Abstract
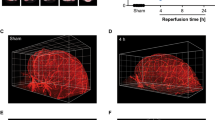
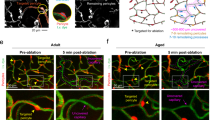
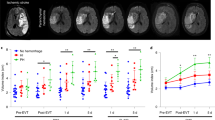
Cerebral microvascular occlusion is a common phenomenon throughout life1,2 that might require greater recognition as a mechanism of brain pathology. Failure to recanalize microvessels promptly may lead to the disruption of brain circuits and significant functional deficits3. Haemodynamic forces and the fibrinolytic system4 are considered to be the principal mechanisms responsible for recanalization of occluded cerebral capillaries and terminal arterioles. Here we identify a previously unrecognized cellular mechanism that may also be critical for this recanalization. By using high-resolution fixed-tissue microscopy and two-photon imaging in living mice we observed that a large fraction of microemboli infused through the internal carotid artery failed to be lysed or washed out within 48 h. Instead, emboli were found to translocate outside the vessel lumen within 2–7 days, leading to complete re-establishment of blood flow and sparing of the vessel. Recanalization occurred by a previously unknown mechanism of microvascular plasticity involving the rapid envelopment of emboli by endothelial membrane projections that subsequently form a new vessel wall. This was followed by the formation of an endothelial opening through which emboli translocated into the perivascular parenchyma. The rate of embolus extravasation was significantly decreased by pharmacological inhibition of matrix metalloproteinase 2/9 activity. In aged mice, extravasation was markedly delayed, resulting in persistent tissue hypoxia, synaptic damage and cell death. Alterations in the efficiency of the protective mechanism that we have identified may have important implications in microvascular pathology, stroke recovery and age-related cognitive decline.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Siebler, M., Kleinschmidt, A., Sitzer, M., Steinmetz, H. & Freund, H. J. Cerebral microembolism in symptomatic and asymptomatic high-grade internal carotid artery stenosis. Neurology 44, 615–618 (1994)
Markus, H. S., Thomson, N. D. & Brown, M. M. Asymptomatic cerebral embolic signals in symptomatic and asymptomatic carotid artery disease. Brain 118, 1005–1011 (1995)
Vermeer, S. E. et al. Silent brain infarcts and the risk of dementia and cognitive decline. N. Engl. J. Med. 348, 1215–1222 (2003)
Collen, D. On the regulation and control of fibrinolysis. Edward Kowalski Memorial Lecture. Thromb. Haemost. 43, 77–89 (1980)
Powers, W. J., Grubb, R. L. Jr, Darriet, D. & Raichle, M. E. Cerebral blood flow and cerebral metabolic rate of oxygen requirements for cerebral function and viability in humans. J. Cereb. Blood Flow Metab. 5, 600–608 (1985)
Levin, E. G. & del Zoppo, G. J. Localization of tissue plasminogen activator in the endothelium of a limited number of vessels. Am. J. Pathol. 144, 855–861 (1994)
Zlokovic, B. V. et al. Expression of tissue plasminogen activator in cerebral capillaries: possible fibrinolytic function of the blood–brain barrier. Neurosurgery 37, 955–961 (1995)
Rapp, J. H. et al. Atheroemboli to the brain: size threshold for causing acute neuronal cell death. J. Vasc. Surg. 32, 68–76 (2000)
Grutzendler, J., Kasthuri, N. & Gan, W. B. Long-term dendritic spine stability in the adult cortex. Nature 420, 812–816 (2002)
Muller, W. A. Leukocyte–endothelial–cell interactions in leukocyte transmigration and the inflammatory response. Trends Immunol. 24, 327–334 (2003)
Engelhardt, B. & Wolburg, H. Mini-review: Transendothelial migration of leukocytes: through the front door or around the side of the house? Eur. J. Immunol. 34, 2955–2963 (2004)
Dejana, E. Endothelial cell–cell junctions: happy together. Nature Rev. Mol. Cell Biol. 5, 261–270 (2004)
Kamei, M. et al. Endothelial tubes assemble from intracellular vacuoles in vivo . Nature 442, 453–456 (2006)
Carman, C. V. & Springer, T. A. A transmigratory cup in leukocyte diapedesis both through individual vascular endothelial cells and between them. J. Cell Biol. 167, 377–388 (2004)
Peppiatt, C. M., Howarth, C., Mobbs, P. & Attwell, D. Bidirectional control of CNS capillary diameter by pericytes. Nature 443, 700–704 (2006)
Yang, Y., Estrada, E. Y., Thompson, J. F., Liu, W. & Rosenberg, G. A. Matrix metalloproteinase-mediated disruption of tight junction proteins in cerebral vessels is reversed by synthetic matrix metalloproteinase inhibitor in focal ischemia in rat. J. Cereb. Blood Flow Metab. 27, 697–709 (2007)
Stratman, A. N. et al. Endothelial cell lumen and vascular guidance tunnel formation requires MT1-MMP-dependent proteolysis in 3-dimensional collagen matrices. Blood 114, 237–247 (2009)
Page-McCaw, A., Ewald, A. J. & Werb, Z. Matrix metalloproteinases and the regulation of tissue remodelling. Nature Rev. Mol. Cell Biol. 8, 221–233 (2007)
