Abstract
Over the past decade, it has become clear that there is an important subset of memory T cells that resides in tissues—tissue-resident memory T (TRM) cells. There is an emerging understanding that TRM cells have a role in human tissue-specific immune and inflammatory diseases. Furthermore, the nature of the molecular signals that maintain TRM cells in tissues is the subject of much investigation. In addition, whereas it is logical for TRM cells to be located in barrier tissues at interfaces with the environment, these cells have also been found in brain, kidney, joint and other non-barrier tissues in humans and mice. Given the biology and behavior of these cells, it is likely that they have a role in chronic relapsing and remitting diseases of both barrier and non-barrier tissues. In this Review we discuss recent insights into the biology of TRM cells with a particular focus on their roles in disease, both proven and putative.
Similar content being viewed by others
Main
Memory T cells provide rapid and highly effective protective immunity against previously encountered pathogens and can recognize a wide variety of antigens, including those from malignant tumors and environmental substances. It was previously thought that memory T cells consisted of two major subsets: central memory T (TCM) cells and effector memory T (TEM) cells1. TCM cells express the chemokine receptor CCR7 and the vascular addressin L selectin (CD62L), which enables them to access and enter lymph nodes from blood. TEM cells express low levels of CCR7 and CD62L but have receptors that allow them to access peripheral tissues (for example, the E-selectin ligand Cutaneous Lymphocyte Antigen (CLA), which grants them access to the skin, and α4β7, an integrin that allows them access to the gut2,3).
Over the past decade, it has become clear that there is another important subset of memory T cells: tissue-resident memory T cells, or TRM cells. Under physiological conditions, TRM cells reside in epithelial barrier tissues at the interface between the host and the environment, such as the gastrointestinal (GI) tract, respiratory tract, reproductive tract and skin. TRM cells can respond rapidly to pathogen challenge at these sites independently of recruitment of T cells from the blood4,5. They thus mediate the rapid protective immunity that is the hallmark of adaptive immune memory4. TRM cells in a barrier tissue are enriched for T cells specific for pathogens and other antigens that have been encountered previously through that barrier epithelium. Thus, the T cell receptor (TCR) repertoire of skin TRM cells is largely different from that of lung TRM cells, and both are different from that of gut TRM cells5. However, TRM cells are not simply memory T cells in an unexpected location; rather, they have a transcriptional program that distinguishes them from peripheral blood TEM cells and TCM cells6.
The cell-signaling interactions that maintain TRM cells in their resident tissues are the subject of much investigation. The role of TRM cells in human tissue-specific immune and inflammatory diseases is just beginning to be appreciated5. In addition, although it is logical for TRM cells to be stationed at interfaces with the environment, they have also been found in brain, kidney, joint and other non-barrier tissues. TRM cells that appear in non-barrier tissues have similar transcriptional programs7, and their location, biology and behavior make it likely that they have a role in chronic relapsing and remitting diseases of non-barrier tissues. Here we discuss how TRM cells are generated after an immune response and review both common and distinctive features of TRM cells in various barrier tissues, including skin, lung and GI tract. We also discuss how TRM cells may be formed in sterile non-barrier tissues such as brain and kidney and speculate as to the role of TRM cells in immune and inflammatory diseases involving tissues. Finally, we review the role of TRM cells in cancer and the goal of generating TRM cells through vaccination for both infectious diseases and cancer. The field is developing at a rapid rate, and new observations are being made on an ongoing basis.
Generation of TRM cells during an immune response
Naive T cells circulate between blood and lymph nodes, where they remain for 12–24 h before returning to the blood and then sampling another lymph node microenvironment8. Naive T cells are abundant but highly diverse with regard to TCR repertoire, and thus to pathogens to which they may be targeted, such that naive T cells specific for any given antigen are rare9. Dendritic cells are the first to encounter infectious challenge in peripheral tissues, and they ferry pathogen fragments to draining lymph nodes, where they present processed peptides (antigens) to naive T cells. Those T cells that recognize the antigen become activated and clonally expand, such that one naive T cell gives rise to tens of thousands of progeny9,10. Although all these T cells derived from a single naive T cell have the same TCR, the expanded T cell population becomes heterogeneous with regard to the homing molecules that are expressed11. Some gain the ability to access peripheral tissues, and others retain the capacity to enter lymph nodes from blood (TCM cells). Effector T cells also acquire new functions that are specific to the pathogen encountered; for example, type 1 helper T cells secrete interferon-γ (IFN-γ, a cytokine that induces a broad range of antiviral factors) in response to viral pathogens, and TH17 cells produce interleukin-17 (IL-17), a potent inducer of neutrophil activation and recruitment, in response to bacterial and fungal pathogens12.
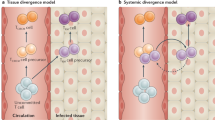
The anatomic location of the draining lymph node has an important role in determining the expression of tissue-homing molecules on formerly naive T cells first activated in that microenvironment11,13 (Fig. 1a). Naive T cells that are activated in skin-draining lymph nodes are induced to express CLA, a glycosylated variant of P-selectin glycoprotein ligand12,14,15,16 and a ligand for E-selectin, as well as a subset of chemokine receptors that facilitate skin homing (for example, CCR4, CCR8 and CCR10)17,18,19. Alternatively, activation of naive T cells in gut-draining lymph nodes induces expression of α4β7 integrin20,21, the receptor of mucosal addressin cell-adhesion molecule (MAdCAM) expressed on post-capillary venules in intestinal lamina propria22, as well as the expression of distinct chemokine receptors, including CCR9 (ref. 23), which binds to CCL25 produced by intestinal epithelium.
(a) Upon first encounter with a pathogen in a barrier tissue, dendritic cells carry antigen to draining lymph nodes and present it to naive T cells. Depending on the anatomic location of the lymph node, various trafficking molecules (indicated adjacent to vessels) are expressed on the expanding activated T cell population, and effector T cells with specific tissue-homing properties preferentially exit blood in peripheral tissues. Gut-draining lymph nodes induce the expression of gut-homing molecules on antigen-activated T cells, and skin-draining lymph nodes induce the expression of skin-homing molecules on antigen-activated T cells. Analogous processes, albeit less well characterized, occur in lymph nodes draining lung and reproductive mucosa. VLA-1, very late antigen 1 (α1β1 integrin). (b) Long after the pathogen has been eliminated from the barrier tissue and inflammation has resolved, populations of TRM cells remain behind in the tissue. These TRM cells retain the tissue-homing molecules originally imprinted on them and acquire a molecular program that contributes to the maintenance of these cells in peripheral tissue. In parallel, circulating memory T cells are generated, and these have the capacity to enter lymph node and recirculate into blood and tissue. Recent evidence suggests that the same naive T cell may give rise to both TRM cells and TCM cells or circulating T cells38. Adapted with permission from ref. 113, Nature Publishing Group.
The clonally expanding T cell population includes cells that differentiate into tissue-homing effector cells, but also cells that retain CD62L and CCR7 and resemble TCM cells. These latter cells leave the draining lymph node and travel to other lymph nodes, where a subset can differentiate to express different tissue-homing molecules11. After effector T cells exit a skin-draining lymph node and enter the blood, those with skin-homing markers are preferentially trapped by inflamed vessels in skin and are extravasated into dermis24. These T cells migrate along chemotactic gradients to the pathogen-infected tissue, where they become activated by antigen and produce cytokines and other effector molecules (for example, granzymes) that facilitate pathogen elimination25,26,27. It is now clear that some of these migrant T cells remain in place as TRM cells long after the pathogen is eliminated. This sequence of events can play out again and again, in multiple barrier tissues, over the lifetime of an organism, and the result is the accumulation of diverse and largely (but not completely) non-overlapping populations of TRM cells in each barrier tissue4,5. Thus, lung contains influenza-specific TRM cells28,29,30, gut contains rotavirus-specific TRM cells31, skin is enriched in Candida-specific TRM cells32,33 and reproductive mucosa contains herpes simplex virus (HSV)-specific TRM cells34,35,36,37 (Fig. 1b). Human peripheral blood T cells enriched in populations of skin (CLA), gut (α4β7) or lung (CLA and/or α4β7) tropic memory T cells are specific to previously encountered pathogens of those tissues32. It was recently shown that the same naive T cells that give rise to these TRM cells also give rise to TCM cells in parallel; thus the TRM cell population in tissue is 'duplicated in antigen specificity' by a population of TCM cells with an identical T cell repertoire38.
