Abstract
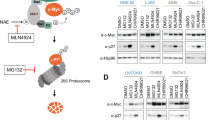
The stability of c-Myc is regulated by multiple Ras effector pathways. Phosphorylation at Ser 62 stabilizes c-Myc, whereas subsequent phosphorylation at Thr 58 is required for its degradation. Here we show that Ser 62 is dephosphorylated by protein phosphatase 2A (PP2A) before ubiquitination of c-Myc, and that PP2A activity is regulated by the Pin1 prolyl isomerase. Furthermore, the absence of Pin1 or inhibition of PP2A stabilizes c-Myc. A stable c-MycT58A mutant that cannot bind Pin1 or be dephosphorylated by PP2A replaces SV40 small T antigen in human cell transformation and tumorigenesis assays. Therefore, small T antigen, which inactivates PP2A, exerts its oncogenic potential by preventing dephosphorylation of c-Myc, resulting in c-Myc stabilization. Thus, Ras-dependent signalling cascades ensure transient and self-limiting accumulation of c-Myc, disruption of which contributes to human cell oncogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Charron, J. et al. Embryonic lethality in mice homozygous for a targeted disruption of the N-myc gene. Genes Dev. 6, 2248–2257 (1992).
Davis, A.C., Wims, M., Spotts, G.D., Hann, S.R. & Bradley, A. A null c-myc mutation causes lethality before 10.5 days of gestation in homozygotes and reduced fertility in heterozygous female mice. Genes Dev. 7, 671–682 (1993).
de Alboran, I.M. et al. Analysis of C-MYC function in normal cells via conditional gene-targeted mutation. Immunity 14, 45–55 (2001).
Trumpp, A. et al. c-Myc regulates mammalian body size by controlling cell number but not cell size. Nature 414, 766 (2001).
Coppola, J.A. & Cole, M.D. Constitutive c-myc oncogene expression blocks mouse erythroleukaemia cell differentiation but not commitment. Nature 320, 760–763 (1986).
Evan, G.L. et al. Induction of apoptosis in fibroblasts by c-myc protein. Cell 69, 119–128 (1992).
Cole, M.D. The myc oncogene: its role in transformation and differentiation. Ann. Rev. Genet. 20, 361–384 (1986).
Nesbit, C.E., Tersak, J.M. & Prochownik, E.V. MYC oncogenes and human neoplastic disease. Oncogene 18, 3004–3016 (1999).
Spencer, C.A. & Groudine, M. Control of c-myc regulation in normal and neoplastic cells. Adv. Canc. Res. 56, 1–48 (1991).
Sears, R., Leone, G., DeGregori, J. & Nevins, J.R. Ras enhances Myc protein stability. Mol.Cell 3, 169–179 (1999).
Sears, R. et al. Multiple Ras-dependent phosphorylation pathways regulate Myc protein stability. Genes Dev. 14, 2501–2514 (2000).
Pulverer, B.J. et al. Site-specific modulation of c-Myc cotransformation by residues phosphorylated in vivo. Oncogene 9, 59–70 (1994).
Henriksson, M., Bakardjiev, A., Klein, G. & Luscher, B. Phosphorylation sites mapping in the N-terminal domain of c-Myc modulate its transforming potential. Oncogene 8, 3199–3209 (1993).
Lutterbach, B. & Hann, S.R. Hierarchical phosphorylation at N-terminal transformation-sensitive sites in c-Myc protein is regulated by mitogens and in mitosis. Mol. Cell. Biol. 14, 5510–5522 (1994).
Cross, D.A.E., Alessi, D.R., Cohen, P., Andejelkovich, M. & Hemmings, B.A. Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature 378, 785–789 (1995).
Salghetti, S.E., Kim, S.Y. & Tansey, W.P. Destruction of Myc by ubiquitin-mediated proteolysis: cancer-associated and transforming muations stabilize Myc. EMBO J. 18, 717–726 (1999).
Gregory, M.A. & Hann, S.R. c-Myc proteolysis by the ubiquitin-proteasome pathway: stabilization of c-Myc in Burkitt's lymphoma cells. Mol. Cell. Biol. 20, 2423–2435 (2000).
Zhou, X.Z. et al. Pin1-dependent prolyl isomerization regulates dephosphorylation of Cdc25C and tau proteins. Mol.Cell 6, 873–883 (2000).
Stukenberg, P.T. & Kirschner, M.W. Pin1 acts catalytically to promote a conformational change in Cdc25. Mol. Cell 7, 1071–1083 (2001).
Sontag, E. et al. The interaction of SV40 small tumor antigen with protein phosphatase 2A stimulates the map kinase pathway and induces cell proliferation. Cell 75, 887–897 (1993).
Myers, J.K., Morris, D.P., Greenleaf, A.L. & Oas, T.G. Phosphorylation of RNA polymerase II CTD fragments results in tight binding to the WW domain from the yeast prolyl isomerase Ess1. Biochemistry 40, 8479–8486 (2001).
