Abstract
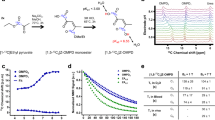
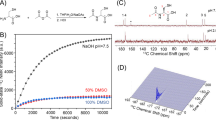
As alterations in tissue pH underlie many pathological processes, the capability to image tissue pH in the clinic could offer new ways of detecting disease and response to treatment1. Dynamic nuclear polarization is an emerging technique for substantially increasing the sensitivity of magnetic resonance imaging experiments2,3. Here we show that tissue pH can be imaged in vivo from the ratio of the signal intensities of hyperpolarized bicarbonate (H13CO3-) and 13CO2 following intravenous injection of hyperpolarized H13CO3-. The technique was demonstrated in a mouse tumour model, which showed that the average tumour interstitial pH was significantly lower than the surrounding tissue. Given that bicarbonate is an endogenous molecule that can be infused in relatively high concentrations into patients4, we propose that this technique could be used clinically to image pathological processes that are associated with alterations in tissue pH, such as cancer, ischaemia and inflammation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gillies, R. J., Raghunand, N., Garcia-Martin, M. L. & Gatenby, R. A. pH imaging. A review of pH measurement methods and applications in cancers. IEEE Eng. Med. Biol. Mag. 23, 57–64 (2004)
Ardenkjaer-Larsen, J. H. et al. Increase in signal-to-noise ratio of > 10,000 times in liquid-state NMR. Proc. Natl Acad. Sci. USA 100, 10158–10163 (2003)
Day, S. E. et al. Detecting tumor response to treatment using hyperpolarized 13C magnetic resonance imaging and spectroscopy. Nature Med. 13, 1382–1387 (2007)
Adrogue, H. J. & Madias, N. E. Management of life-threatening acid-base disorders. First of two parts. N. Engl. J. Med. 338, 26–34 (1998)
White, A., Handler, P. & Smith, E. L. Principles of Biochemistry (McGraw-Hill Kogakusha, New York, 1968)
Meldrum, N. U. & Roughton, F. J. Carbonic anhydrase. Its preparation and properties. J. Physiol. (Lond.) 80, 113–142 (1933)
Grinstein, S., Swallow, C. J. & Rotstein, O. D. Regulation of cytoplasmic pH in phagocytic cell function and dysfunction. Clin. Biochem. 24, 241–247 (1991)
Hohn-Berlage, M., Okada, Y., Kloiber, O. & Hossmann, K. A. Imaging of brain tissue pH and metabolites. A new approach for the validation of volume-selective NMR spectroscopy. NMR Biomed. 2, 240–245 (1989)
Raghunand, N., Mahoney, B., van Sluis, R., Baggett, B. & Gillies, R. J. Acute metabolic alkalosis enhances response of C3H mouse mammary tumors to the weak base mitoxantrone. Neoplasia 3, 227–235 (2001)
Raghunand, N. et al. Enhancement of chemotherapy by manipulation of tumour pH. Br. J. Cancer 80, 1005–1011 (1999)
Stubbs, M. et al. An assessment of 31P MRS as a method of measuring pH in rat tumours. NMR Biomed. 5, 351–359 (1992)
Beauregard, D. A., Parker, D. & Brindle, K. M. Relaxation-based mapping of tumor pH. Proc. Int. Soc. Magn. Reson. Med. 653 (1998)
Raghunand, N., Zhang, S., Sherry, A. D. & Gillies, R. J. In vivo magnetic resonance imaging of tissue pH using a novel pH-sensitive contrast agent, GdDOTA-4AmP. Acad. Radiol. 9 (Suppl 2). S481–S483 (2002)
Ward, K. M. & Balaban, R. S. Determination of pH using water protons and chemical exchange dependent saturation transfer (CEST). Magn. Reson. Med. 44, 799–802 (2000)
Zhou, J., Payen, J. F., Wilson, D. A., Traystman, R. J. & van Zijl, P. C. Using the amide proton signals of intracellular proteins and peptides to detect pH effects in MRI. Nature Med. 9, 1085–1090 (2003)
Mason, R. P. Transmembrane pH gradients in vivo: Measurements using fluorinated vitamin B6 derivatives. Curr. Med. Chem. 6, 481–499 (1999)
van Sluis, R. et al. In vivo imaging of extracellular pH using 1H MRSI. Magn. Reson. Med. 41, 743–750 (1999)
Gillies, R. J., Liu, Z. & Bhujwalla, Z. 31P-MRS measurements of extracellular pH of tumors using 3-aminopropylphosphonate. Am. J. Physiol. 267, C195–C203 (1994)
Rottenberg, D. A. et al. In vivo measurement of brain tumor pH using [11C]DMO and positron emission tomography. Ann. Neurol. 17, 70–79 (1985)
