Abstract
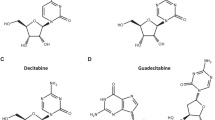
DNA methyltransferase (DNMT) inhibitors, azacitidine (Vidaza®, Pharmion, Boulder, CO, USA) and decitabine (Dacogen™; SuperGen Inc, Dublin, CA, USA, and MGI Pharma Inc, Bloomington, MN, USA), have had a significant impact on the treatment paradigm of myelodysplastic syndromes (MDSs), previously managed mainly by supportive care and hematopoietic-stem-cell transplantation. The positive clinical experience seen in MDS to date coupled with the persistent challenges faced in the treatment of other hematologic malignancies has served as the impetus for further exploration of the therapeutic value of DNMT inhibitors beyond MDS. In that respect, the majority of data for these agents are in the setting of acute myelogenous leukemia (AML). Experience with these agents in patients with refractory anemia with excess blasts in transformation (reclassified by the World Health Organization as AML) was also reported in the clinical trials submitted to the FDA for approval of azacitidine for MDS. Some use has also been described in chronic myelogenous leukemia and acute lymphocytic leukemia. Further studies are needed to clarify the appropriate dose and the number and duration of cycles in the treatment of leukemias, and to identify ideal candidates for therapy, explore the role of DNMT inhibitors in combination with other agents, especially histone deacetylase inhibitors, delineate differences between the commercially available agents, and establish the long-term safety of these agents. To this end, experience with DNMT inhibitors in hematologic malignancies other than MDS is reviewed in an effort to better understand the therapeutic potential of these agents and to define areas of future exploration in these settings.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Piskala A et al. (1964) Nucleic acids components and their analogues. LI. Synthesis of 1-glycosyl derivatives of 5-azauracil and 5-azacytosine. Collect Czech Chem Commun 29: 2060–2076
Hanka LJ et al. (1966) Microbiological production of 5-azacytidine. I. Production and biological activity. Antimicrob Agents Chemother 6: 619–624
Silverman LR et al. (1993) Effects of treatment with 5-azacytidine on the in vivo and in vitro hematopoiesis in patients with myelodysplastic syndromes. Leukemia 7 (Suppl 1): 21–29
Silverman LR et al. (2002) Randomized controlled trial of azacitidine in patients with myelodysplastic syndrome: a study of the Cancer and Leukemia Group B. J Clin Oncol 20: 2429–2440
Kornblith AB et al. (2002) Impact of azacytidine on the quality of life of patients with myelodysplastic syndrome treated in a randomized phase III trial: a Cancer and Leukemia Group B study. J Clin Oncol 20: 2441–2452
Pharmion Corporation. (2004) Vidaza® (azacitidine, injectable, subcutaneous) package insert; Pharmion, Boulder, CO, USA.
Jones PA and Laird PW (1999) Cancer epigenetics comes of age. Nat Genet 21: 163–167
Behringer B et al. (2003) Prognosis of older patients with acute myeloid leukemia receiving either inducion or noncurative treatment: a single-center retrospective study. Ann Hematol 82: 381–389
Leith CP et al. (1997) Acute myeloid leukemia in the elderly: assessment of multi-drug resistance (MDR) and cytogenetics distinguishes biological subgroups with remarkably distinct responses to standard chemotherapy: a Southwest oncology group study. Blood 89: 3323–3329
Stone RM et al. Acute myeloid leukemia. In American Society of Hematology Education Program Book: 2004 December 4–7; San Diego, 98–117 (Eds Broudy VC et al.) Washington, DC: American Society of Hematology
Deininger M et al. (2005) The develoment of imatinib as a therapeutic agent for chronic myeloid leukemia. Blood 105: 2640–2653
