Abstract
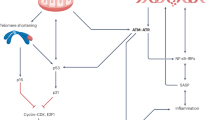
Cellular senescence suppresses cancer by stably arresting the proliferation of damaged cells1. Paradoxically, senescent cells also secrete factors that alter tissue microenvironments2. The pathways regulating this secretion are unknown. We show that damaged human cells develop persistent chromatin lesions bearing hallmarks of DNA double-strand breaks (DSBs), which initiate increased secretion of inflammatory cytokines such as interleukin-6 (IL-6). Cytokine secretion occurred only after establishment of persistent DNA damage signalling, usually associated with senescence, not after transient DNA damage responses (DDRs). Initiation and maintenance of this cytokine response required the DDR proteins ATM, NBS1 and CHK2, but not the cell-cycle arrest enforcers p53 and pRb. ATM was also essential for IL-6 secretion during oncogene-induced senescence and by damaged cells that bypass senescence. Furthermore, DDR activity and IL-6 were elevated in human cancers, and ATM-depletion suppressed the ability of senescent cells to stimulate IL-6-dependent cancer cell invasiveness. Thus, in addition to orchestrating cell-cycle checkpoints and DNA repair, a new and important role of the DDR is to allow damaged cells to communicate their compromised state to the surrounding tissue.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
24 August 2009
In the version of this article initially published, the labelling of the radiation doses in Fig. 1a was reversed. These errors have been corrected in the HTML and PDF versions of the article.
References
Beausejour, C. M. et al. Reversal of human cellular senescence: roles of the p53 and p16 pathways. EMBO J. 22, 4212–4222 (2003).
Campisi, J. Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell 120, 513–522 (2005).
Krtolica, A., Parrinello, S., Lockett, S., Desprez, P. Y. & Campisi, J. Senescent fibroblasts promote epithelial cell growth and tumorigenesis: a link between cancer and aging. Proc. Natl Acad. Sci. USA 98, 12072–12077 (2001).
Liu, D. & Hornsby, P. J. Senescent human fibroblasts increase the early growth of xenograft tumors via matrix metalloproteinase secretion. Cancer Res. 67, 3117–3126 (2007).
Bavik, C. et al. The gene expression program of prostate fibroblast senescence modulates neoplastic epithelial cell proliferation through paracrine mechanisms. Cancer Res. 66, 794–802 (2006).
Coppe, J. P. et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 6, 2853–2868 (2008).
Parrinello, S., Coppe, J. P., Krtolica, A. & Campisi, J. Stromal-epithelial interactions in aging and cancer: senescent fibroblasts alter epithelial cell differentiation. J. Cell Sci. 118, 485–496 (2005).
Coppe, J. P., Kauser, K., Campisi, J. & Beausejour, C. M. Secretion of vascular endothelial growth factor by primary human fibroblasts at senescence. J. Biol. Chem. 281, 29568–29574 (2006).
Wajapeyee, N., Serra, R. W., Zhu, X., Mahalingam, M. & Green, M. R. Oncogenic BRAF induces senescence and apoptosis through pathways mediated by the secreted protein IGFBP7. Cell 132, 363–374 (2008).
Kortlever, R. M., Higgins, P. J. & Bernards, R. Plasminogen activator inhibitor-1 is a critical downstream target of p53 in the induction of replicative senescence. Nature Cell Biol. 8, 877–884 (2006).
Kuilman, T. et al. Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell 133, 1019–1031 (2008).
Acosta, J. C. et al. Chemokine signaling via the CXCR2 receptor reinforces senescence. Cell 133, 1006–1018 (2008).
Caruso, C., Lio, D., Cavallone, L. & Franceschi, C. Aging, longevity, inflammation, and cancer. Ann. NY Acad. Sci. 1028, 1–13 (2004).
Campisi, J. & d'Adda di Fagagna, F. Cellular senescence: when bad things happen to good cells. Nature Rev. Mol. Cell Biol. 8, 729–740 (2007).
Rodier, F., Kim, S. H., Nijjar, T., Yaswen, P. & Campisi, J. Cancer and aging: the importance of telomeres in genome maintenance. Int. J. Biochem. Cell Biol. 37, 977–990 (2005).
d'Adda di Fagagna, F. et al. A DNA damage checkpoint response in telomere-initiated senescence. Nature 426, 194–198 (2003).