The process of TRM cell formation involves some additional nuances not mentioned above. For example, when mouse skin is infected with vaccinia virus by scarification, effector T cells accumulate not only at the vaccinated site but throughout the skin4, and this was recently shown to be true for skin immunization with proteins and haptens38. Non-inflamed skin contains post-capillary venules that express low levels of E-selectin, chemokines and intercellular adhesion molecule 1, all of the requisite molecules that allow skin-homing T cells24 to be extravasated and home to uninfected skin sites. In the same fashion, endothelial molecules specific for gut-homing T cells are expressed on resting lamina propria endothelium22, allowing gut-homing T cells to home to normal, non-inflamed gut. Additionally, multiple sequential encounters with a pathogen at distinct sites on skin lead to a further accumulation of pathogen-specific TRM cells throughout the entire skin surface; thus there is a greater number of TRM cells specific for pathogens encountered more frequently4,38.
Furthermore, whereas each naive T cell (and its progeny) has a unique TCR, the expanded clone is otherwise heterogeneous. TCM cells have limited effector function or protective capacity4, but they have the potential to replenish the TRM cell compartment upon activation38. The relationship between TRM cells and TCM cells is unclear, but both express low levels of KLRG-1 (ref. 6), a molecule strongly expressed by effector and TEM cells. These data, in addition to a recent report demonstrating skin TRM and lymph node TCM cell clones sharing the same variable sequence (CDR3) of the TCR38, indicate that many, if not all, TRM cells and TCM cells have a common precursor. With regard to the balance of TRM cells and TCM cells, a recent report suggests that the mTOR pathway may regulate this balance, as mTOR inhibitors such as rapamycin favor TCM cell generation in mouse models39. Another recent study suggests that high expression of the transcription factor T-box specific protein 21, or T-bet, favors TEM cell differentiation over TRM cell differentiation, with lower expression of this protein found in TRM cells40.
Although TRM cells are most likely to have evolved to protect humans against infection from dangerous environmental pathogens, it appears that normal flora of tissue microbiomes, as well as innocuous environmental proteins, can induce the production of TRM cells as well. A recent study in mice and humans found that allergic contact dermatitis was mediated by TRM cells that had been generated in response to topically applied allergens38. TRM cells were described as early as 2001 (ref. 41), and the same group demonstrated that TRM cells in gut do not recirculate between parabiotic mice, in contrast to T cells in lymph node and spleen42.
Common features of TRM cells in barrier tissues
Homing of TRM cells. TRM cells are characterized by their inability to recirculate from tissue to lymph node to blood4,43,44,45,46,47, although the factors responsible for this are an area of active research. The glycoprotein CD69 is a marker of TRM cells and is expressed on TRM cells in skin, lung, GI tract and everywhere else TRM cells have been identified4,5,6,28,48,49,50. CD69 was originally thought to be a marker of recent T cell activation in lymph node51; however, most TRM cells in tissues have not recently been activated by antigen. CD69 seems to be involved in peripheral tissue retention of TRM cells, which seems to involve downregulation of the G protein–coupled receptor for sphingosine 1 phosphate (S1P1)52. There is a gradient of levels of sphingosine 1 phosphate (S1P) in the body in humans and mice, with the lowest levels in peripheral tissue, intermediate levels in lymph node and the highest levels in blood50,53,54. These S1P gradients normally function to guide T cells out of tissues and into lymph nodes and out of lymph nodes into blood. Expression of CD69 by TRM cells interferes with the cell-surface expression and function of S1P1, thereby blocking the capacity of these T cells to sense S1P gradients and supporting their stationary nature50. The transcription factor Kruppel-like factor 2, which normally enhances S1P1 expression, is downregulated in TRM cells, which indirectly enables CD69 expression53. The mechanism by which CD69 and S1P1 compete with each other for cell-surface expression is not completely understood54.
The chemokine receptor CCR7 is another G protein–coupled receptor that senses molecular gradients of its ligands CCL19 and CCL21 and directs T cells and dendritic cells from peripheral tissues to lymph node via afferent lymphatics55. T cells expressing CCR7 are able to migrate in response to gradients of its chemokine ligands, which are normally not abundant in tissue but are at their highest levels in lymph node and afferent lymphatics. It was recently shown in a mouse model that CD4+ T cells in skin require CCR7 to migrate to afferent lymphatics and that blocking CCR7 expression prevents T cells from leaving skin56. In human skin, expression of CCR7 was seen on a population of T cells that migrated out of skin (so-called migratory memory T (TMM) cells), whereas CCR7− T cells remained in skin as TRM cells57. The respective contributions of S1P1-CD69 and CCR7 expression on T cells to the migration of these cells out of tissues have not been determined.
The integrin CD103 (also known as αE, and which pairs with the β7 integrin chain) is another marker of TRM cells; however, its expression is more predominant on CD8+ TRM cells than on CD4+ TRM cells. It is a known ligand of E-cadherin, a homotypic adhesion molecule expressed by epithelial cells in barrier tissues58. In mouse models, CD8+ T cells specific for HSV-1 do not express CD103 before they enter the skin, but they upregulate CD103 expression upon entering epidermis in response to transforming growth factor–β (TGF-β)6. CD103 is also expressed by TRM cells in the lung and GI tract, and even in TRM cells in the brain after CNS viral infection7,40,48,59. It is tempting to assume that αEβ7 on these cells binds to epithelial cells via interactions with E-cadherin. However, binding to E-cadherin is not required for tissue residence, as CD103+CD4+ and CD8+ TRM cells can be found in the dermis, and CD103+ dendritic cells are plentiful in the dermis without ever entering the epidermis60. Although E-cadherin is expressed during brain development, it is absent in adult CNS tissue61, despite abundant CD103 on brain CD8+ TRM cells in mice. Thus, although its role is incompletely understood, it does appear that CD103 expression is a marker of differentiation of TRM cells6 rather than a functional requirement for tissue residence. It is notable that CD103+ TRM cells have less proliferative potential and more significant effector cytokine–production capacity than do CD103− T cells in several human and mouse models7,40,41,48,59,62,63. Human skin T cells also are not strictly required to express CD103 in order to be TRM cells57. A recent study of a mouse model suggests that CD8+CD103− TRM cells may have a unique role in gut, as they are generated in inflammatory microenvironments in the lamina propria and have a distinctive role in controlling infection compared to their CD8+CD103+ counterparts64.