Hochhaus A et al. (2004) Clinical resistance to imatinib: mechanisms and implications. Hematol Oncol Clin N Am 18: 641–656
Lee SJ et al. (1998) Initial therapy for chronic myelogenous leukemia: playing the odds. J Clin Oncol 16: 2897–2903
Ghaddar HM et al. (1994) Cytogenetic evolution following the transformation of myelodysplastic syndrome to acute myelogenous leukemia: implications on the overlap between the two disease states. Leukemia 8: 1649–1653
Baylin SB and Herman JG (2000) DNA hypermethylation in tumorigenesis. Trends Genet 16: 168–174
Robertson KD (2001) DNA methylation, methyltransferases, and cancer. Oncogene 20: 3139–3155
Costello JF and Plass C (2001) Methylation matters. J Med Genet 38: 285–303
Bestor T et al. (1988) Cloning and sequencing of a cDNA encoding DNA methyltransferase of mouse cells. The carboxyl-terminal domain of the mammalian enzymes is related to bacterial restriction methyltransferases. J Mol Biol 203: 971–983
Okano M et al. (1999) DNA methyltransferases Dnmt3a and Dnmt3b are essential for de novo methylation and mammalian development. Cell 99: 247–257
Issa J-P et al. (1997) DNA methylation changes in hematologic malignancies: biologic and clinical implications. Leukemia 11 (Suppl 1): 7–11
Melki JR et al. (1999) Concurrent DNA hypermethylation of multiple genes in acute myeloid leukemia. Cancer Res 59: 3730–3740
Herman JG et al. (1996) Hypermethylation-associated inactivation indicates a tumor suppressor role for p15INK4B. Cancer Res 56: 722–777
Nguyen TT et al. (2000) Quantitative measure of c-abl and p15 methylation in chronic myelogenous leukemia: biological implications. Blood 95: 2990–2992
Baylin SB et al. (1987) Hypermethylation of the 5' region of the calcitonin gene is a property of human lymphoid and acute myeloid malignancies. Blood 70: 412–417
Malinen T et al. (1991) Acceleration of chronic myeloid leukemia correlates with calcitonin gene hypermethylation. Blood 77: 2435–2440
Roman-Gomez J et al. (2002) 5' CpG Island hypermethylation is associated with transcription silencing of the p21 CIPI/WAFI/SDI1 gene and confers poor prognosis in acute lymphobalstic leukemia. Blood 99: 2291–2296
Christman JK (2002) 5-Azacytidine and 5-aza-2'-deoxycytidine as inhibitors of DNA methylation: mechanistic studies and their implications for cancer therapy. Oncogene 21: 5483–5495
Von Hoff DD and Slavik M (1977) 5-azacytidine—a new anticancer drug with significant activity in acute myeloblastic leukemia. Adv Pharmacol Chemother 14: 285–326
McCredie KB et al. (1973) Treatment of acute leukemia with 5-azacytidine (NSC-102816). Cancer Chemother Rep 57: 319–323
Tan C et al. (1973) Clinical trial of 5-azacytidine (5-azaCR) [abstract]. Cancer Res 14: 97
Karon M et al. (1973) 5-Azacytidine: a new active agent for the treatment of acute leukemia. Blood 42: 359–365
Von Hoff DD et al. (1976) 5-Azacytidine. A new anticancer drug with effectiveness in acute myelogenous leukemia. Ann Intern Med 85: 237–245
Vogler WR et al. (1976) 5-Azacytidine (NSC 102816): a new drug for the treatment of myeloblastic leukemia. Blood 48: 331–337
Saiki JH et al. (1981) Effect of schedule on activity and toxicity of 5-azacytidine in acute leukemia: a Southwest Oncology Group Study. Cancer 47: 1739–1742
Case DC Jr (1982) 5-azacytidine in refractory acute leukemia. Oncology 39: 218–221
Winton EF et al. (1985) Sequentially administered 5-azacitidine and amsacrine in refractory adult acute leukemia: a phase I-II trial of the Southeastern Cancer Study Group. Cancer Treat Rep 69: 807–811
Hakami N et al. (1987) Combined etoposide and 5-azacitidine in children and adolescents with refractory or relapsed acute nonlymphocytic leukemia: A Pediatric Oncology Group study. J Clin Oncol 5: 1022–1025
