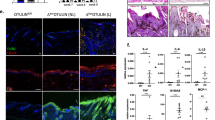
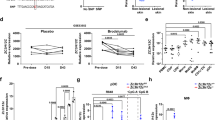
Abstract
The mitogen-activated protein kinase p38 mediates cellular responses to injurious stress and immune signaling. Among the many p38 isoforms, p38α is the most widely expressed in adult tissues and can be targeted by various pharmacological inhibitors. Here we investigated how p38α activation is linked to cell type–specific outputs in mouse models of cutaneous inflammation. We found that both myeloid and epithelial p38α elicit inflammatory responses, yet p38α signaling in each cell type served distinct inflammatory functions and varied depending on the mode of skin irritation. In addition, myeloid p38α limited acute inflammation via activation of anti-inflammatory gene expression dependent on mitogen- and stress-activated kinases. Our results suggest a dual function for p38α in the regulation of inflammation and show mixed potential for its inhibition as a therapeutic strategy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Lee, J.C. et al. A protein kinase involved in the regulation of inflammatory cytokine biosynthesis. Nature 372, 739–746 (1994).
Han, J., Lee, J.D., Bibbs, L. & Ulevitch, R.J.A. MAP kinase targeted by endotoxin and hyperosmolarity in mammalian cells. Science 265, 808–8 (1994).
Rouse, J. et al. A novel kinase cascade triggered by stress and heat shock that stimulates MAPKAP kinase-2 and phosphorylation of the small heat shock proteins. Cell 78, 1027–1037 (1994).
Freshney, N.W. et al. Interleukin-1 activates a novel protein kinase cascade that results in the phosphorylation of Hsp27. Cell 78, 1039–1049 (1994).
Kumar, S., Boehm, J. & Lee, J.C. p38 MAP kinases: key signalling molecules as therapeutic targets for inflammatory diseases. Nat. Rev. Drug Discov. 2, 717–726 (2003).
Ashwell, J.D. The many paths to p38 mitogen-activated protein kinase activation in the immune system. Nat. Rev. Immunol. 6, 532–540 (2006).
Goedert, M., Cuenda, A., Craxton, M., Jakes, R. & Cohen, P. Activation of the novel stress-activated protein kinase SAPK4 by cytokines and cellular stresses is mediated by SKK3 (MKK6); comparison of its substrate specificity with that of other SAP kinases. EMBO J. 16, 3563–3571 (1997).
Kumar, S. et al. Novel homologues of CSBP/p38 MAP kinase: activation, substrate specificity and sensitivity to inhibition by pyridinyl imidazoles. Biochem. Biophys. Res. Commun. 235, 533–538 (1997).
Jiang, Y. et al. Characterization of the structure and function of the fourth member of p38 group mitogen-activated protein kinases, p38δ. J. Biol. Chem. 272, 30122–30128 (1997).
Hale, K.K., Trollinger, D., Rihanek, M. & Manthey, C.L. Differential expression and activation of p38 mitogen-activated protein kinase α, β, γ, and δ in inflammatory cell lineages. J. Immunol. 162, 4246–4252 (1999).
Zarubin, T. & Han, J. Activation and signaling of the p38 MAP kinase pathway. Cell Res. 15, 11–18 (2005).
Roux, P.P. & Blenis, J. ERK and p38 MAPK-activated protein kinases: a family of protein kinases with diverse biological functions. Microbiol. Mol. Biol. Rev. 68, 320–344 (2004).
Zaru, R., Ronkina, N., Gaestel, M., Arthur, J.S. & Watts, C. The MAPK-activated kinase Rsk controls an acute Toll-like receptor signaling response in dendritic cells and is activated through two distinct pathways. Nat. Immunol. 8, 1227–1235 (2007).
Karaman, M.W. et al. A quantitative analysis of kinase inhibitor selectivity. Nat. Biotechnol. 26, 127–132 (2008).
Tamura, K. et al. Requirement for p38α in erythropoietin expression: a role for stress kinases in erythropoiesis. Cell 102, 221–231 (2000).
Adams, R.H. et al. Essential role of p38α MAP kinase in placental but not embryonic cardiovascular development. Mol. Cell 6, 109–116 (2000).
