Abstract
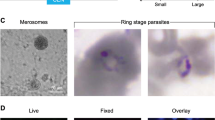
Malaria relapses, resulting from the activation of quiescent hepatic hypnozoites of Plasmodium vivax and Plasmodium ovale, hinder global efforts to control and eliminate malaria. As primaquine, the only drug capable of eliminating hypnozoites, is unsuitable for mass administration, an alternative drug is needed urgently. Currently, analyses of hypnozoites, including screening of compounds that would eliminate them, can only be made using common macaque models, principally Macaca rhesus and Macaca fascicularis, experimentally infected with the relapsing Plasmodium cynomolgi. Here, we present a protocol for long-term in vitro cultivation of P. cynomolgi–infected M. fascicularis primary hepatocytes during which hypnozoites persist and activate to resume normal development. In a proof-of-concept experiment, we obtained evidence that exposure to an inhibitor of histone modification enzymes implicated in epigenetic control of gene expression induces an accelerated rate of hypnozoite activation. The protocol presented may further enable investigations of hypnozoite biology and the search for compounds that kill hypnozoites or disrupt their quiescence.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Garnham, P.C.C. Malaria Parasites and Other Haemosporidia (Blackwell Scientific Publications, Oxford, 1966).
Coatney, G.R., Collins, W.E., Warren, M. & Contacos, P.G. The Primate Malarias (US Government Printing Office, Washington, DC, 1971).
Shortt, H.E. & Garnham, P.C.C. Demonstration of a persisting exo-erythrocytic cycle in P. cynomolgi and its bearing on the production of relapses. BMJ 1, 1225–1228 (1948).
Krotoski, W.A. et al. Relapses in primate malaria: discovery of two populations of exoerythrocytic stages. Preliminary note. BMJ 280, 153–154 (1980).
Krotoski, W.A. et al. Observations on early and late post-sporozoite tissue stages in primate malaria. I. Discovery of a new latent form of Plasmodium cynomolgi (the hypnozoite), and failure to detect hepatic forms within the first 24 hours after infection. Am. J. Trop. Med. Hyg. 31, 24–35 (1982).
Cogswell, F.B. The hypnozoite and relapse in primate malaria. Clin. Microbiol. Rev. 5, 26–35 (1992).
Shute, P.G. et al. A strain of Plasmodium vivax characterized by prolonged incubation: the effect of numbers of sporozoites on the length of the prepatent period. Trans. R. Soc. Trop. Med. Hyg. 70, 474–481 (1976).
Mazier, D., Rénia, L. & Snounou, G. A pre-emptive strike against malaria's stealthy hepatic forms. Nat. Rev. Drug Discov. 8, 854–864 (2009).
Wells, T.N.C., Burrows, J.N. & Baird, J.K. Targeting the hypnozoite reservoir of Plasmodium vivax: the hidden obstacle to malaria elimination. Trends Parasitol. 26, 145–151 (2010).
Schmidt, L.H. et al. Plasmodium cynomolgi infections in the rhesus monkey. Am. J. Trop. Med. Hyg. 31, 609–703 (1982).
Kocken, C.H.M. et al. Statistical model to evaluate in vivo activities of antimalarial drugs in a Plasmodium cynomolgi-macaque model for Plasmodium vivax malaria. Antimicrob. Agents Chemother. 53, 421–427 (2009).
Dembélé, L. et al. Towards an in vitro model of Plasmodium hypnozoites suitable for drug discovery. PLoS ONE 6, e18162 (2011).
Bray, R.S. Studies on the Exo-erythrocytic Cycle in the Genus Plasmodium (Lewis & Co. Ltd, London, 1957).
Prudêncio, M., Rodriguez, A. & Mota, M.M. The silent path to thousands of merozoites: the Plasmodium liver stage. Nat. Rev. Microbiol. 4, 849–856 (2006).