Lee EJ et al. (1990) Low dose 5-azacytidine is ineffective for remission induction in patients with acute myeloid leukemia. Leukemia 4: 835–838
Goldberg J et al. (1993) Mitoxantrone and 5-azacytidine for refractory/relapsed ANLL or CML in blast crisis: a leukemia intergroup study. Am J Hematol 43: 286–290
Steuber CP et al. (1996) Induction treatment of refractory or recurrent childhood acute myeloid leukemia using amsacrine and etoposide with or without azacitidine: a Pediatric Oncology Group randomized phase II study. J Clin Oncol 14: 1521–1525
Oblon D et al. (1984) Long-term follow up of brief, intensive chemotherapy without maintenance therapy for acute myelogenous leukemia (AML): A SECSG pilot study [abstract]. Proc Am Soc Clin Oncol 20: C-748
Grier HE et al. (1992) Intensive sequential chemotherapy for children with acute myelogenous leukemia: VAPA, 80-035, and HI-C-Daze. Leukemia 6 (Suppl 2): 48–51
Shadduck RK et al. (2004) AML induction therapy with outpatient azacitidine [abstract #1800]. In Meeting Abstracts of the American Society of Hematology: 2004 December 5; San Diego http://www.abstracts2view.com/hem_sandiego2004/view.php?nu=HEM4L1_4764 ] (accessed 28 September 2005); also Blood 104 (Suppl 1): 499a
Schiffer CA et al. (1982) Treatment of the blast crisis of chronic myelogenous leukemia with 5-azacitidine and VP-16-213. Cancer Treat Rep 66: 267–271
Dutcher JP et al. (1992) Phase II study of mitoxantrone and 5-azacytidine for accelerated and blast crisis of chronic myelogenous leukemia: a study of the Eastern Cooperative Oncology Group. Leukemia 6: 770–775
Rivard GE et al. (1981) Phase I study on 5-Aza-2'-deoxycytidine in children with acute leukemia. Leukemia Res 5: 453–462
Momparler RL et al. (1985) Clinical trial on 5-aza-2'-deoxycytidine in patients with acute leukemia. Pharmacol Ther 30: 277–286
Richel DJ et al. (1991) The antileukemic activity of 5-aza-2 deoxycytidine (Aza-dC) in patients with relapsed and resistant leukaemia. Br J Cancer 64: 144–148
Petti AC et al. (1993) Pilot study of 5-aza-2'-deoxycytidine (decitabine) in the treatment of poor prognosis acute myelogenous leukemia patients: preliminary results. Leukemia 7 (Suppl 1): 36–41
Schwartsmann G et al. (1997) Decitabine (5-Aza-2-deoxycytidine; DAC) plus daunorubicin as a first line treatment in patients with acute myeloid leukemia: preliminary observations. Leukemia 11 (Suppl 1): 28–31
Willemze R et al. (1997) A randomized phase II study on the effects of 5-aza-2' deoxycytidine combined with either amsacrine or idarubicin in patients with relapsed acute leukemia: an EORTC Leukemia Cooperative Group phase II study (06893). Leukemia 11 (Suppl 1): 24–27
Kantarjian HM et al. (1997) Results of decitabine therapy in the accelerated and blastic phases of chronic myelogenous leukemia. Leukemia 11: 1617–1620
Sacchi S et al. (1999) Chronic myelogenous leukemia in nonlymphoid blastic phase: analysis of the results of first salvage therapy with three different treatment approaches for 162 patients. Cancer 86: 2632–2641
Wijermans P et al. (2000) Low-dose 5-aza-2' deoxycytidine, a DNA hypomethylating agent, for the treatment of high-risk myelodysplastic syndrome: a multi-center phase II study in elderly patients. J Clin Oncol 18: 956–962
Issa JJ et al. (2004) Phase I study of low-dose prolonged exposure schedules of the hypomethylating agent 5-aza-2' deoxycytidine (decitabine) in hematopoietic malignancies. Blood 103: 1635–1640
Kantarjian HM et al. (2003) Results of decitabine (5-aza-2' deoxycytidine) therapy in 130 patients with chronic myelogenous leukemia. Cancer 98: 522–528
