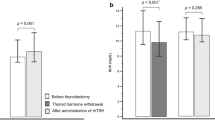
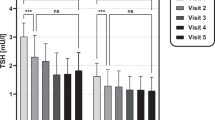
Abstract
Experimental studies and clinical data have demonstrated that thyroid-cell proliferation is dependent on thyroid-stimulating hormone (TSH), thereby providing the rationale for TSH suppression as a treatment for differentiated thyroid cancer. Several reports have shown that hormone-suppressive treatment with the L-enantiomer of tetraiodothyronine (L-T4) benefits high-risk thyroid cancer patients by decreasing progression and recurrence rates, and cancer-related mortality. Evidence suggests, however, that complex regulatory mechanisms (including both TSH-dependent and TSH-independent pathways) are involved in thyroid-cell regulation. Indeed, no significant improvement has been obtained by suppressing TSH in patients with low-risk thyroid cancer. Moreover, TSH suppression implies a state of subclinical thyrotoxicosis. In low-risk patients, the goal of L-T4 treatment is therefore to obtain a TSH level in the normal range (0.5–2.5 mU/l). Only selected patients with high-risk papillary and follicular thyroid cancer require long-term TSH-suppressive doses of L-T4. In these patients, careful monitoring is necessary to avoid undesirable effects on bone and heart.
Key Points
-
Thyroid-cell proliferation is thyroid-stimulating hormone (TSH)-dependent, hence L-tetraiodothyronine (L-T4)-induced TSH suppression should be included in the treatment strategies for differentiated thyroid carcinomas
-
TSH suppression implies a state of subclinical thyrotoxicosis and becomes necessary only when there is evidence of persistent or recurrent disease; in low-risk patients, L-T4 treatment serves to return TSH level to within the normal range
-
To ensure optimal dosing, each patient must always receive the same preparation and the daily L-T4 dose should be carefully tailored
-
Adjustments of L-T4 dosage will be required under particular circumstances, for example in pregnant women, patients with significant weight gain or weight loss, those with known heart disease and older patients
-
If long-term TSH suppression is necessary because of a high-risk cancer, a cardioselective β-blocking drug can be added to reduce cardiovascular risk and to improve quality of life
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Larsen PR et al. (2002) Thyroid physiology and diagnostic evaluation of patients with thyroid disorders. In Williams' Textbook of Endocrinology, edn 10, 331–373 (Eds Larsen PR et al.) Philadelphia: WB Saunders
Larsen PR et al. (1981) Relationships between circulating and intracellular thyroid hormones: physiological and clinical implications. Endocr Rev 2: 87–102
Saberi M and Utiger RD (1974) Serum thyroid hormone and thyrotropin concentrations during thyroxine and triiodothyronine therapy. J Clin Endocrinol Metab 39: 923–927
Braverman LE et al. (1970) Conversion of thyroxine (T4) to triiodothyronine in athyreotic human subjects. J Clin Invest 49: 855–864
Mandel SJ et al. (1993) Levothyroxine therapy in patients with thyroid disease. Ann Intern Med 119: 492–502
Singer PA et al. (1996) Treatment guidelines for patients with thyroid nodules and well-differentiated thyroid cancer. Arch Intern Med 156: 2165–2172
Thyroid Carcinoma Task Force (2001) AACE/AAES medical/surgical guidelines for clinical practice: management of thyroid carcinoma. Endocr Pract 7: 202–220
Dunhill TP (1937) Surgery of the thyroid gland (The Lettsomian Lectures). BMJ 1: 460–461
Balme HW (1954) Metastastic carcinoma of the thyroid successfully treated with thyroxine. Lancet 266: 812–813
Crile G Jr (1966) Endocrine dependency of papillary carcinomas of the thyroid. JAMA 195: 721–724
Goldberg LD and Ditchek NT (1981) Thyroid carcinoma with spinal cord compression. JAMA 245: 953–954
Mazzaferri EL and Jhiang SM (1994) Long-term impact of initial surgical and medical therapy on papillary and follicular thyroid cancer. Am J Med 97: 418–428
Pujol P et al. (1996) Degree of thyrotropin suppression as a prognostic determinant in differentiated thyroid cancer. J Clin Endocrinol Metab 81: 4318–4322
Cooper DS et al. (1998) Thyrotropin suppression and disease progression in patients with differentiated thyroid cancer: results from the National Thyroid Cancer Treatment Cooperative Registry. Thyroid 8: 737–744
Roger P et al. (1988) Mitogenic effects of thyrotropin and adenosine 3',5'-monophosphate in differentiated normal human thyroid cells in vitro. J Clin Endocrinol Metab 66: 1158–1165
Nadler NJ et al. (1970) The effect of hypophysectomy on the experimental production of rat thyroid neoplasms. Cancer Res 30: 1909–1911
Ichikawa Y et al. (1976) Presence of TSH receptor in thyroid neoplasms. J Clin Endocrinol Metab 42: 395–398