Less is known about CD4+ TRM cells than about CD8+ TRM cells, in part because these cells are less efficiently generated after viral infection in mouse models, in which TRM cells have been most completely characterized. Studies of HSV infection of the female mouse reproductive tract suggest that local chemokine gradients from tissue mononuclear cells maintain CD4+ TRM cells in place37. In skin, evidence suggests that CD4+ TRM cells do not preferentially localize to the epidermis and express lower levels of CD103 than do CD8+ T cells4,63. HSV-specific CD4+ T cells in mouse skin may be more mobile than CD8+ TRM cells, and they may also be limited to the dermis65. CD4+ T cells in skin may express CCR7 and/or CD69 (refs. 5,56). In a recent study, highly immunocompromised NOD SCID IL-2Rγ–deficient (NSG) mice bearing human skin xenografts were treated with alemtuzumab (an antibody that binds human CD52, a molecule present on all T cells). This humanized antibody has been shown to deplete human T cells in blood but not in tissue57,66. Two populations of CD4+ T cells were depleted by alemtuzumab from the skin xenografts of these mice: those that expressed both CCR7 and CD62L (markers of TCM cells), and those that expressed CCR7 but not CD62L (TMM cells). These data demonstrate that these two populations normally migrate out of skin into blood, and thus although they transiently reside in skin, they are not authentic TRM cells. The two populations of CD4+ T cells that remained in the skin after alemtuzumab treatment both expressed CD69 and lacked CCR7 (and were thus unresponsive to S1P and CCL19/21 gradients) and included both CD103+ and CD103− populations. Thus, four distinct populations of CD4+ T cells could be identified in human skin, two of which were short-term residents and could exit skin and enter blood, and two of which were true TRM cells57.
Maintaining TRM cells in tissues. The molecular factors that maintain TRM cells in their resident tissue are not completely understood, but IL-15, TGF-β, tumor necrosis factor–α (TNF-α) and IL-33 have all been implicated6,53. TGF-β, TNF-α and IL-33 have all been shown to have a role in the induction of CD103 expression and acquisition of a TRM cell phenotype. Factors that induce upregulation of CD69 expression include TNF-α and type I interferons50,67. The aryl hydrocarbon receptor is important for the maintenance of γδ T cells in mouse skin68, and a recent report suggests that it is important for the generation of αβ-TCR+CD8+ TRM cells69. A recently described additional common activity of CD8+ TRM cells is highlighted by several recent papers. One of the first cytokines made by virally induced CD8+ TRM cells upon antigen reactivation is IFN-γ. In both skin and reproductive mucosa, the IFN-γ released by activated TRM cells creates a generalized antiviral microenvironment in tissue by upregulating a series of antiviral and antimicrobial genes in surrounding keratinocytes, enhancing the expression of vascular adhesion molecule in endothelium and activating other resident cells, including natural killer (NK) cells and dendritic cells70,71. In this fashion, activated TRM cells can amplify and activate the innate immune system, creating an environment inhospitable to even completely unrelated viruses and other pathogens.
Properties of TRM cells in distinct barrier tissues
Skin TRM cells. In 2006, it was reported that normal resting human skin contains twice as many T cells as blood does5,72,73, and it is now appreciated that most but not all of these cells are TRM cells57 (Table 1). Thus, memory T cells generated in response to previous pathogen exposure in the cutaneous environment are present in abundance in the skin, allowing for immediate response to pathogenic invasion5. These cells have a diverse TCR repertoire and can be activated by pathogens at a much lower threshold than circulating T cells via TCRs72. Moreover, they are heterogeneous: they include CD4+ and CD8+ T cells that produce IL-17, IFN-γ, TNF-α, IL-9, IL-13 and other cytokines, alone or in combination5,32,72,73,74,75,76.
Mouse models have been instrumental in furthering our understanding of skin TRM cells. Early studies showed that in mice transfused with HSV peptide–specific transgenic T cells and then infected with HSV, the transgenic CD8+ T cells could be transferred from one mouse to another by a previously infected skin graft, and these transferred HSV-specific TRM cells maintained their ability to clear virus upon challenge63. In another study it was shown that vaccinia virus (VACV) administration by skin scarification was far superior to other routes of immunization in generating skin-resident CD8+ TRM cells77. The same investigators showed that skin TRM cells, in the absence of circulating T cells and antibody, can clear virus on re-challenge to skin. Furthermore, infection by skin scarification leads to the generation of lung TRM cells that, in the complete absence of circulating antibodies and circulating T cells, can partially protect immunized mice from a lethal pulmonary challenge with VACV77. Thus, skin immunization can lead to widespread TRM cells throughout the skin as well as in distant barrier tissues. This occurs not only at the site of infection but also at distant sites, and the number of accumulated CD8+ TRM cells throughout the skin increases after multiple infections at distinct sites4. However, CD8+ TRM cells do not recirculate, and mice that contain TCM cells but lack TRM cells cannot effectively clear VACV from skin, in contrast to mice that have immune skin TRM cells4. Another study showed that after HSV challenge in mice, HSV-specific CD8+ TRM cells migrate to the epidermis and acquire a sessile phenotype, whereas HSV-specific CD4+ TRM cells localize to the dermis and show greater mobility65.
Interestingly, the transcriptomes of TRM cells from skin, lung and gut have common core features in mice6. A study showed that localization of CD8+ TRM cells in the epidermis and CD103 expression on TRM cells were induced in the epidermis by TGF-β, and these CD8+ TRM cells homed to epidermis via a chemokine-mediated process6. Mouse CD8+ TRM cells were also shown to occupy epidermal niches formerly filled by a population of T cells that seed the epidermis before birth—γδ dendritic epidermal T cells (DETCs)—and when viewed by intravital microscopy these cells were found to move laterally between keratinocytes, unlike sessile γδ DETCs. These CD8+ TRM cells interacted transiently with Langerhans cells, suggesting that they were scanning the environment for antigen69. In humans, there are two isoforms of the dimeric CD8 molecule on T cells, composed of αβ and αα chains, respectively. After cutaneous HSV infection, CD8-αα+ TRM cells localize at the dermal epidermal junction. These cells, but not CD8-αβ+ T cells, protect against the reactivation of HSV and lesion formation78.
Somewhat less is known about CD4+ skin TRM cells in mice than about CD8+ skin TRM cells. It has been shown, however, that CD4+ regulatory T (Treg) cells are a major population of T cells emigrating from skin to lymph node after an immune response to contact hypersensitivity, as well as in the absence of stimulus79. Another group characterized CD4+ T cells in mouse skin and demonstrated at least two populations, including one that did not leave skin (lacking CCR7 expression) and another that left skin by a CCR7-dependent mechanism, expressed low levels of CD62L and high levels of E-selectin ligand and was negative for CD69 expression56. This is consistent with a very recently published study of T cells in human skin57.
TRM cells in the GI tract. TRM cells in gut are defined here as T cells that reside in the intestinal epithelium or in lamina propria80. It has been shown that a subset of CD103+ dendritic cells in gut-draining lymph node can skew naive T cells toward differentiation into α4β7+ gut-homing memory T cells, primarily under the influence of retinoic acid secreted by these dendritic cells81. Gut-infiltrating T cells have been most exhaustively studied in mice during disease states, such as experimentally induced colitis, in which mice lacking CD103 have attenuated inflammation, suggesting a role for TRM cells in inflammation. Until recently48,70, less attention had been paid to TRM cells that emerged after pathogen infection. Several pathogens, including lymphocytic choriomeningitis virus (LCMV), Listeria and others, have been shown to induce the generation of long-lived intraepithelial T cells with potent effector activities in mice42. Although many of these pathogens were delivered intravenously in past experiments, in a recent study mice were infected orally with Listeria, and the authors acquired information on gut TRM cells48. The study showed that long-lived gut TRM cells expressed KLRG-1 at low levels (similar to skin TRM cells), whereas cells with high expression of KLRG-1 (as in TEM cells) that entered gut underwent apoptosis. The oral immunization induced abundant long-lived gut TRM cells, unlike nasal immunization, which induced gut entry by T cells with high KLRG-1 expression that did not persist long term. Gut CD8+ TRM cells express CD69 and CD103, as do mouse skin TRM cells, and their maintenance in the gut is enhanced by TGF-β (ref. 80). When transcriptional profiles of skin TRM cells generated in response to HSV infection were compared to those of gut TRM cells induced by an LCMV infection6, of 127 genes up- or downregulated in TRM cells relative to expression in TCM cells, 68 showed a pattern common to skin and gut, and the remainder were unique to gut (or possibly to the difference between LCMV and HSV infection)6. Thus, the TRM cells that form in gut epithelium and lamina propria have many features in common with TRM cells in other barrier tissues, although they also express gut-specific homing molecules.