Farkas, E. & Luiten, P. G. Cerebral microvascular pathology in aging and Alzheimer’s disease. Prog. Neurobiol. 64, 575–611 (2001)
Zhao, B. Q. et al. Role of matrix metalloproteinases in delayed cortical responses after stroke. Nature Med. 12, 441–445 (2006)
Zhang, S., Boyd, J., Delaney, K. & Murphy, T. H. Rapid reversible changes in dendritic spine structure in vivo gated by the degree of ischemia. J. Neurosci. 25, 5333–5338 (2005)
Nishimura, N. et al. Targeted insult to subsurface cortical blood vessels using ultrashort laser pulses: three models of stroke. Nature Methods 3, 99–108 (2006)
Gleerup, G. & Winther, K. The effect of ageing on platelet function and fibrinolytic activity. Angiology 46, 715–718 (1995)
Arvanitakis, Z., Wilson, R. S., Bienias, J. L., Evans, D. A. & Bennett, D. A. Diabetes mellitus and risk of Alzheimer disease and decline in cognitive function. Arch. Neurol. 61, 661–666 (2004)
Iadecola, C. Neurovascular regulation in the normal brain and in Alzheimer’s disease. Nature Rev. Neurosci. 5, 347–360 (2004)
Lo, E. H., Dalkara, T. & Moskowitz, M. A. Mechanisms, challenges and opportunities in stroke. Nature Rev. Neurosci. 4, 399–415 (2003)
Abbott, N. J., Ronnback, L. & Hansson, E. Astrocyte–endothelial interactions at the blood-brain barrier. Nature Rev. Neurosci. 7, 41–53 (2006)
Zlokovic, B. V. The blood–brain barrier in health and chronic neurodegenerative disorders. Neuron 57, 178–201 (2008)
Pugsley, W. et al. The impact of microemboli during cardiopulmonary bypass on neuropsychological functioning. Stroke 25, 1393–1399 (1994)
Kleinfeld, D., Mitra, P. P., Helmchen, F. & Denk, W. Fluctuations and stimulus-induced changes in blood flow observed in individual capillaries in layers 2 through 4 of rat neocortex. Proc. Natl Acad. Sci. USA 95, 15741–15746 (1998)
Acknowledgements
We thank S. Lo, C. Freitas, D. Choi and C. Whiteus for help with experiments; A. Schain for suggesting the use of ACTB–eGFP mice; E. Mugnaini for advice with transmission electron microscopy experiments; and G. D’avossa, J. García-Añoveros, J. Kessler and P. Opal for helpful discussions and critical review of the manuscript. This work was supported by National Institutes of Health/National Institute on Aging grant AG027855 (J.G.) and a Howard Hughes Medical Institute medical student research fellowship (C.K.L.).
Author information
Authors and Affiliations
Contributions
J.G., C.K.L. and T.Y. conceived and designed the project. C.K.L., T.Y., B.H. and J.G. performed in vivo two-photon imaging. C.K.L., B.H. and J.G. adapted the technique and performed electron microscopy experiments. C.K.L. and T.Y. performed in situ zymography, ageing mice and SB-3CT experiments. T.Y. performed cell culture and the imaging experiment on human umbilical-vein endothelial cells. Z.L., C.K.L., T.Y., B.H. and J.G. performed histological and confocal microscopy experiments. J.G. wrote the manuscript with significant input from C.K.L. and T.Y.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-14 with legends and legends for Supplementary Movies 1-5. (PDF 1611 kb)
Supplementary Movie 1
This movie shows embolus in the process of extravasation – see Supplementary Information file for full legend. (MOV 22465 kb)
Supplementary Movie 2
This movie shows embolus extravasation and reestablishment of blood flow – see Supplementary Information file for full legend. (MOV 2156 kb)
Supplementary Movie 3
This movie shows microsphere extravasation – see Supplementary Information file for full legend. (MOV 6848 kb)
Supplementary Movie 4
This movie shows early extravasation and reestablishment of blood flow – see Supplementary Information file for full legend. (MOV 1378 kb)
Supplementary Movie 5
This movie shows cholesterol embolus occlusion – see Supplementary Information file for full legend. (MOV 778 kb)
Rights and permissions
About this article
Cite this article
Lam, C., Yoo, T., Hiner, B. et al. Embolus extravasation is an alternative mechanism for cerebral microvascular recanalization. Nature 465, 478–482 (2010). https://doi.org/10.1038/nature09001
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature09001
This article is cited by
-
Erythrocyte–brain endothelial interactions induce microglial responses and cerebral microhemorrhages in vivo
Journal of Neuroinflammation (2023)
-
Debris generated by laser and/or balloon cause cerebral infarction with different severity
Lasers in Medical Science (2023)
-
Matrix stiffness, endothelial dysfunction and atherosclerosis
Molecular Biology Reports (2023)
-
Assessment of the epi-pericardial fibrotic substrate by collagen-targeted probes
Scientific Reports (2022)
-
Targeting hypercoagulation to alleviate Alzheimer’s disease progression in metabolic syndrome
International Journal of Obesity (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.