de Lima M et al. (2003) Long-term follow-up of a phase I study of high-dose decitabine, busulfan, and cyclophosphamide plus allogeneic transplantation for the treatment of patients with leukemias. Cancer 97: 1242–1247
Ravandi F et al. (2001) Decitabine with allogeneic peripheral blood stem cell transplantation in the therapy of leukemia relapse following a prior transplant: results of a phase I study. Bone Marrow Transplant 27: 1221–1225
Kritz AD et al. (1996) Pilot study of 5-azacytidine (5-AZA) and carboplatin (CBDCA) in patients with relapsed/refractory leukemia. Am J Hematol 5: 117–121
Camacho LH et al. (2001) Transcription modulation: a pilot study of sodium phenylbutyrate plus 5-azacytidine [abstract]. Blood 98: 460a
Miller CB et al. (2001) A Phase I dose-descalation trial of combined DNA methyltransferase (MeT)/histone deacetylase (HDAC) inhibition in myeloid malignancies [abstract]. Blood 98: 622a
Silverman LR et al. (1994) Azacitidine in myelodysplastic syndromes: CALGB studies 8421 and 8921 [abstract]. Am Hematol 68: A12
Kaminskas E et al. (2005) Approval summary: azacitidine for treatment of myelodysplastic syndrome subtypes. Clin Cancer Res 11: 3604–3608
Jaffe ES et al. (2001) World Health Organization Classification of Tumours: Pathology and Genetics of Tumours of Haematopoietic and Lymphoid Tissues. Lyon, France: IARC Press
Momparler RL et al. (1997) Pharmacological approach for optimization of the dose schedule of 5-Aza-2'-deoxycytidine (Decitabine) for the therapy of leukemia. Leukemia 11: 175–180
Estey E et al. (1997) Effect of diagnosis (refractory anemia with excess blasts, refractory anemia with excess blasts in transformation, or acute myeloid leukemia [AML]) on outcome of AML-type chemotherapy. Blood 15: 2969–2977
Rossi G et al. (2000) Cytogenetic analogy between myelodysplastic syndrome and acute myeloid leukemia of elderly patients. Leukemia 14: 636–641
Parker JE et al. (2000) The role of apoptosis, proliferation, and the Bcl-2 related proteins in myelodysplastic syndromes and acute myeloid leukemias secondary to MDS. Blood 96: 3932–3938
Vardiman JW et al. (2002) The World Health Organization (WHO) classification of the myeloid neoplasms. Blood 100: 2292–2302
Claus R et al. (2003) Epigenetic targets in hematopoietic malignancies. Oncogene 22: 6489–6496
Chen RZ et al. (1998) DNA hypomethylation leads to elevated mutation rates. Nature 395: 89–93
Sato N et al. (2003) Effects of 5-aza-2'-deoxycytidine on matrix metalloproteinase expression and pancreatic cancer cell invasiveness. J Natl Cancer Inst 95: 327–330
Eden A et al. (2003) Chromosomal instability and tumors promoted by DNA hypomethylation. Science 300: 455
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Prof Pierre Fenaux is a regular consultant of Pharmion Corp.
Rights and permissions
About this article
Cite this article
Fenaux, P. Inhibitors of DNA methylation: beyond myelodysplastic syndromes. Nat Rev Clin Oncol 2 (Suppl 1), S36–S44 (2005). https://doi.org/10.1038/ncponc0351
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncponc0351
This article is cited by
-
Depletion of ZBTB38 potentiates the effects of DNA demethylating agents in cancer cells via CDKN1C mRNA up-regulation
Oncogenesis (2018)
-
Reactivation of HIV-1 from Latency by an Ingenol Derivative from Euphorbia Kansui
Scientific Reports (2017)
-
Enzymatic activation of double-targeted 5′-O-l-valyl-decitabine prodrug by biphenyl hydrolase-like protein and its molecular design basis
Drug Delivery and Translational Research (2017)
-
HIV-1 functional cure: will the dream come true?
BMC Medicine (2015)
-
Progress and challenges in the use of latent HIV-1 reactivating agents
Acta Pharmacologica Sinica (2015)