Regarding what is known about gut TRM cells in humans, a recent study that surveyed resident T cell populations in various human tissues demonstrated the presence of TRM cells in both colon and small intestine82. We have further analyzed human GI tissue by deep sequencing of TCRB and identified a highly diverse T cell repertoire in normal non-inflamed tissue (R.A. Clark et al., data not shown). Liver TRM cells have been demonstrated after malaria infection83, and the T cells that infiltrate the liver during viral hepatitis are likely to become tissue resident as well, causing significant and recurrent tissue injury in the absence of effective therapy84.
TRM cells in lung and respiratory tissue. The possibility that TRM cells might exist in lung was first considered after the identification of CD69+CD8+ TRM cells that remained in lung after influenza infection28. The original explanation for the expression of CD69 by these cells was that the cells were in an activated state, perhaps as a result of retained antigen; however, we now know that CD69 expression is a generic characteristic of resting TRM cells29. There is good evidence that CD8+ TRM cells can be protective against subsequent infection with influenza. Intranasal but not intraperitoneal infection with influenza in mice results in the presence of lung TRM cells, although both routes of infection efficiently produce influenza-specific splenic TEM cells30. Furthermore, in the referenced study, the nasal influenza–immunized mice, but not the intraperitoneally immunized mice, were protected against a lethal intranasal challenge with influenza. This echoes work in skin showing that resident, but not circulating, memory T cells are most effective at limiting viral replication at the site of viral entry4. However, in this lung-infection study, the TRM cells had essentially vanished from lung 90 d after a single influenza infection. Whether they could be made more abundant at this late stage via boosting strategies such as additional antigen challenge was not explored.
The anatomic location in lung where TRM cells need to reside in order to be most protective against a flu challenge is also controversial; it is clear that lung TRM cells express CD103, and its epithelial ligand, E-cadherin, is expressed most strongly on large and intermediate bronchial epithelial cells and less strongly on small or alveolar epithelia85. However, as discussed above, CD103+ cells can persist at a distance from cells expressing E-cadherin. Regarding the levels of CD4+ T cells versus those of CD8+ T cells, TGF-β promotes the development of lung CD103+CD8+ TRM cells, but in a fashion not dependent on Smad4 (ref. 86). CD4+ T cells in lung aid in the development of CD103+CD8+ TRM cells after influenza virus infection40, but the respective roles of CD4+ and CD8+ T cells, and whether (as in skin) these two populations have different migratory capacities, have not been explored.
There is a growing body of evidence that points to the existence of TRM cells in normal human lung. T cells have been observed in bronchoalveolar lavage samples, but these are typically acquired in the setting of diseased lung, and thus it is not known whether the cells noted are authentic TRM cells. After pneumonectomy for isolated tumors, human lung tissue very distal to the tumors is histologically normal in appearance87, and T cells isolated from such lung samples include CD4+ and CD8+ cells that produce TNF-α and IFN-γ (ref. 87), express CD69 and have a diverse TCR repertoire; from these data it was estimated that the number of TRM cells in lung approximates the number of T cells in blood—on the order of 10 billion cells. Moreover, these populations of T cells are enriched in the cells that proliferate in response to inactivated influenza virus87. Lung TRM cells express abundant α1β1, although this is expressed in other tissues and is thus not lung specific87. CD8+CD103+ T cells in human lung are specific for influenza, as opposed to CD8+CD103− T cells, which are also found in that tissue59. TRM cells in human lung were also found in a large survey study that looked at multiple human tissues82.
TRM cells in the genitourinary tract. The mucosa of the female reproductive tract is an important barrier tissue. In mouse vaginal HSV infection, CD4+ T cells must first enter the tissue and provide a recruiting cytokine and chemokine signal to facilitate the entry of CD8+ T cells into infected vaginal mucosa34. This is different from the process in skin, in which CD4 help is not required to recruit antigen-specific CD8+ T cells after VACV infection4. Protective immunity against HSV can also be generated by direct topical infection of the vaginal mucosa followed by the accumulation of TRM cells35, which suggests that the generation of TRM cells should be a goal of vaccination88. Independently performed studies comparing HSV-infected skin and mucosa have yielded analogous results36. Approaches such as these are likely to be used in attempts to generate a protective vaccine against HSV-2. Very recently, it was shown that a local chemokine gradient maintained HSV-specific CD4+ TRM cells in situ, a previously unrecognized mechanism for the maintenance of TRM cell residence89. Furthermore, it was recently shown that intravaginal administration of the human papillomavirus (HPV) vaccine in mice led to the generation of CD8+ TRM cells in vaginal mucosa90,91. This work is very promising, as not only HSV and HPV but also HIV can infect through this route, and the possibility of rapidly killing virally infected cells with TRM cells in situ, however they are generated, is appealing (Box 1)88. More recently, work on cervical tissue—normal, dysplastic and malignant—has demonstrated that vaccination against oncogenic papillomavirus leads to the generation of TRM cells in these tissues that are highly protective against reinfection92.
The role of TRM cells in human disease
Pathologic TRM cells in barrier tissues. The best-characterized role for TRM cells in disease is in mediating skin diseases (Fig. 2), with fixed drug eruption being the first and best described93. Recently, established psoriasis has been shown to be mediated largely by TRM cells. Transcriptomic analysis of resolved lesional psoriatic skin in humans has revealed the presence of T cells and cytokines thought to be important in pathogenesis, suggesting the persistence of these TRM cells94,95. More recently, analysis of cells extracted from resolved psoriatic lesions showed CD8+ T cells that produce IL-17 and CD4+ T cells that produce IL-22, providing additional support for the role of TRM cells in psoriasis96. A recent report showed that allergic contact dermatitis in both human and murine settings is also mediated by TRM cells38. In psoriasis, the putative antigen is considered to be an autoantigen, whereas in allergic contact dermatitis it is often an innocuous environmental molecule97, and in fixed drug eruption it is typically an orally ingested chemical. Recurrence of treated and resolved psoriasis lesions in the same place suggests that although the activity of disease-causing TRM cells was suppressed by therapy, their localization was unaffected. Vitiligo, as well as some forms of atopic and eczematous dermatitis, may also be mediated by TRM cells5; here the antigen is a melanocyte-specific antigen. Interestingly, a variant of cutaneous T cell lymphoma was found to be a malignancy of TRM cells98, and another variant (leukemic cutaneous T cell lymphoma or Sezary syndrome) is a malignancy of skin-homing TCM cells66,98 (Box 2).
Right, diseases of lung, gut and skin clearly or potentially mediated by pathologically activated TRM cells. Left, diseases of normally sterile non-barrier tissues mediated by infiltrating T cells that have acquired the properties of TRM cells. Disease states in normal font indicate that there is experimental evidence supporting TRM cell causation, whereas disease states in italic font are speculation on the part of the authors. GVHD, graft-versus-host disease.
The GI tract is another site where certain diseases exhibit the behavior of TRM cell–mediated diseases (Fig. 2). The discrete waxing and waning skip lesions—areas of disease separated by areas of normal mucosa—in Crohn's disease suggest a role for TRM cells, whereas ulcerative colitis involves a more contiguous circumferential area of the large intestine99. It is unknown, however, whether immune-mediated diseases of the lung (e.g., asthma) involve TRM cells (Fig. 2). There are no data that address this possibility, although the presence of TRM cells in normal human lung makes the hypothesis a reasonable one. Certainly, the excessive inflammation in lung in the setting of fatal influenza infection may well involve hyperactive TRM cells. Additionally, the fact that T cell–mediated diseases of the skin are mediated by TRM cells, and the finding that these diseases can often be treated with skin-directed rather than systemic therapy, suggests that the approach of local rather than systemic treatment may apply to other tissues as well.