Beliveau, A. et al. p53-dependent integration of telomere and growth factor deprivation signals. Proc. Natl Acad. Sci. USA 104, 4431–4436 (2007).
Gorgoulis, V. G. et al. Activation of the DNA damage checkpoint and genomic instability in human precancerous lesions. Nature 434, 907–913 (2005).
Bartkova, J. et al. DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 434, 864–870 (2005).
Xu, Y. & Baltimore, D. Dual roles of ATM in the cellular response to radiation and in cell growth control. Genes Dev. 10, 2401–2410 (1996).
Mallette, F. A., Gaumont-Leclerc, M. F. & Ferbeyre, G. The DNA damage signaling pathway is a critical mediator of oncogene-induced senescence. Genes Dev. 21, 43–48 (2007).
Di Micco, R. et al. Oncogene-induced senescence is a DNA damage response triggered by DNA hyper-replication. Nature 444, 638–642 (2006).
Bartkova, J. et al. Oncogene-induced senescence is part of the tumorigenesis barrier imposed by DNA damage checkpoints. Nature 444, 633–637 (2006).
Rothkamm, K. & Lobrich, M. Evidence for a lack of DNA double-strand break repair in human cells exposed to very low x-ray doses. Proc. Natl Acad. Sci. USA 100, 5057–5062 (2003).
Sedelnikova, O. A. et al. Senescing human cells and ageing mice accumulate DNA lesions with unrepairable double-strand breaks. Nature Cell Biol. 6, 168–170 (2004).
Herbig, U., Ferreira, M., Condel, L., Carey, D. & Sedivy, J. M. Cellular senescence in aging primates. Science 311, 1257 (2006).
Rodier, F., Campisi, J. & Bhaumik, D. Two faces of p53: aging and tumor suppression. Nucleic Acids Res. 35, 7475–7484 (2007).
Hayflick, L. The limited in vitro lifetime of human diploid cell strains. Exp. Cell Res. 37, 614–636 (1965).
Xue, W. et al. Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature 445, 656–660 (2007).
Krizhanovsky, V. et al. Senescence of activated stellate cells limits liver fibrosis. Cell 134, 657–667 (2008).
Kim, S. H. et al. TIN2 mediates the functions of TRF2 at telomeres. J. Biol. Chem. 279, 43799–43804 (2004).
Acknowledgements
We thank C. Beausejour (grant CIHR # MPO-79317) for help in the design of a modified lentiviral expression system, R. Teachenor, V. Chu and G. Tang for valuable technical assistance, and P. Desprez for insightful comments on the manuscript. This work was supported by National Institutes of Health grants AG017242 (J.C.) and AG025708 (Buck Institute), a CABCRP grant 11IB-0153 (A.R.D.), a Larry L. Hillblom Foundation fellowship (C.K.P.), the Netherlands Organization for International Cooperation in Higher Education (Nuffic, HSP-TP 06/78), the Dutch Cancer Society (W.A.M.H.), and the Department of Energy under contract DE-AC03-76SF00098 to the University of California.
Author information
Authors and Affiliations
Contributions
F.R. designed, performed and analysed the experiments; J.P.C. and C.K.P. performed antibody arrays; J.P.C. and W.A.M.H. performed ELISAs; S.R.R. analysed immunofluorescence; A.F. analysed tissue arrays, A.R.D.; DPM and E.C. generated and tested p53 and ATM RNAi constructs; J.C. analysed data and F.R. and J.C. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1446 kb)
Rights and permissions
About this article
Cite this article
Rodier, F., Coppé, JP., Patil, C. et al. Persistent DNA damage signalling triggers senescence-associated inflammatory cytokine secretion. Nat Cell Biol 11, 973–979 (2009). https://doi.org/10.1038/ncb1909
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1909
This article is cited by
-
Modulation of fracture healing by senescence-associated secretory phenotype (SASP): a narrative review of the current literature
European Journal of Medical Research (2024)
-
Blocking methionine catabolism induces senescence and confers vulnerability to GSK3 inhibition in liver cancer
Nature Cancer (2024)
-
Judith Campisi (1948–2024)
Nature Aging (2024)
-
Cancer-associated fibroblasts in radiotherapy: Bystanders or protagonists?
Cell Communication and Signaling (2023)
-
Enhancement of PD-L1-attenuated CAR-T cell function through breast cancer-associated fibroblasts-derived IL-6 signaling via STAT3/AKT pathways
Breast Cancer Research (2023)