Carayon P et al. (1980) Human thyroid cancer: membrane thyrotropin binding and adenylate cyclase activity. J Clin Endocrinol Metab 51: 915–920
Clark OH et al. (1983) Characterization of the thyrotropin receptor–adenylate cyclase system in neoplastic human thyroid tissue. J Clin Endocrinol Metab 57: 140–147
Tanaka K et al. (1997). Relationship between prognostic score and thyrotropin receptor (TSH-R) in papillary thyroid carcinoma: immunohistochemical detection of TSH-R. Br J Cancer 76: 594–599
Lazar V et al. (1999) Expression of the Na+/I-symporter gene in human thyroid tumors: a comparison study with other thyroid-specific genes. J Clin Endocrinol Metab 84: 3228–3234
Filetti S et al. (1999) Sodium/iodide symporter a key transport system in thyroid cancer cell metabolism. Eur J Endocrinol 141: 443–457
Filetti S et al. (1988) The role of thyroid-stimulating antibodies of Graves' disease in differentiated thyroid cancer. N Engl J Med 318: 753–759
Belfiore A et al. (2001) Graves' disease, thyroid nodules and thyroid cancer. Clin Endocrinol (Oxf) 55: 711–718
Westermark K et al. (1983) Epidermal growth factor modulates thyroid growth and function in culture. Endocrinology 112: 1680–1686
Wynford-Thomas D (1993) Molecular basis of epithelial tumorigenesis: the thyroid model. Crit Rev Oncol 4: 1–23
Fagin JA (2004) How thyroid tumors start and why it matters: kinase mutants as targets for solid cancer pharmacotherapy. J Endocrinol 183: 249–256
Russo D et al. (1995) Activating mutations of the TSH receptor in differentiated thyroid carcinomas. Oncogene 11: 1907–1911
Spambalg D et al. (1996) Structural studies of the thyrotropin receptor and Gs alpha in human thyroid cancer: low prevalence of mutation predicts infrequent involvement in malignant transformation. J Clin Endocrinol Metab 81: 3898–3901
Challeton C et al. (1995) Pattern of ras and gsp oncogene mutations in radiation-associated human thyroid tumors. Oncogene 11: 601–603
Nicoloff JT and Spencer CA (1992) Non-thyrotropin-dependent thyroid secretion. J Clin Endocrinol Metab 75: 343
Burmeister LA et al. (1992) Levothyroxine dose requirements for thyrotropin suppression in the treatment of differentiated thyroid cancer. J Clin Endocrinol Metab 75: 344–350
Demers LM and Spencer CA (2003) Laboratory medicine practice guidelines. Laboratory support for the diagnosis and monitoring of thyroid disease. Thyroid 13: 33–44
Surks M et al. (2004) Subclinical thyroid disease: scientific review and guidelines for diagnosis and management. JAMA 291: 228–238
Hollowell JG et al. (2002) Serum TSH, T4, and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab 87: 489–499
Bjoro T et al. (2000) Prevalence of thyroid disease, thyroid dysfunction, and thyroid peroxidase antibodies in a large unselected population. Eur J Endocrinol 143: 639–637
Parle JV et al. (2001) Prediction of all-cause and cardiovascular mortality in elderly people from one low serum thyrotropin result: a 10-year cohort study. Lancet 358: 861–865
Biondi B et al. (2005) Subclinical hyperthyroidism: clinical features and treatment options. Eur J Endocrinol 152: 1–9
Spencer CA et al. (1986) Thyrotropin secretion in thyrotoxic and thyroxine treated patients: assessment by a sensitive immunoenzymometric assay. J Clin Endocrinol Metab 63: 349–355
Cailleux AF et al. (2000) Is diagnostic iodine-131 scanning useful after total thyroid ablation for differentiated thyroid cancer? J Clin Endocrinol Metab 85: 175–178
Pacini F et al. (2001) Prediction of disease status by recombinant human TSH-stimulated serum Tg in the postsurgical follow-up of differentiated thyroid carcinoma. J Clin Endocrinol Metab 86: 5686–5690
American Thyroid Association; Endocrine Society; American Association of Clinical Endocrinologists (2004) Joint statement on the U.S. Food and Drug Administration's decision regarding bioequivalence of levothyroxine sodium. Thyroid 14: 486
Hays MT (1991) Localization of human thyroxine absorption. Thyroid 1: 242–248
Ladenson PW (2003) Problems in the management of hypothyroidism. In Diseases of the Thyroid, edn 2, 161–176 (Ed. Braverman LE) Totowa: Humana Press
Arafah BM (2001) Increased need for thyroxine in women with hypothyroidism during estrogen therapy. N Engl J Med 344: 1743–1749
Howanitz PJ et al. (1982) Incidence and mechanism of spurious increase in serum thyrotropin. Clin Chem 28: 427–431
Sherman SI et al. (1997) Augmented hepatic and skeletal thyromimetic effects of tiratricol in comparison with levothyroxine. J Clin Endocrinol Metab 82: 2153–2158
Mechelany C et al. (1991) TRIAC (3,5,3'-triiodothyroacetic acid ) has parallel effects at pituitary and peripheral tissue levels in thyroid cancer patients treated with L-thyroxine. Clin Endocrinol 35: 123–128