Pathologic TRM cells in non-barrier tissues. It has been shown experimentally that TRM cell accumulation can also occur in tissues generally considered sterile, such as the brain7. TRM cells were identified in the brain after intranasal infection with vesicular stomatitis virus, and CD103+ TRM cells had a potent effector function after in vitro stimulation. The transcriptional profile of these brain CD8+ TRM cells resembled that of TRM cells in skin, gut and lung6,7. Whether such TRM cells can form in human brain after viral infection is unknown, and a putative role for these cells in diseases of the CNS requires additional evidence, although recent reports have linked putative pathogenic brain TRM cells to multiple sclerosis (Fig. 2) and even schizophrenia100,101. Although T cell responses in non-barrier tissues may be necessary episodically to deal with a potentially lethal infection, the unintended consequence of such an event may be the generation of long-lived TRM cells and the attendant predisposition to potential autoreactive and autoimmune diseases. One hypothesis is that the potential program for generating TRM cells exists in all activated T cells, and a subset of those that gain entry into tissue (whether a barrier tissue or a normally sterile tissue) show activation of this TRM cell program (Fig. 2).
Spondyloarthropathies such as human ankylosing spondylitis involve inflammation of entheseal tissues (sites of attachment of tendon to bone), and one recent study in mice demonstrated that entheseal resident TH17 TRM cells are essential for disease progression102. In human rheumatoid arthritis, the clinical recurrence of disease in individual joints bears the hallmarks of a TRM cell–driven process, and a preliminary report describes the presence of TRM cells in human joint synovium in rheumatoid arthritis103. It is thus likely that sterile chronic inflammation of peripheral tissues in human disease is mediated by these cells. For example, T cells from blood and kidney were examined in lupus nephritis, and a relatively limited set of T cell clones appeared to be responsible for progressive disease in individual patients, even over periods separated by months to years104. Although that study did not examine CD103 or CD69, it provides indirect proof of pathological renal TRM cells (Fig. 2). In murine models of insulin-dependent diabetes mellitus and pancreatic islet beta cell rejection, infiltrating CD8+ T cells acquire CD103 and remain in place during the immune response105. It is conceivable that in human type 1 diabetes mellitus, T cells that infiltrate pancreas and attack beta cells may take on the phenotype of TRM cells (Fig. 2), favoring their long-term persistence in situ. In solid organ allograft rejection, infiltrating allogeneic T cells are able to acquire TRM cell properties such as CD103 expression106, and urinary CD103 is associated with acute graft rejection107. If these human diseases involve pathological TRM cells, immunosuppressive regimens may suppress the activation of these cells but have no effect on their location and persistence, thereby setting the stage for recurrence and persistence of disease (Fig. 2).
Finally, there is evidence that some tissues of immune privilege, such as the eye, may have mechanisms to inactivate TRM cells induced by inflammation108 through expression of PD-L1 and promotion of TRM cell PD-1 expression109. Through mechanisms that include PD-1–PD-LI interactions, cancer tissues can acquire similar immune privilege. Binding of T cell PD-1 to its natural ligand PD-L1 induces a state of T cell unresponsiveness. Tumor-infiltrating lymphocytes (TILs) by definition become 'resident' in neoplastic tissue. It is notable that TILs with surface markers of TRM cells were found to be predictive of a more favorable prognosis in ovarian cancer110. The role of PD-1–PD-L1 interactions in suppressing the activity of TILs in malignant melanoma, and its reversal by therapeutic antibodies that block this interaction, is now well established111,112.
Conclusions
In barrier tissues at interfaces with the environment, TRM cells are an important part of adaptive immune memory, providing the capacity to rapidly address and clear tissue infections caused by previously encountered pathogens. When TRM cells pathologically accumulate in barrier tissues in response to innocuous antigens, disease can result. The molecular program that facilitates the TRM cell phenotype in barrier tissues can be activated in other tissues as well, where persistent immune-driven inflammation can cause chronic disease. Previously, when T cells were seen in pathologic infiltrates, it was assumed that this represented chronic and dynamic T cell infiltration, regardless of the tissue involved. It is more than a semantic difference to propose that these infiltrates in fact include resident populations of T cells in which a TRM cell molecular program has been activated. If this is the case, therapies that suppress T cell function do not necessarily change T cell localization, and reactivated TRM cells will mediate recurrent disease. Whether in CNS, joint, pancreas, kidney or heart, persistent and activated TRM cells in sterile tissues (where they were not intended to be) may drive human autoimmune and inflammatory diseases. Therapies directed at selectively eliminating these TRM cells, by depletion or through modification of the cells' ability to persistently reside in tissue, represent potential approaches to the treatment of such diseases.
It is worth noting that skin TRM cells have been unknowingly targeted for decades, with relevant treatments far predating appreciation of these cells' existence. Skin-directed therapies, ranging from topical corticosteroids to UV-based phototherapy to low-dose radiation, have all led to the remission of what are now understood as TRM cell–mediated diseases. Would gut mucosal–directed therapy via endoscope or synovial-directed therapy via arthroscope suppress TRM cells in those tissues? The advantage of skin-targeted therapy is that repetitive therapy is straightforward and noninvasive, and the results can be assessed without sophisticated imaging techniques. Other approaches might be directed at features that maintain TRM cells in tissue, namely, CD69 and CD103. Blocking or interfering with the function of these molecules might flush pathogenic TRM cells out of tissues, although of course a balance would have to be struck in the depletion of pathogenic TRM cells and physiologically protective normal TRM cells. Finally, it was noted that TRM cells in immune-privileged sites such as the eye express PD-1, and presumably remain quiescent in this fashion. We speculate that TILs are a form of TRM cell and that the tumor may induce PD-1 expression on these tumor TRM cells to suppress their activity. In tumor immunotherapy, the goal is to interfere with this immune suppression with antibodies that block PD-1–PD-L1 interactions. However, if this suppressive pathway exploited for immune privilege could be exploited in non-cancer diseases where unrestrained TRM cell activity causes tissue inflammation and injury, yet another approach to suppressing disease-causing TRM cells would exist. None of these approaches (save those long employed in skin) is more than hypothetical at present. However, what is clear is that although simply suppressing the activation of TRM cells in psoriasis, inflammatory bowel disease or inflammatory arthritis may lead to transient clinical remission, disease recurrence is nearly inevitable if TRM cells persist (as they are designed to do) in tissue. The next decade of TRM cell biology will be devoted to modifying the cells' behavior and, perhaps, long-term residence and location.
References
Sallusto, F., Lenig, D., Forster, R., Lipp, M. & Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401, 708–712 (1999).
Fuhlbrigge, R.C., Kieffer, J.D., Armerding, D. & Kupper, T.S. Cutaneous lymphocyte antigen is a specialized form of PSGL-1 expressed on skin-homing T cells. Nature 389, 978–981 (1997).
Mackay, C.R. et al. Tissue-specific migration pathways by phenotypically distinct subpopulations of memory T cells. Eur. J. Immunol. 22, 887–895 (1992).
Jiang, X. et al. Skin infection generates non-migratory memory CD8+ T(RM) cells providing global skin immunity. Nature 483, 227–231 (2012).
Clark, R.A. Resident memory T cells in human health and disease. Sci. Transl. Med. 7, 269rv261 (2015).
Mackay, L.K. et al. The developmental pathway for CD103+CD8+ tissue-resident memory T cells of skin. Nat. Immunol. 14, 1294–1301 (2013).
Wakim, L.M. et al. The molecular signature of tissue resident memory CD8 T cells isolated from the brain. J. Immunol. 189, 3462–3471 (2012).
von Andrian, U.H. & Mempel, T.R. Homing and cellular traffic in lymph nodes. Nat. Rev. Immunol. 3, 867–878 (2003).