Guguen-Guillouzo, C. & Guillouzo, A. General review on in vitro hepatocyte models and their applications. Methods Mol. Biol. 640, 1–40 (2010).
Clement, B. et al. Long-term co-cultures of adult human hepatocytes with rat liver epithelial cells: modulation of albumin secretion and accumulation of extracellular material. Hepatology 4, 373–380 (1984).
Mazier, D. et al. Cultivation of the liver forms of Plasmodium vivax in human hepatocytes. Nature 307, 367–369 (1984).
Gripon, P. et al. Infection of a human hepatoma cell line by hepatitis B virus. Proc. Natl. Acad. Sci. USA 99, 15655–15660 (2002).
Sturm, A. et al. Manipulation of host hepatocytes by the malaria parasite for delivery into liver sinusoids. Science 313, 1287–1290 (2006).
Dunn, J.C., Yarmush, M.L., Koebe, H.G. & Tompkins, R.G. Hepatocyte function and extracellular matrix geometry: long-term culture in a sandwich configuration. FASEB J. 3, 174–177 (1989).
Kono, Y., Yang, S. & Roberts, E.A. Extended primary culture of human hepatocytes in a collagen gel sandwich system. In Vitro Cell. Dev. Biol. Anim. 33, 467–472 (1997).
Bissell, D.M., Arenson, D.M., Maher, J.J. & Roll, F.J. Support of cultured hepatocytes by a laminin-rich gel. Evidence for a functionally significant subendothelial matrix in normal rat liver. J. Clin. Invest. 79, 801–812 (1987).
Lopez-Rubio, J.J., Mancio-Silva, L. & Scherf, A. Genome-wide analysis of heterochromatin associates clonally variant gene regulation with perinuclear repressive centers in malaria parasites. Cell Host Microbe 5, 179–190 (2009).
Malmquist, N.A., Moss, T.A., Mecheri, S., Scherf, A. & Fuchter, M.J. Small-molecule histone methyltransferase inhibitors display rapid antimalarial activity against all blood stage forms in Plasmodium falciparum. Proc. Natl. Acad. Sci. USA 109, 16708–16713 (2012).
White, N.J. Determinants of relapse periodicity in Plasmodium vivax malaria. Malar. J. 10, 297 (2011).
Atkinson, C.T., Millet, P.G., Collins, W.E. & Aikawa, M. Localization of circumsporozoite antigen in exoerythrocytic schizonts of Plasmodium cynomolgi. Am. J. Trop. Med. Hyg. 40, 131–140 (1989).
Barter, Z.E. et al. Scaling factors for the extrapolation of in vivo metabolic drug clearance from in vitro data: reaching a consensus on values of human microsomal protein and hepatocellularity per gram of liver. Curr. Drug Metab. 8, 33–45 (2007).
Hollingdale, M.R., Collins, W.E., Campbell, C.C. & Schwartz, A.L. In vitro culture of two populations (dividing and nondividing) of exoerythrocytic parasites of Plasmodium vivax. Am. J. Trop. Med. Hyg. 34, 216–222 (1985).
Hollingdale, M.R., Collins, W.E. & Campbell, C.C. In vitro culture of exoerythrocytic parasites of the North Korean strain of Plasmodium vivax in hepatoma cells. Am. J. Trop. Med. Hyg. 35, 275–276 (1986).
Liu, D., Luo, S., Ye, B., Shu, H. & Fu, R. Demonstration of in vitro cultured exoerythrocytic schizonts and hypnozoites of Plasmodium vivax (southern China isolate) by an immunoperoxidase antibody technique. Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi 13, 86–88 (1995).
Shu, H., Lou, S., Liu, D. & Fu, R. [Observation on hypnozoite of different isolates of Plasmodium vivax in cultured materials]. Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi 13, 185–188 (1995).
Chattopadhyay, R. et al. Establishment of an in vitro assay for assessing the effects of drugs on the liver stages of Plasmodium vivax malaria. PLoS ONE 5, e14275 (2010).