Goldman JM et al. (1980) Influence of triiodothyronine withdrawal time on 131I uptake post-thyroidectomy for thyroid cancer. J Clin Endocrinol Metab 50: 734–739
Bunevicius R et al. (1999) Effects of thyroxine as compared with thyroxine plus triiodothyronine in patients with hypothyroidism. N Engl J Med 340: 424–429
Sawka AM et al. (2003) Does a combination regimen of thyroxine (T4) and 3,5,3'-triiodothyronine improve depressive symptoms better than T4 alone in patients with hypothyroidism? Results of a double blind randomized controlled trial. J Clin Endocrinol Metab 88: 4551–4555
Walsh JP et al. (2003) Combined thyroxine/liothyronine treatment does not improve well-being, quality of life, or cognitive function compared to thyroxine alone: a randomized controlled trial in patients with primary hypothyroidism. J Clin Endocrinol Metab 88: 4543–4550
Clyde PW et al. (2003) Combined levothyroxine plus liothyronine compared with levothyroxine alone in primary hypothyroidism. A randomized controlled trial. JAMA 290: 2952–2958
Henneman G et al. (2004) Thyroxine plus low-dose, slow release triiodothyronine replacement in hypothyroidism: proof of principle. Thyroid 14: 271–275
Saravan P et al. (2005) Partial substitution of thyroxine (T4) with triiodothyronine in patients on T4 replacement therapy: results of a large community-based randomized controlled trial. J Clin Endocrinol Metab 90: 805–812
Appelhof BC et al. (2005) Combined therapy with levothyroxine and liothyronine in two ratios, compared with levothyroxine monotherapy in primary hypothyroidism: a double-blind, randomized, controlled clinical trial. J Clin Endocrinol Metab 90: 2666–2674
Escobar-Morreale HF et al. (2005) Thyroid hormone replacement therapy in primary hypothyroidism: a randomized trial comparing L-thyroxine plus liothyronine with L-thyroxine alone. Ann Intern Med 142: 412–424
Santini F et al. (2005) Lean body mass is a major determinant of levothyroxine dosage in the treatment of thyroid diseases. J Clin Endocrinol Metab 90: 124–127
Sawin CT et al. (1989) The aging thyroid. The use of thyroid hormone in older person. JAMA 261: 2653–2655
Alexander EK et al. (2004) Timing and magnitude of increases in levothyroxine requirements during pregnancy in women with hypothyroidism. N Engl J Med 351: 241–249
Andersen S et al. (2002) Narrow individual variations in serum T4 and T3 in normal subjects: a clue to understanding of subclinical thyroid disease. J Clin Endocrinol Metab 87: 1068–1072
Sheppard MC et al. (2002) Levothyroxine treatment and occurrence of fracture of the hip. Arch Intern Med 162: 338–343
Bauer DC et al. (2001) Risk for fracture in women with low serum levels of thyroid-stimulating hormone. Ann Intern Med 134: 561–568
Marcocci C et al. (1994) Carefully monitored levothyroxine therapy is not associated with bone loss in premenopausal women. J Clin Endocrinol Metab 78: 818–823
Biondi B et al. (1993) Cardiac effects of long-term thyrotropin-suppressive therapy with levothyroxine. J Clin Endocrinol Metab 77: 334–338
Sawin CT et al. (1994) Low serum thyrotropin concentrations as a risk factor for atrial fibrillation in older persons. N Engl J Med 331: 1249–1252
Biondi B et al. (2002) Effects of subclinical thyroid dysfunction on the heart. Ann Intern Med 137: 904–914
Biondi B et al. (1994) Control of adrenergic overactivity by β-blockade improves quality of life in patients receiving long term suppressive therapy with levothyroxine. J Clin Endocrinol Metab 78: 1028–1033
Fazio S et al. (1992) Evaluation by noninvasive methods of the effects of acute loss of thyroid hormone on the heart. Angiology 43: 287–293
Bengel FM et al. (2000) Effect of thyroid hormones on cardiac function, geometry, and oxidative metabolism assessed noninvasively by positron emission tomography and magnetic resonance imaging. J Clin Endocrinol Metab 85: 1822–1827
Biondi B et al. (2003) Cardiovascular safety of acute recombinant human thyrotropin administration to patients monitored for differentiated thyroid cancer. J Clin Endocrinol Metab 88: 211–214
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Biondi, B., Filetti, S. & Schlumberger, M. Thyroid-hormone therapy and thyroid cancer: a reassessment. Nat Rev Endocrinol 1, 32–40 (2005). https://doi.org/10.1038/ncpendmet0020
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpendmet0020
This article is cited by
-
CACA guidelines for holistic integrative management of thyroid cancer
Holistic Integrative Oncology (2022)
-
Polyethylene glycol triggers the anti-cancer impact of curcumin nanoparticles in sw-1736 thyroid cancer cells
Journal of Materials Science: Materials in Medicine (2021)
-
Thyroid hormone therapy in differentiated thyroid cancer
Endocrine (2019)
-
Diagnosis and Management of Hypothyroidism: Addressing the Knowledge–Action Gaps
Advances in Therapy (2018)