Tubo, N.J. et al. Single naive CD4+ T cells from a diverse repertoire produce different effector cell types during infection. Cell 153, 785–796 (2013).
von Andrian, U.H. & Mackay, C.R. T-cell function and migration. Two sides of the same coin. N. Engl. J. Med. 343, 1020–1034 (2000).
Liu, L., Fuhlbrigge, R.C., Karibian, K., Tian, T. & Kupper, T.S. Dynamic programming of CD8+ T cell trafficking after live viral immunization. Immunity 25, 511–520 (2006).
Sallusto, F. & Lanzavecchia, A. Heterogeneity of CD4+ memory T cells: functional modules for tailored immunity. Eur. J. Immunol. 39, 2076–2082 (2009).
Campbell, D.J. & Butcher, E.C. Rapid acquisition of tissue-specific homing phenotypes by CD4+ T cells activated in cutaneous or mucosal lymphoid tissues. J. Exp. Med. 195, 135–141 (2002).
Picker, L.J. et al. Control of lymphocyte recirculation in man. II. Differential regulation of the cutaneous lymphocyte-associated antigen, a tissue-selective homing receptor for skin-homing T cells. J. Immunol. 150, 1122–1136 (1993).
Borowitz, M.J., Weidner, A., Olsen, E.A. & Picker, L.J. Abnormalities of circulating T-cell subpopulations in patients with cutaneous T-cell lymphoma: cutaneous lymphocyte-associated antigen expression on T cells correlates with extent of disease. Leukemia 7, 859–863 (1993).
Berg, E.L. et al. The cutaneous lymphocyte antigen is a skin lymphocyte homing receptor for the vascular lectin endothelial cell-leukocyte adhesion molecule 1. J. Exp. Med. 174, 1461–1466 (1991).
Campbell, J.J. et al. The chemokine receptor CCR4 in vascular recognition by cutaneous but not intestinal memory T cells. Nature 400, 776–780 (1999).
Campbell, J.J., Pan, J. & Butcher, E.C. Cutting edge: developmental switches in chemokine responses during T cell maturation. J. Immunol. 163, 2353–2357 (1999).
Homey, B. et al. CCL27-CCR10 interactions regulate T cell–mediated skin inflammation. Nat. Med. 8, 157–165 (2002).
Siewert, C. et al. Induction of organ-selective CD4+ regulatory T cell homing. Eur. J. Immunol. 37, 978–989 (2007).
Iwata, M. et al. Retinoic acid imprints gut-homing specificity on T cells. Immunity 21, 527–538 (2004).
Briskin, M. et al. Human mucosal addressin cell adhesion molecule-1 is preferentially expressed in intestinal tract and associated lymphoid tissue. Am. J. Pathol. 151, 97–110 (1997).
Zabel, B.A. et al. Human G protein-coupled receptor GPR-9-6/CC chemokine receptor 9 is selectively expressed on intestinal homing T lymphocytes, mucosal lymphocytes, and thymocytes and is required for thymus-expressed chemokine-mediated chemotaxis. J. Exp. Med. 190, 1241–1256 (1999).
Chong, B.F., Murphy, J.E., Kupper, T.S. & Fuhlbrigge, R.C. E-selectin, thymus- and activation-regulated chemokine/CCL17, and intercellular adhesion molecule-1 are constitutively coexpressed in dermal microvessels: a foundation for a cutaneous immunosurveillance system. J. Immunol. 172, 1575–1581 (2004).
Kupper, T.S. & Fuhlbrigge, R.C. Immune surveillance in the skin: mechanisms and clinical consequences. Nat. Rev. Immunol. 4, 211–222 (2004).
Mackay, C.R. & von Andrian, U.H. Immunology. Memory T cells—local heroes in the struggle for immunity. Science 291, 2323–2324 (2001).
Robert, C. & Kupper, T.S. Inflammatory skin diseases, T cells, and immune surveillance. N. Engl. J. Med. 341, 1817–1828 (1999).
Hogan, R.J. et al. Activated antigen-specific CD8+ T cells persist in the lungs following recovery from respiratory virus infections. J. Immunol. 166, 1813–1822 (2001).
Wei, C.H. et al. Tissue-resident memory CD8+ T cells can be deleted by soluble, but not cross-presented antigen. J. Immunol. 175, 6615–6623 (2005).
Wu, T. et al. Lung-resident memory CD8 T cells (TRM) are indispensable for optimal cross-protection against pulmonary virus infection. J. Leukoc. Biol. 95, 215–224 (2014).
Kuklin, N.A. et al. α4β7 independent pathway for CD8+ T cell-mediated intestinal immunity to rotavirus. J. Clin. Invest. 106, 1541–1552 (2000).
Schlapbach, C. et al. Human TH9 cells are skin-tropic and have autocrine and paracrine proinflammatory capacity. Sci. Transl. Med. 6, 219ra218 (2014).
Seneschal, J., Clark, R.A., Gehad, A., Baecher-Allan, C.M. & Kupper, T.S. Human epidermal Langerhans cells maintain immune homeostasis in skin by activating skin resident regulatory T cells. Immunity 36, 873–884 (2012).
Nakanishi, Y., Lu, B., Gerard, C. & Iwasaki, A. CD8+ T lymphocyte mobilization to virus-infected tissue requires CD4+ T-cell help. Nature 462, 510–513 (2009).
Shin, H. & Iwasaki, A. A vaccine strategy that protects against genital herpes by establishing local memory T cells. Nature 491, 463–467 (2012).
Mackay, L.K. et al. Long-lived epithelial immunity by tissue-resident memory T (TRM) cells in the absence of persisting local antigen presentation. Proc. Natl. Acad. Sci. USA 109, 7037–7042 (2012).
Iijima, N. & Iwasaki, A. T cell memory. A local macrophage chemokine network sustains protective tissue-resident memory CD4 T cells. Science 346, 93–98 (2014).
Gaide, O. et al. Common clonal origin of central and resident memory T cells following skin immunization. Nat. Med. 21, 647–653 (2015).
Sowell, R.T., Rogozinska, M., Nelson, C.E., Vezys, V. & Marzo, A.L. Cutting edge: generation of effector cells that localize to mucosal tissues and form resident memory CD8 T cells is controlled by mTOR. J. Immunol. 193, 2067–2071 (2014).
Laidlaw, B.J. et al. CD4+ T cell help guides formation of CD103+ lung-resident memory CD8+ T cells during influenza viral infection. Immunity 41, 633–645 (2014).
Masopust, D., Vezys, V., Marzo, A.L. & Lefrancois, L. Preferential localization of effector memory cells in nonlymphoid tissue. Science 291, 2413–2417 (2001).
Klonowski, K.D. et al. Dynamics of blood-borne CD8 memory T cell migration in vivo. Immunity 20, 551–562 (2004).
Mueller, S.N., Gebhardt, T., Carbone, F.R. & Heath, W.R. Memory T cell subsets, migration patterns, and tissue residence. Annu. Rev. Immunol. 31, 137–161 (2013).
Bevan, M.J. Memory T cells as an occupying force. Eur. J. Immunol. 41, 1192–1195 (2011).
Gebhardt, T., Mueller, S.N., Heath, W.R. & Carbone, F.R. Peripheral tissue surveillance and residency by memory T cells. Trends Immunol. 34, 27–32 (2013).
Carbone, F.R., Mackay, L.K., Heath, W.R. & Gebhardt, T. Distinct resident and recirculating memory T cell subsets in non-lymphoid tissues. Curr. Opin. Immunol. 25, 329–333 (2013).
Mueller, S.N., Zaid, A. & Carbone, F.R. Tissue-resident T cells: dynamic players in skin immunity. Front. Immunol. 5, 332 (2014).
Sheridan, B.S. et al. Oral infection drives a distinct population of intestinal resident memory CD8+ T cells with enhanced protective function. Immunity 40, 747–757 (2014).