Vermathen, P., Capizzano, A. A. & Maudsley, A. A. Administration and 1H MRS detection of histidine in human brain: Application to in vivo pH measurement. Magn. Reson. Med. 43, 665–675 (2000)
Stabenau, E. K. & Heming, T. A. Determination of the constants of the Henderson-Hasselbalch equation, αCO2 and pKa, in sea turtle plasma. J. Exp. Biol. 180, 311–314 (1993)
Golman, K. & Petersson, J. S. Metabolic imaging and other applications of hyperpolarized 13C. Acad. Radiol. 13, 932–942 (2006)
Merritt, M. E. et al. Hyperpolarized 13C allows a direct measure of flux through a single enzyme-catalyzed step by NMR. Proc. Natl Acad. Sci. USA 104, 19773–19777 (2007)
Golman, K., Zandt, R. I., Lerche, M., Pehrson, R. & Ardenkjaer-Larsen, J. H. Metabolic imaging by hyperpolarized 13C magnetic resonance imaging for in vivo tumor diagnosis. Cancer Res. 66, 10855–10860 (2006)
McCoy, C. L. et al. The effect of blood flow modification on intra- and extracellular pH measured by 31P magnetic resonance spectroscopy in murine tumours. Br. J. Cancer 72, 905–911 (1995)
Swietach, P., Vaughan-Jones, R. D. & Harris, A. L. Regulation of tumor pH and the role of carbonic anhydrase 9. Cancer Metastasis Rev. 26, 299–310 (2007)
Hoffman, D. W. & Henkens, R. W. The rates of fast reactions of carbon dioxide and bicarbonate in human erythrocytes measured by carbon-13 NMR. Biochem. Biophys. Res. Commun. 143, 67–73 (1987)
Brindle, K. M. NMR methods for measuring enzyme kinetics in vivo . Prog. NMR Spectrosc. 20, 257–293 (1988)
Maren, T. H. Use of inhibitors in physiological studies of carbonic anhydrase. Am. J. Physiol. 232, F291–F297 (1977)
Zhao, M., Beauregard, D. A., Loizou, L., Davletov, B. & Brindle, K. M. Non-invasive detection of apoptosis using magnetic resonance imaging and a targeted contrast agent. Nature Med. 7, 1241–1244 (2001)
Acknowledgements
We thank R. McLaughlin and T. Witney for comments on the manuscript and S. Vowler for help with statistics. F.A.G. is in receipt of a Cancer Research UK and Royal College of Radiologists (UK) clinical research training fellowship, and S.E.D. a National Institutes of Health-Cambridge studentship. The work was supported by a Cancer Research UK Programme grant (to K.M.B.; C197/A3514). The polarizer and related materials were provided by GE Healthcare.
Author Contributions F.A.G. and S.E.D. operated the polarizer and J.H.A.-L. and K.G. provided advice on its use. M.I.K. conducted the MRI experiments and D.-E.H. prepared the animals. M.I.K. and F.A.G. analysed the data. R.I.Z., P.R.J., M.K. and M.H.L. devised the bicarbonate formulation. F.A.G. and K.M.B. wrote the paper and K.M.B. devised and organized the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The hyperpolarizer is on loan from GE Healthcare and is the subject of a research agreement between the University of Cambridge, Cancer Research UK and GE Healthcare. GE Healthcare also supplied the trityl radical used in the hyperpolarization process. Imagnia AB has supplied information about formulations for polarizing bicarbonate.
Rights and permissions
About this article
Cite this article
Gallagher, F., Kettunen, M., Day, S. et al. Magnetic resonance imaging of pH in vivo using hyperpolarized 13C-labelled bicarbonate. Nature 453, 940–943 (2008). https://doi.org/10.1038/nature07017
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07017
This article is cited by
-
Imaging cancer metabolism using magnetic resonance
npj Imaging (2024)
-
Severely polarized extracellular acidity around tumour cells
Nature Biomedical Engineering (2024)
-
A pH-dependent anti-CD47 antibody that selectively targets solid tumors and improves therapeutic efficacy and safety
Journal of Hematology & Oncology (2023)
-
Using optimal controlled singlet spin order to accurately target molecular signal in MRI and MRS
Scientific Reports (2023)
-
Opportunities and challenges with hyperpolarized bioresponsive probes for functional imaging using magnetic resonance
Nature Chemistry (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.