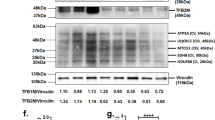
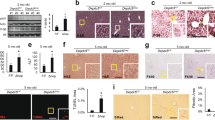
Abstract
Hypoxia-inducible factor (HIF) transcription factors respond to multiple environmental stressors, including hypoxia and hypoglycemia. We report that mice lacking the HIF family member HIF-2α (encoded by Epas1) have a syndrome of multiple-organ pathology, biochemical abnormalities and altered gene expression patterns. Histological and ultrastructural analyses showed retinopathy, hepatic steatosis, cardiac hypertrophy, skeletal myopathy, hypocellular bone marrow, azoospermia and mitochondrial abnormalities in these mice. Serum and urine metabolite studies showed hypoglycemia, lactic acidosis, altered Krebs cycle function and dysregulated fatty acid oxidation. Biochemical assays showed enhanced generation of reactive oxygen species (ROS), whereas molecular analyses indicated reduced expression of genes encoding the primary antioxidant enzymes (AOEs). Transfection analyses showed that HIF-2α could efficiently transactivate the promoters of the primary AOEs. Prenatal or postnatal treatment of Epas1−/− mice with a superoxide dismutase (SOD) mimetic reversed several aspects of the null phenotype. We propose a rheostat role for HIF-2α that allows for the maintenance of ROS as well as mitochondrial homeostasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Semenza, G.L. Hypoxia-inducible factor 1: oxygen homeostasis and disease pathophysiology. Trends Mol. Med. 7, 345–350 (2001).
Wang, G.L., Jiang, B.H., Rue, E.A. & Semenza, G.L. Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc. Natl. Acad. Sci. USA 92, 5510–5514 (1995).
Tian, H., McKnight, S.L. & Russell, D.W. Endothelial PAS domain protein 1 (EPAS1), a transcription factor selectively expressed in endothelial cells. Genes Dev. 11, 72–82 (1997).
Lando, D., Pongratz, I., Poellinger, L. & Whitelaw, M.L. A redox mechanism controls differential DNA binding activities of hypoxia-inducible factor (HIF) 1α and the HIF-like factor. J. Biol. Chem. 275, 4618–4627 (2000).
Wood, S.M. et al. Selection and analysis of a mutant cell line defective in the hypoxia-inducible factor-1 α-subunit (HIF-1α). Characterization of hif-1α-dependent and -independent hypoxia-inducible gene expression. J. Biol. Chem. 273, 8360–8368 (1998).
O'Rourke, J.F., Tian, Y.M., Ratcliffe, P.J. & Pugh, C.W. Oxygen-regulated and transactivating domains in endothelial PAS protein 1: comparison with hypoxia-inducible factor-1α. J. Biol. Chem. 274, 2060–2071 (1999).
Jain, S., Maltepe, E., Lu, M.M., Simon, C. & Bradfield, C.A. Expression of ARNT, ARNT2, HIFα, HIF2α and Ah receptor mRNAs in the developing mouse. Mech. Dev. 73, 117–123 (1998).
Iyer, N.V. et al. Cellular and developmental control of O2 homeostasis by hypoxia-inducible factor 1α. Genes Dev. 12, 149–162 (1998).
Ryan, H.E., Lo, J. & Johnson, R.S. HIF-1α is required for solid tumor formation and embryonic vascularization. EMBO J. 17, 3005–3015 (1998).
Tian, H., Hammer, R.E., Matsumoto, A.M., Russell, D.W. & McKnight, S.L. The hypoxia-responsive transcription factor EPAS1 is essential for catecholamine homeostasis and protection against heart failure during embryonic development. Genes Dev. 12, 3320–3324 (1998).
Peng, J., Zhang, L., Drysdale, L. & Fong, G.H. The transcription factor EPAS-1/hypoxia-inducible factor 2α plays an important role in vascular remodeling. Proc. Natl. Acad. Sci. USA 97, 8386–8391 (2000).
Scortegagna, M., Morris, M.A., Oktay, Y., Bennett, M. & Garcia, J.A. The HIF family member EPAS1/HIF-2α is required for normal hematopoiesis in mice. Blood 102, 1634–1640 (2003).
Rifai, Z., Welle, S., Kamp, C. & Thornton, C.A. Ragged red fibers in normal aging and inflammatory myopathy. Ann. Neurol. 37, 24–29 (1995).
Wallace, D.C. Mitochondrial diseases in man and mouse. Science 283, 1482–1488 (1999).
Boveris, A., Alvarez, S., Bustamante, J. & Valdez, L. Measurement of superoxide radical and hydrogen peroxide production in isolated cells and subcellular organelles. Methods Enzymol. 349, 280–287 (2002).