Beura, L.K. & Masopust, D. SnapShot: resident memory T cells. Cell 157, 1488–1488 (2014).
Turner, D.L. & Farber, D.L. Mucosal resident memory CD4 T cells in protection and immunopathology. Front. Immunol. 5, 331 (2014).
Shiow, L.R. et al. CD69 acts downstream of interferon-α/β to inhibit S1P1 and lymphocyte egress from lymphoid organs. Nature 440, 540–544 (2006).
Mackay, L.K. et al. Cutting Edge: CD69 interference with sphingosine-1-phosphate receptor function regulates peripheral T cell retention. J. Immunol. 194, 2059–2063 (2015).
Skon, C.N. et al. Transcriptional downregulation of S1pr1 is required for the establishment of resident memory CD8+ T cells. Nat. Immunol. 14, 1285–1293 (2013).
Cyster, J.G. & Schwab, S.R. Sphingosine-1-phosphate and lymphocyte egress from lymphoid organs. Annu. Rev. Immunol. 30, 69–94 (2012).
Bromley, S.K., Thomas, S.Y. & Luster, A.D. Chemokine receptor CCR7 guides T cell exit from peripheral tissues and entry into afferent lymphatics. Nat. Immunol. 6, 895–901 (2005).
Bromley, S.K., Yan, S., Tomura, M., Kanagawa, O. & Luster, A.D. Recirculating memory T cells are a unique subset of CD4+ T cells with a distinct phenotype and migratory pattern. J. Immunol. 190, 970–976 (2013).
Watanabe, R. et al. Human skin is protected by four functionally and phenotypically discrete populations of resident and recirculating memory T cells. Sci. Transl. Med. 7, 279ra239 (2015).
Hadley, G.A. & Higgins, J.M. Integrin αEβ7: molecular features and functional significance in the immune system. Adv. Exp. Med. Biol. 819, 97–110 (2014).
Piet, B. et al. CD8+ T cells with an intraepithelial phenotype upregulate cytotoxic function upon influenza infection in human lung. J. Clin. Invest. 121, 2254–2263 (2011).
Nestle, F.O., Di Meglio, P., Qin, J.Z. & Nickoloff, B.J. Skin immune sentinels in health and disease. Nat. Rev. Immunol. 9, 679–691 (2009).
Shimamura, K. & Takeichi, M. Local and transient expression of E-cadherin involved in mouse embryonic brain morphogenesis. Development 116, 1011–1019 (1992).
Reinhardt, R.L., Khoruts, A., Merica, R., Zell, T. & Jenkins, M.K. Visualizing the generation of memory CD4 T cells in the whole body. Nature 410, 101–105 (2001).
Gebhardt, T. et al. Memory T cells in nonlymphoid tissue that provide enhanced local immunity during infection with herpes simplex virus. Nat. Immunol. 10, 524–530 (2009).
Bergsbaken, T. & Bevan, M.J. Proinflammatory microenvironments within the intestine regulate the differentiation of tissue-resident CD8+ T cells responding to infection. Nat. Immunol. 16, 406–414 (2015).
Gebhardt, T. et al. Different patterns of peripheral migration by memory CD4 and CD8 T cells. Nature 477, 216–219 (2011).
Clark, R.A. et al. Skin effector memory T cells do not recirculate and provide immune protection in alemtuzumab-treated CTCL patients. Sci. Transl. Med. 4, 117ra117 (2012).
Casey, K.A. et al. Antigen-independent differentiation and maintenance of effector-like resident memory T cells in tissues. J. Immunol. 188, 4866–4875 (2012).
Kadow, S. et al. Aryl hydrocarbon receptor is critical for homeostasis of invariant γδ T cells in the murine epidermis. J. Immunol. 187, 3104–3110 (2011).
Zaid, A. et al. Persistence of skin-resident memory T cells within an epidermal niche. Proc. Natl. Acad. Sci. USA 111, 5307–5312 (2014).
Schenkel, J.M. et al. T cell memory. Resident memory CD8 T cells trigger protective innate and adaptive immune responses. Science 346, 98–101 (2014).
Ariotti, S. et al. T cell memory. Skin-resident memory CD8+ T cells trigger a state of tissue-wide pathogen alert. Science 346, 101–105 (2014).
Clark, R.A. Skin-resident T cells: the ups and downs of on site immunity. J. Invest. Dermatol. 130, 362–370 (2010).
Clark, R.A. et al. The vast majority of CLA+ T cells are resident in normal skin. J. Immunol. 176, 4431–4439 (2006).
Clark, R.A. et al. A novel method for the isolation of skin resident T cells from normal and diseased human skin. J. Invest. Dermatol. 126, 1059–1070 (2006).
Clark, R.A. & Kupper, T.S. IL-15 and dermal fibroblasts induce proliferation of natural regulatory T cells isolated from human skin. Blood 109, 194–202 (2007).
Hijnen, D. et al. CD8+ T cells in the lesional skin of atopic dermatitis and psoriasis patients are an important source of IFN-γ, IL-13, IL-17, and IL-22. J. Invest. Dermatol. 133, 973–979 (2013).
Liu, L. et al. Epidermal injury and infection during poxvirus immunization is crucial for the generation of highly protective T cell-mediated immunity. Nat. Med. 16, 224–227 (2010).
Zhu, J. et al. Immune surveillance by CD8αα+ skin-resident T cells in human herpes virus infection. Nature 497, 494–497 (2013).
Tomura, M. et al. Activated regulatory T cells are the major T cell type emigrating from the skin during a cutaneous immune response in mice. J. Clin. Invest. 120, 883–893 (2010).
Masopust, D. et al. Dynamic T cell migration program provides resident memory within intestinal epithelium. J. Exp. Med. 207, 553–564 (2010).
Ruane, D.T. & Lavelle, E.C. The role of CD103+ dendritic cells in the intestinal mucosal immune system. Front. Immunol. 2, 25 (2011).
Sathaliyawala, T. et al. Distribution and compartmentalization of human circulating and tissue-resident memory T cell subsets. Immunity 38, 187–197 (2013).
Tse, S.W., Cockburn, I.A., Zhang, H., Scott, A.L. & Zavala, F. Unique transcriptional profile of liver-resident memory CD8+ T cells induced by immunization with malaria sporozoites. Genes Immun. 14, 302–309 (2013).
Yanagisawa, K. et al. Ex vivo analysis of resident hepatic pro-inflammatory CD1d-reactive T cells and hepatocyte surface CD1d expression in hepatitis C. J. Viral Hepat. 20, 556–565 (2013).
Turner, D.L. et al. Lung niches for the generation and maintenance of tissue-resident memory T cells. Mucosal Immunol. 7, 501–510 (2014).
Hu, Y., Lee, Y.T., Kaech, S.M., Garvy, B. & Cauley, L.S. Smad4 promotes differentiation of effector and circulating memory CD8 T cells but is dispensable for tissue-resident memory CD8 T cells. J. Immunol. 194, 2407–2414 (2015).
Purwar, R. et al. Resident memory T cells (T(RM)) are abundant in human lung: diversity, function, and antigen specificity. PLoS One 6, e16245 (2011).
Kupper, T.S. Old and new: recent innovations in vaccine biology and skin T cells. J. Invest. Dermatol. 132, 829–834 (2012).
Iijima, N. & Iwasaki, A. A local macrophage chemokine network sustains protective tissue-resident memory CD4 T cells. Science 346, 93–98 (2014).
Cuburu, N. et al. Intravaginal immunization with HPV vectors induces tissue-resident CD8+ T cell responses. J. Clin. Invest. 122, 4606–4620 (2012).
Cuburu, N. et al. Topical herpes simplex virus 2 (HSV-2) vaccination with human papillomavirus vectors expressing gB/gD ectodomains induces genital-tissue-resident memory CD8+ T cells and reduces genital disease and viral shedding after HSV-2 challenge. J. Virol. 89, 83–96 (2015).