Allen, M. et al. Deficiency of the stress kinase p38α results in embryonic lethality: characterization of the kinase dependence of stress responses of enzyme-deficient embryonic stem cells. J. Exp. Med. 191, 859–870 (2000).
Mudgett, J.S. et al. Essential role for p38α mitogen-activated protein kinase in placental angiogenesis. Proc. Natl. Acad. Sci. USA 97, 10454–10459 (2000).
Hui, L. et al. p38α suppresses normal and cancer cell proliferation by antagonizing the JNK-c-Jun pathway. Nat. Genet. 39, 741–749 (2007).
Ventura, J.J. et al. p38α MAP kinase is essential in lung stem and progenitor cell proliferation and differentiation. Nat. Genet. 39, 750–758 (2007).
Beardmore, V.A. et al. Generation and characterization of p38β (MAPK11) gene-targeted mice. Mol. Cell. Biol. 25, 10454–10464 (2005).
Nishida, K. et al. p38α mitogen-activated protein kinase plays a critical role in cardiomyocyte survival but not in cardiac hypertrophic growth in response to pressure overload. Mol. Cell. Biol. 24, 10611–10620 (2004).
Clausen, B.E., Burkhardt, C., Reith, W., Renkawitz, R. & Förster, I. Conditional gene targeting in macrophages and granulocytes using LysMcre mice. Transgenic Res. 8, 265–277 (1999).
Jonkers, J. Synergistic tumor suppressor activity of BRCA2 and p53 in a conditional mouse model for breast cancer. Nat. Genet. 29, 418–425 (2001).
Thepen, T. et al. Resolution of cutaneous inflammation after local elimination of macrophages. Nat. Biotechnol. 18, 48–51 (2000).
Cramer, T. et al. HIF-1α is essential for myeloid cell-mediated inflammation. Cell 112, 645–657 (2003).
Park, J.M. et al. Signaling pathways and genes that inhibit pathogen-induced macrophage apoptosis–CREB and NF-κB as key regulators. Immunity 23, 319–329 (2005).
Carl, V.S., Gautam, J.K., Comeau, L.D. & Smith, M.F. Jr. Role of endogenous IL-10 in LPS-induced STAT3 activation and IL-1 receptor antagonist gene expression. J. Leukoc. Biol. 76, 735–742 (2004).
Berg, D.J. et al. Interleukin 10 but not interleukin 4 is a natural suppressant of cutaneous inflammatory responses. J. Exp. Med. 182, 99–108 (1995).
Grimbaldeston, M.A., Nakae, S., Kalesnikoff, J., Tsai, M. & Galli, S.J. Mast cell-derived interleukin 10 limits skin pathology in contact dermatitis and chronic irradiation with ultraviolet B. Nat. Immunol. 8, 1095–1104 (2007).
Siewe, L. et al. Interleukin-10 derived from macrophages and/or neutrophils regulates the inflammatory response to LPS but not the response to CpG DNA. Eur. J. Immunol. 36, 3248–3255 (2006).
Zhang, H., Shi, X., Hampong, M., Blanis, L. & Pelech, S. Stress-induced inhibition of ERK1 and ERK2 by direct interaction with p38 MAP kinase. J. Biol. Chem. 276, 6905–6908 (2001).
Cheung, P.C., Campbell, D.G., Nebreda, A.R. & Cohen, P. Feedback control of the protein kinase TAK1 by SAPK2a/p38α. EMBO J. 22, 5793–5805 (2003).
Mathur, R.K., Awasthi, A., Wadhone, P., Ramanamurthy, B. & Saha, B. Reciprocal CD40 signals through p38MAPK and ERK-1/2 induce counteracting immune responses. Nat. Med. 10, 540–544 (2004).
Perdiguero, E. et al. Genetic analysis of p38 MAP kinases in myogenesis: fundamental role of p38α in abrogating myoblast proliferation. EMBO J. 26, 1245–1256 (2007).
Kamata, H. et al. Reactive oxygen species promote TNFα-induced death and sustained JNK activation by inhibiting MAP kinase phosphatases. Cell 120, 649–661 (2005).
Dolado, I. et al. p38α MAP kinase as a sensor of reactive oxygen species in tumorigenesis. Cancer Cell 11, 191–205 (2007).
Salojin, K.V. et al. Essential role of MAPK phosphatase-1 in the negative control of innate immune responses. J. Immunol. 176, 1899–1907 (2006).