Lu, P.J., Zhou, X.Z., Shen, M. & Lu, K.P. Function of WW domains as phosphoserine- or phosphothreonine-binding modules. Science 283, 1325–1328 (1999).
Atchison, F.W., Capel, B. & Means, A.R. Pin1 regulates the timing of mammalian primordial germ cell proliferation. Development 130, 3579–3586 (2003).
Sears, R., Ohtani, K. & Nevins, J.R. Identification of positively and negatively acting elements regulating expression of the E2F2 gene in response to cell growth signals. Mol. Cell. Biol. 17, 5227–5235 (1997).
Hahn, W.C. et al. Creation of human tumour cells with defined genetic elements. Nature 400, 464–468 (1999).
Hahn, W.C. et al. Enumeration of the simian virus 40 early region elements necessary for human cell transformation. Mol. Cell. Biol. 22, 2111–2123 (2002).
Papas, T.S. & Lautenberger, J.A. Sequence curiosity in v-myc oncogenes. Nature 318, 237 (1985).
Bhatia, K. et al. Point mutations in the c-Myc transactivation domain are common in Burkitt's lymphoma and mouse plasmacytomas. Nature Genet. 5, 56–61 (1993).
Chang, D.W., Claassen, G.F., Hann, S.R. & Cole, M.D. The c-Myc transactivation domain is a direct modulator of apoptotic versus proliferative signals. 20, 4309–4319 (2000).
Winkler, K.E., Swenson, K.I., Kornbluth, S. & Means, A.R. Requirement of the Prolyl isomerase Pin1 for the replication checkpoint. Science 287, 1644–1647 (2000).
Nevins, J.R., DeGregori, J., Jakoi, L. & Leone, G. Functional analysis of E2F. Meth.. Enzymol. 283, 205–219 (1997).
DeGregori, J., Leone, G., Ohtani, K., Miron, A. & Nevins, J.R. E2F1 accumulation bypasses a G1 arrest resulting from the inhibition of G1 cyclin-dependent kinase activity. Genes Dev. 9, 2873–2887 (1995).
Cook, J.G. et al. Analysis of Cdc6 function in the assembly of mammalian prereplication complexes. Proc. Natl Acad. Sci. USA 99, 1347–1352 (2002).
He, T.-C. et al. A simplified system for generating recombinant adenoviruses. Biochem. Biophys. Res. Comm. 95, 2509–2514 (1998).
Fujimori, F., Takahashi, K., Uchida, C. & Uchida, T. Mice lacking Pin1 develop normally, but are defective in entering cell cycle from G0 arrest. 265, 658–663 (1999).
Robertson, E.J. in Teratocarcinomas and Embryonic Stem Cells: A Practical Approach (ed. Robertson, E.J.) 104–108 (IRL Press, Oxford, 1987).
Hann, S., Dixit, M., Sears, R. & Sealy, L. The alternatively initiated c-Myc proteins differentially regulated transcription through a noncanonical DNA binding site. Genes Dev. 8, 2441–2452 (1994).
Cifone, M.A. & Fidler, I.J. Correlation of patterns of anchorage independent growth with in vivo behavior of cells from a murine fibrosarcoma. Proc. Natl Acad. Sci. USA 77, 1039–1043 (1980).
Acknowledgements
We thank K. Culler for assistance in the preparation of the manuscript. R.S. was the recipient of a Howard Temin Award (CA 86957) that provided partial support for this work. Additional support was provided by start-up funds from Oregon Health & Sciences University and a grant from the NIH (CA100855) to R.S. Additional support came from the Howard Hughes Medical Institute (J.N.) and a grant from the NIH (CA-82845) to A.R.M.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information Figures
Supplementary Information, Fig. S1 (PDF 1154 kb)
Supplementary Information, Fig. S2
Supplementary Information, Fig. S3
Supplementary Information, Fig. S4
Rights and permissions
About this article
Cite this article
Yeh, E., Cunningham, M., Arnold, H. et al. A signalling pathway controlling c-Myc degradation that impacts oncogenic transformation of human cells. Nat Cell Biol 6, 308–318 (2004). https://doi.org/10.1038/ncb1110
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1110
This article is cited by
-
Organelle resolved proteomics uncovers PLA2R1 as a novel cell surface marker required for chordoma growth
Acta Neuropathologica Communications (2024)
-
Calcineurin-mediated dephosphorylation enhances the stability and transactivation of c-Myc
Scientific Reports (2023)
-
SET-PP2A complex as a new therapeutic target in KMT2A (MLL) rearranged AML
Oncogene (2023)
-
Unravelling similarities and differences in the role of circular and linear PVT1 in cancer and human disease
British Journal of Cancer (2022)
-
Prostate-specific oncogene OTUD6A promotes prostatic tumorigenesis via deubiquitinating and stabilizing c-Myc
Cell Death & Differentiation (2022)