 39, 767–778 (1996).
Parchi, P. et al. Typing prion isoforms. Nature 386, 232–233 (1997).
Weissmann, C. Molecular biology of transmissible spongiform encephalopathies. FEBS Let. 389, 3–11 (1996).
Parchi, P. et al. Regional distribution of protease-resistant prion protein in fatal familial insomnia. Ann. Neural. 38, 21–29 (1995).
Gambetti, P., Parchi, P., Petersen, R.B., Chen, S.G. & Lugaresi, E. Fatal familial insomnia and familial Creutzfeldt-Jakob disease: Clinical, pathological and molecular features. Brain Pathol. 5, 43–51 (1995).
Petersen, R.B., Parchi, P., Richardson, S.L., Urig, C.B. & Gambetti, P. Effect of the D178N mutation and the codon 129 polymorphism on the metabolism of the prion protein. J. Biol. Chem. 271, 12661–12668 (1996).
Collinge, J. et al. Transmission of fatal familial insomnia to laboratory animals. Lancet 346, 569–570 (1995).
Tateishi, J. et al. First experimental transmission of fatal familial insomnia. Nature 376, 434–435 (1995).
Brown, P. et al. Phenotypic characteristics of familial Creutzfeldt-jakob disease associated with the codon 178Asn PRNP mutation. Ann Neurol. 31, 282–285 (1992).
Telling, G.C. et al. Evidence for the conformation of the pathologic isoform of the prion protein enciphering and propagating prion diversity. Science 274, 2079–2082 (1996).
Palmer, M.S., Dryden, A.J., Hughes, J.T. & Collinge, J. (1991). Homozygous prion protein genotype predisposes to sporadic Creutzfeldt-jakob disease. Nature 352, 340–342 (1991).
Cochran, E.J. et al. Familial Creutzfeldt-jakob disease with a five-repeat octapeptide insert mutation. Neurology 47, 727–733 (1996).
Bertoni, J.M., Brown, P., Goldfarb, L.G., Rubenstein, R. & Gajdusek, D.C. Familial Creutzfeldt-Jakob disease (codon 200 mutation) with supranuclear palsy. JAMA 268, 2413–2415 (1992).
Chapman, J. et al. Clinical heterogeneity and unusual presentations of Creutzfeldt-Jakob disease in Jewish patients with the PRNP codon 200 mutation. J. Neural. Neurosurg. Psychiatry 56, 1109–1112 (1993).
Chapman, J. et al. Fatal insomnia in a case of familial Creutzfeldt-Jakob disease with the codon 200Lys mutation. Neurology 46, 758–761 (1996).
Kascsak, R.J. et al. Mouse polyclonal and monoclonal antibody to scrapie-associated fibril proteins. J. Virol. 61, 3688–3693 (1987).
Merrill, C.R., Goldman, D., Sedman, S.A. & Ebert, M.H. Ultrasensitive stain for proteins in polyacrylamide gels shows regional variation in cerebrospinal fluid proteins. Science 211, 1437–1438 (1981).
Bayer, E.A., Safars, M. & Wilchek, M. Selective labeling of sulfhydryls and disulfides on blot transfers using avidin-biotin technology: Studies on purified proteins and erythrocyte membranes. Anal. Biochem. 161, 262–271 (1987).
Schagger, H. & von Jagow, G. Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal. Biochem. 166, 368–379 (1987).
Goldfarb, L.G. et al. Creutzfeldt-Jakob disease cosegregates with the codon 178Asn PRNP mutation in families of European origin. Ann. Neurol. 31, 274–281 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chen, S., Parchi, P., Brown, P. et al. Allelic origin of the abnormal prion protein isoform in familial prion diseases. Nat Med 3, 1009–1015 (1997). https://doi.org/10.1038/nm0997-1009
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0997-1009
This article is cited by
-
Prion strains viewed through the lens of cryo-EM
Cell and Tissue Research (2023)
-
Characterization of Anchorless Human PrP With Q227X Stop Mutation Linked to Gerstmann-Sträussler-Scheinker Syndrome In Vivo and In Vitro
Molecular Neurobiology (2021)
-
Familial human prion diseases associated with prion protein mutations Y226X and G131V are transmissible to transgenic mice expressing human prion protein
Acta Neuropathologica Communications (2018)
-
The influence of PRNP polymorphisms on human prion disease susceptibility: an update
Acta Neuropathologica (2015)
-
Molecular pathology of human prion disease
Acta Neuropathologica (2011)