Maldonado, L. et al. Intramuscular therapeutic vaccination targeting HPV16 induces T cell responses that localize in mucosal lesions. Sci. Transl. Med. 6, 221ra213 (2014).
Shiohara, T. Fixed drug eruption: pathogenesis and diagnostic tests. Curr. Opin. Allergy Clin. Immunol. 9, 316–321 (2009).
Suarez-Farinas, M., Fuentes-Duculan, J., Lowes, M.A. & Krueger, J.G. Resolved psoriasis lesions retain expression of a subset of disease-related genes. J. Invest. Dermatol. 131, 391–400 (2011).
Clark, R.A. Gone but not forgotten: lesional memory in psoriatic skin. J. Invest. Dermatol. 131, 283–285 (2011).
Cheuk, S. et al. Epidermal Th22 and Tc17 cells form a localized disease memory in clinically healed psoriasis. J. Immunol. 192, 3111–3120 (2014).
Honda, T., Egawa, G., Grabbe, S. & Kabashima, K. Update of immune events in the murine contact hypersensitivity model: toward the understanding of allergic contact dermatitis. J. Invest. Dermatol. 133, 303–315 (2013).
Campbell, J.J., Clark, R.A., Watanabe, R. & Kupper, T.S. Sezary syndrome and mycosis fungoides arise from distinct T-cell subsets: a biologic rationale for their distinct clinical behaviors. Blood 116, 767–771 (2010).
Kleinschek, M.A. et al. Circulating and gut-resident human Th17 cells express CD161 and promote intestinal inflammation. J. Exp. Med. 206, 525–534 (2009).
Sasaki, K. et al. Relapsing-remitting central nervous system autoimmunity mediated by GFAP-specific CD8 T cells. J. Immunol. 192, 3029–3042 (2014).
Debnath, M. & Berk, M. Th17 pathway-mediated immunopathogenesis of schizophrenia: mechanisms and implications. Schizophr. Bull. 40, 1412–1421 (2014).
Sherlock, J.P. et al. IL-23 induces spondyloarthropathy by acting on ROR-γt+ CD3+CD4−CD8− entheseal resident T cells. Nat. Med. 18, 1069–1076 (2012).
Henderson, L.A. et al. A161: novel 3-dimensional explant method facilitates the study of lymphocyte populations in the synovium and reveals a large population of resident memory T cells in rheumatoid arthritis. Arthritis Rheum. 66 (suppl. 11), S209 (2014).
Zhou, G. et al. Identification of systemically expanded activated T cell clones in MRL/lpr and NZB/W F1 lupus model mice. Clin. Exp. Immunol. 136, 448–455 (2004).
Feng, Y. et al. CD103 expression is required for destruction of pancreatic islet allografts by CD8+ T cells. J. Exp. Med. 196, 877–886 (2002).
Wang, D. et al. Regulation of CD103 expression by CD8+ T cells responding to renal allografts. J. Immunol. 172, 214–221 (2004).
Ding, R. et al. CD103 mRNA levels in urinary cells predict acute rejection of renal allografts. Transplantation 75, 1307–1312 (2003).
Boldison, J. et al. Tissue-resident exhausted effector memory CD8+ T cells accumulate in the retina during chronic experimental autoimmune uveoretinitis. J. Immunol. 192, 4541–4550 (2014).
Freeman, G.J. et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J. Exp. Med. 192, 1027–1034 (2000).
Webb, J.R., Milne, K., Watson, P., Deleeuw, R.J. & Nelson, B.H. Tumor-infiltrating lymphocytes expressing the tissue resident memory marker CD103 are associated with increased survival in high-grade serous ovarian cancer. Clin. Cancer Res. 20, 434–444 (2014).
Hamid, O. et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N. Engl. J. Med. 369, 134–144 (2013).
Robert, C. et al. Nivolumab in previously untreated melanoma without BRAF mutation. N. Engl. J. Med. 372, 320–330 (2015).
Sheridan, B.S. & Lefrançois, L. Regional and mucosal memory T cells. Nat. Immunol. 12, 485–491 (2011).
Jiang, X., Campbell, J.J. & Kupper, T.S. Embryonic trafficking of γδ T cells to skin is dependent on E/P selectin ligands and CCR4. Proc. Natl. Acad. Sci. USA 107, 7443–7448 (2010).
Gray, E.E., Suzuki, K. & Cyster, J.G. Cutting edge: identification of a motile IL-17-producing γδ T cell population in the dermis. J. Immunol. 186, 6091–6095 (2011).
Sumaria, N. et al. Cutaneous immunosurveillance by self-renewing dermal γδ T cells. J. Exp. Med. 208, 505–518 (2011).
Cai, Y. et al. Pivotal role of dermal IL-17-producing γδ T cells in skin inflammation. Immunity 35, 596–610 (2011).
Sanchez Rodriguez, R. et al. Memory regulatory T cells reside in human skin. J. Clin. Invest. 124, 1027–1036 (2014).
Hansen, S.G. et al. Profound early control of highly pathogenic SIV by an effector memory T-cell vaccine. Nature 473, 523–527 (2011).
Gardner, J.M., Evans, K.G., Musiek, A., Rook, A.H. & Kim, E.J. Update on treatment of cutaneous T-cell lymphoma. Curr. Opin. Oncol. 21, 131–137 (2009).
Watanabe, R., Teague, J.E., Fisher, D.C., Kupper, T.S. & Clark, R.A. Alemtuzumab therapy for leukemic cutaneous T-cell lymphoma: diffuse erythema as a positive predictor of complete remission. JAMA Dermatol. 150, 776–779 (2014).
Rao, U.N., Lee, S.J., Luo, W., Mihm, M.C. Jr. & Kirkwood, J.M. Presence of tumor-infiltrating lymphocytes and a dominant nodule within primary melanoma are prognostic factors for relapse-free survival of patients with thick (t4) primary melanoma: pathologic analysis of the e1690 and e1694 intergroup trials. Am. J. Clin. Pathol. 133, 646–653 (2010).
Tumeh, P.C. et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 515, 568–571 (2014).
Webb, J.R., Milne, K. & Nelson, B.H. Location, location, location: CD103 demarcates intraepithelial, prognostically favorable CD8 tumor-infiltrating lymphocytes in ovarian cancer. Oncoimmunology 3, e27668 (2014).
Djenidi, F. et al. CD8+CD103+ tumor-infiltrating lymphocytes are tumor-specific tissue-resident memory T cells and a prognostic factor for survival in lung cancer patients. J. Immunol. 194, 3475–3486 (2015).
Acknowledgements
This work was supported by funding from the US National Institutes of Health (R01 AR065807 and TR01 AI097128 to T.S.K.). Discussions with R.A. Clark are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
T.S.K. is an inventor on US Patent 8691502, assigned to TremRx, Inc.
Rights and permissions
About this article
Cite this article
Park, C., Kupper, T. The emerging role of resident memory T cells in protective immunity and inflammatory disease. Nat Med 21, 688–697 (2015). https://doi.org/10.1038/nm.3883
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3883
This article is cited by
-
Gut immune cell trafficking: inter-organ communication and immune-mediated inflammation
Nature Reviews Gastroenterology & Hepatology (2023)
-
Assessing the generation of tissue resident memory T cells by vaccines
Nature Reviews Immunology (2023)
-
CD103+CD8+ tissue-resident memory T cell infiltration predicts clinical outcome and adjuvant therapeutic benefit in muscle-invasive bladder cancer
British Journal of Cancer (2022)
-
Tissue-resident memory T cells in the kidney
Seminars in Immunopathology (2022)
-
CD39+PD-1+CD8+ T cells mediate metastatic dormancy in breast cancer
Nature Communications (2021)