Hoffman, S.L. et al. Development of a metabolically active, non-replicating sporozoite vaccine to prevent Plasmodium falciparum malaria. Hum. Vaccin. 6, 97–106 (2010).
Voorberg-van der Wel, A. et al. Transgenic fluorescent Plasmodium cynomolgi liver stages enable live imaging and purification of malaria hypnozoite-forms. PLoS ONE 8, e54888 (2013).
Painter, H.J., Campbell, T.L. & Llinás, M. The Apicomplexan AP2 family: integral factors regulating Plasmodium development. Mol. Biochem. Parasitol. 176, 1–7 (2011).
Liu, F. et al. Protein lysine methyltransferase G9a inhibitors: design, synthesis, and structure activity relationships of 2,4-diamino-7-aminoalkoxy-quinazolines. J. Med. Chem. 53, 5844–5857 (2010).
Silvie, O. et al. Hepatocyte CD81 is required for Plasmodium falciparum and Plasmodium yoelii sporozoite infectivity. Nat. Med. 9, 93–96 (2003).
Castaing, M. et al. Efficient restricted gene expression in beta cells by lentivirus-mediated gene transfer into pancreatic stem/progenitor cells. Diabetologia 48, 709–719 (2005).
Acknowledgements
This work was supported in part by a translational research grant (WT078285) from the Wellcome Trust, a grant from the Medicines for Malaria Venture to the Novartis Institute for Tropical Diseases (Singapore), the Biomedical Primate Research Centre (the Netherlands), the Université Pierre et Marie Curie–Paris 6 (France), a Seventh Framework Programme EU European Research Council Executive Agency Advanced Grant (Plasmoescape 250320) to A.S. and a Laboratoires d'Excellence (LABEX) grant (ANR-11-LABX0024) from the Agence Nationale de la Recherche (France). We are grateful to T.A. Moss (Imperial College London) for the chemical synthesis of TM2-115. HepaRG cells were provided by C. Guguen-Guillouzo (Biopredic International). pTRIP-CMV-GFP ΔU3 vector was provided by P. Ravassard (le Centre de Recherche de l'Institut du Cerveau et de la Moelle Epinière). We thank the staff of the Flow Cytometry Core CyPS at the Université Pierre et Marie Curie (Pitié-Salpêtrière Hospital, Paris, France) for their help and assistance. Finally, we thank A.C. Grüner for her excellent critical comments on the manuscript.
Author information
Authors and Affiliations
Contributions
D.M., G.S. and A.S. designed the study. L.D., J.-F.F., A.L. and A.G. designed and performed the experiments. A.-M.Z., C.H.M.K., R.L.G., N.D.-B., G.-J.v.G., R.S., J.-C.V., L.H., M.J.F., T.T.D. and N.A.M. contributed essential materials. G.S. and D.M. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
T.T.D. is a Novartis employee and owns Novartis shares.
Rights and permissions
About this article
Cite this article
Dembélé, L., Franetich, JF., Lorthiois, A. et al. Persistence and activation of malaria hypnozoites in long-term primary hepatocyte cultures. Nat Med 20, 307–312 (2014). https://doi.org/10.1038/nm.3461
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3461
This article is cited by
-
Comparative analyses of functional antibody-mediated inhibition with anti-circumsporozoite monoclonal antibodies against transgenic Plasmodium berghei
Malaria Journal (2023)
-
Ethiopian Plasmodium vivax hypnozoites formation dynamics and their susceptibility to reference antimalarial drugs
BMC Infectious Diseases (2023)
-
Systems biology of malaria explored with nonhuman primates
Malaria Journal (2022)
-
Probing the distinct chemosensitivity of Plasmodium vivax liver stage parasites and demonstration of 8-aminoquinoline radical cure activity in vitro
Scientific Reports (2021)
-
Plasmodium vivax liver stage assay platforms using Indian clinical isolates
Malaria Journal (2020)