Chi, H. et al. Dynamic regulation of pro- and anti-inflammatory cytokines by MAPK phosphatase 1 (MKP-1) in innate immune responses. Proc. Natl. Acad. Sci. USA 103, 2274–2279 (2006).
Zhao, Q. et al. MAP kinase phosphatase 1 controls innate immune responses and suppresses endotoxic shock. J. Exp. Med. 203, 131–140 (2006).
Hammer, M. et al. Dual specificity phosphatase 1 (DUSP1) regulates a subset of LPS-induced genes and protects mice from lethal endotoxin shock. J. Exp. Med. 203, 15–20 (2006).
Martin, M., Rehani, K., Jope, R.S. & Michalek, S.M. Toll-like receptor-mediated cytokine production is differentially regulated by glycogen synthase kinase 3. Nat. Immunol. 6, 777–784 (2005).
Hu, X. et al. IFN-γ suppresses IL-10 production and synergizes with TLR2 by regulating GSK3 and CREB/AP-1 proteins. Immunity 24, 563–574 (2006).
Greten, F.R. et al. NF-κB is a negative regulator of IL-1β secretion as revealed by genetic and pharmacological inhibition of IKKβ. Cell 130, 918–931 (2007).
Ananieva, O. et al. The kinases MSK1 and MSK2 act as negative regulators of Toll-like receptor signaling. Nat. Immunol. (in the press).
Dambach, D.M. Potential adverse effects associated with inhibition of p38α/β MAP kinases. Curr. Top. Med. Chem. 5, 929–939 (2005).
Park, J.M., Greten, F.R., Li, Z.W. & Karin, M. Macrophage apoptosis by anthrax lethal factor through p38 MAP kinase inhibition. Science 297, 2048–2051 (2002).
Hu, Y. et al. IKKα controls formation of the epidermis independently of NF-κB. Nature 410, 710–714 (2001).
Vakeva, A.P. et al. Myocardial infarction and apoptosis after myocardial ischemia and reperfusion: role of the terminal complement components and inhibition by anti-C5 therapy. Circulation 97, 2259–2267 (1998).
Saccani, S. & Natoli, G. Dynamic changes in histone H3 Lys 9 methylation occurring at tightly regulated inducible inflammatory genes. Genes Dev. 16, 2219–2224 (2002).
Acknowledgements
We thank S. Krane (Massachusetts General Hospital) for the MMP-13-specific antibody and advice on its use; D. Ginsburg (University of Michigan School of Medicine) for the PAI-2-specific antibody; R. Bravo (Bristol-Myers Squibb Pharmaceutical Research Institute) and C. Caelles (Institute for Research in Biomedicine, Barcelona) for Dusp1-KO mice; and M. Karin for discussion about Jnk-activation mechanisms in p38α-deficient cells. Supported by the Cutaneous Biology Research Center through the Massachusetts General Hospital–Shiseido Agreement (J.M.P.), the US National Institutes of Health (DK043351 to D. Podolsky) and the Center for the Study of Inflammatory Bowel Disease at Massachusetts General Hospital (J.M.P.).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–11 and Table 1 (PDF 3097 kb)
Rights and permissions
About this article
Cite this article
Kim, C., Sano, Y., Todorova, K. et al. The kinase p38α serves cell type–specific inflammatory functions in skin injury and coordinates pro- and anti-inflammatory gene expression. Nat Immunol 9, 1019–1027 (2008). https://doi.org/10.1038/ni.1640
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1640
This article is cited by
-
Targeting the RAS/RAF/MAPK pathway for cancer therapy: from mechanism to clinical studies
Signal Transduction and Targeted Therapy (2023)
-
Genistein protects against ultraviolet B–induced wrinkling and photoinflammation in in vitro and in vivo models
Genes & Nutrition (2022)
-
Interleukin-10 deficiency exacerbates inflammation-induced tau pathology
Journal of Neuroinflammation (2021)
-
Circular RNA circACSL1 aggravated myocardial inflammation and myocardial injury by sponging miR-8055 and regulating MAPK14 expression
Cell Death & Disease (2021)
-
Rebelled epigenome: histone H3S10 phosphorylation and H3S10 kinases in cancer biology and therapy
Clinical Epigenetics (2020)