Guyton, K.Z., Spitz, D.R. & Holbrook, N.J. Expression of stress response genes GADD153, c-jun, and heme oxygenase-1 in H2O2- and O2-resistant fibroblasts. Free Radic. Biol. Med. 20, 735–741 (1996).
Ghoshal, K., Majumder, S., Li, Z., Bray, T.M. & Jacob, S.T. Transcriptional induction of metallothionein-I and -II genes in the livers of Cu,Zn-superoxide dismutase knockout mice. Biochem. Biophys. Res. Commun. 264, 735–742 (1999).
Warner, B.B., Stuart, L., Gebb, S. & Wispe, J.R. Redox regulation of manganese superoxide dismutase. Am. J. Physiol. 271, L150–L158 (1996).
Rogers, R.J., Monnier, J.M. & Nick, H.S. Tumor necrosis factor-α selectively induces MnSOD expression via mitochondria-to-nucleus signaling, whereas interleukin-1β utilizes an alternative pathway. J. Biol. Chem. 276, 20419–20427 (2001).
Jackson, R.M., Parish, G. & Helton, E.S. Peroxynitrite modulates MnSOD gene expression in lung epithelial cells. Free Radic. Biol. Med. 25, 463–472 (1998).
Wang, G.L., Jiang, B.H. & Semenza, G.L. Effect of altered redox states on expression and DNA-binding activity of hypoxia-inducible factor 1. Biochem. Biophys. Res. Commun. 212, 550–556 (1995).
Melov, S. et al. A novel neurological phenotype in mice lacking mitochondrial manganese superoxide dismutase. Nat. Genet. 18, 159–163 (1998).
Huang, T.-T. et al. Genetic modification of prenatal lethality and dilated cardiomyopathy in Mn superoxide dismutase mutant mice. Free Radic. Biol. Med. 31, 1101–1110 (2001).
Schisler, N.J. & Singh, S.M. A quantitative genetic analysis of tissue-specific catalase activity in Mus musculus. Biochem. Genet. 29, 65–89 (1991).
Schisler, N.J. & Singh, S.M. Tissue-specific developmental regulation of superoxide dismutase (SOD-1 and SOD-2) activities in genetic strains of mice. Biochem. Genet. 23, 291–308 (1985).
Phelan, S.A., Johnson, K.A., Beier, D.R. & Paigen, B. Characterization of the murine gene encoding Aop2 (antioxidant protein 2) and identification of two highly related genes. Genomics 54, 132–139 (1998).
DiMauro, S. & Schon, E.A. Mitochondrial respiratory-chain diseases. N. Engl. J. Med. 348, 2656–2668 (2003).
Horiuchi, M. et al. Primary defect of juvenile visceral steatosis (jvs) mouse with systemic carnitine deficiency is probably in renal carnitine transport system. Biochim. Biophys. Acta 1226, 25–30 (1994).
Miyagawa, J. et al. Mitochondrial abnormalities of muscle tissue in mice with juvenile visceral steatosis associated with systemic carnitine deficiency. Virchows Arch. 426, 271–279 (1995).
Kaido, M. et al. Mitochondrial abnormalities in a murine model of primary carnitine deficiency. Systemic pathology and trial of replacement therapy. Eur. Neurol. 38, 302–309 (1997).
Kamiya, H. et al. Antioxidant changes in the hypertrophied heart due to energy metabolic disorder. Basic Res. Cardiol. 96, 431–438 (2001).
Graham, B.H. et al. A mouse model for mitochondrial myopathy and cardiomyopathy resulting from a deficiency in the heart/muscle isoform of the adenine nucleotide translocator. Nat. Genet. 16, 226–234 (1997).
Esposito, L.A., Melov, S., Panov, A., Cottrell, B.A. & Wallace, D.C. Mitochondrial disease in mouse results in increased oxidative stress. Proc. Natl. Acad. Sci. USA 96, 4820–4825 (1999).
Li, Y. et al. Dilated cardiomyopathy and neonatal lethality in mutant mice lacking manganese superoxide dismutase. Nat. Genet. 11, 376–381 (1995).
Lebovitz, R.M. et al. Neurodegeneration, myocardial injury, and perinatal death in mitochondrial superoxide dismutase-deficient mice. Proc. Natl. Acad. Sci. USA 93, 9782–9787 (1996).
Friedman, J.S. et al. Absence of mitochondrial superoxide dismutase results in a murine hemolytic anemia responsive to therapy with a catalytic antioxidant. J. Exp. Med. 193, 925–934 (2001).
Williams, M.D. et al. Increased oxidative damage is correlated to altered mitochondrial function in heterozygous manganese superoxide dismutase knockout mice. J. Biol. Chem. 273, 28510–28515 (1998).
Kokoszka, J.E., Coskun, P., Esposito, L.A. & Wallace, D.C. Increased mitochondrial oxidative stress in the Sod2+/− mouse results in the age-related decline of mitochondrial function culminating in increased apoptosis. Proc. Natl. Acad. Sci. USA 98, 2278–2283 (2001).
Kuhn, D.M., Aretha, C.W. & Geddes, T.J. Peroxynitrite inactivation of tyrosine hydroxylase: mediation by sulfhydryl oxidation, not tyrosine nitration. J. Neurosci. 19, 10289–10294 (1999).
Ischiropoulos, H., Duran, D. & Horwitz, J. Peroxynitrite-mediated inhibition of DOPA synthesis in PC12 cells. J. Neurochem. 65, 2366–2372 (1995).
Lai, C.T. & Yu, P.H. Dopamine- and L-β-3,4-dihydroxyphenylalanine hydrochloride (L-Dopa)-induced cytotoxicity towards catecholaminergic neuroblastoma SH-SY5Y cells. Effects of oxidative stress and antioxidative factors. Biochem. Pharmacol. 53, 363–372 (1997).
Brodsky, S.V., Gao, S., Li, H. & Goligorsky, M.S. Hyperglycemic switch from mitochondrial nitric oxide to superoxide production in endothelial cells. Am. J. Physiol. Heart Circ. Physiol. 283, H2130–H2139 (2002).
Chang, S.H., Garcia, J., Melendez, J.A., Kilberg, M.S. & Agarwal, A. Haem oxygenase 1 gene induction by glucose deprivation is mediated by reactive oxygen species via the mitochondrial electron-transport chain. Biochem. J. 371, 877–885 (2003).
Mohazzab, H.K., Kaminski, P.M. & Wolin, M.S. Lactate and PO2 modulate superoxide anion production in bovine cardiac myocytes: potential role of NADH oxidase. Circulation 96, 614–620 (1997).
Lu, H., Forbes, R.A. & Verma, A. Hypoxia-inducible factor 1 activation by aerobic glycolysis implicates the Warburg effect in carcinogenesis. J. Biol. Chem. 277, 23111–23115 (2002).
Ohman, T., Parish, G. & Jackson, R.M. Hypoxic modulation of manganese superoxide dismutase promoter activity and gene expression in lung epithelial cells. Am. J. Respir. Cell Mol. Biol. 21, 119–127 (1999).
St Clair, D.K., Porntadavity, S., Xu, Y. & Kiningham, K. Transcription regulation of human manganese superoxide dismutase gene. Methods Enzymol. 349, 306–312 (2002).
Dalton, T.P., Shertzer, H.G. & Puga, A. Regulation of gene expression by reactive oxygen. Annu. Rev. Pharmacol. Toxicol. 39, 67–101 (1999).
Taylor, B.L. & Zhulin, I.B. PAS domains: internal sensors of oxygen, redox potential, and light. Microbio. Mol. Biol. Rev. 63, 479–506 (1999).
Bunn, H.F. & Poyton, R.O. Oxygen sensing and molecular adaptation to hypoxia. Physiol. Rev. 76, 839–885 (1996).
Acknowledgements
We thank A. Das, L. Wang and V. Jones for technical assistance; the University of Texas Southwestern Medical Center Molecular Pathology Core group for histologic expertise; D. Bellotto and the University of Texas Southwestern Medical Center Imaging Core for guidance; R. Estabrook, R. Haller and G. Thomas for providing suggestions with mitochondrial assays; J. Repa for assistance with real-time RT-PCR; J. Herz and S. Hoffman for donating antibodies; D. Foster, R. S. Williams and S. McKnight for support; and A. Zinn, L. Terada and C. K. Garcia for critically reviewing this manuscript. J.A.G. is supported by funds from the National Institutes of Health, American Heart Association and Donald W. Reynolds Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Scortegagna, M., Ding, K., Oktay, Y. et al. Multiple organ pathology, metabolic abnormalities and impaired homeostasis of reactive oxygen species in Epas1−/− mice. Nat Genet 35, 331–340 (2003). https://doi.org/10.1038/ng1266
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1266
This article is cited by
-
EPAS1 expression contributes to maintenance of the primordial follicle pool in the mouse ovary
Scientific Reports (2024)
-
Pericytes contribute to pulmonary vascular remodeling via HIF2α signaling
EMBO Reports (2024)
-
Dysregulation of hypoxia-inducible factor 1α in the sympathetic nervous system accelerates diabetic cardiomyopathy
Cardiovascular Diabetology (2023)
-
UBE3B promotes breast cancer progression by antagonizing HIF-2α degradation
Oncogene (2023)
-
Hypoxia-induced signaling in the cardiovascular system: pathogenesis and therapeutic targets
Signal Transduction and Targeted Therapy (2023)