Abstract
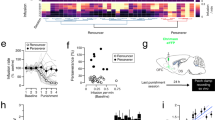
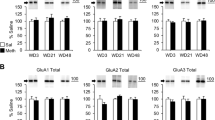
Defining the drug-induced neuroadaptations specifically associated with the behavioral manifestation of addiction is a daunting task. To address this issue, we used a behavioral model that differentiates rats controlling their drug use (Non-Addict-like) from rats undergoing transition to addiction (Addict-like). Dysfunctions in prefrontal cortex (PFC) synaptic circuits are thought to be responsible for the loss of control over drug taking that characterizes addicted individuals. Here, we studied the synaptic alterations in prelimbic PFC (pPFC) circuits associated with transition to addiction. We discovered that some of the changes induced by cocaine self-administration (SA), such as the impairment of the endocannabinoid-mediated long-term synaptic depression (eCB-LTD) was similarly abolished in Non-Addict- and Addict-like rats and thus unrelated to transition to addiction. In contrast, metabotropic glutamate receptor 2/3-mediated LTD (mGluR2/3-LTD) was specifically suppressed in Addict-like rats, which also show a concomitant postsynaptic plasticity expressed as a change in the relative contribution of AMPAR and NMDAR to basal glutamate-mediated synaptic transmission. Addiction-associated synaptic alterations in the pPFC were not fully developed at early stages of cocaine SA, when addiction-like behaviors are still absent, suggesting that pathological behaviors appear once the pPFC is compromised. These data identify specific synaptic impairments in the pPFC associated with addiction and support the idea that alterations of synaptic plasticity are core markers of drug dependence.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Deroche-Gamonet V, Piazza PV . Transition to addiction. In: Koon G, Le Moal M (eds) Encyclopedia of Behavioral Neuroscience. Academic Press: Oxford, 2010.
Kalivas PW . The glutamate homeostasis hypothesis of addiction. Nat Rev Neurosci 2009; 10: 561–572.
Koob GF, Le Moal M . Plasticity of reward neurocircuitry and the ‘dark side’ of drug addiction. Nat Neurosci 2005; 8: 1442–1444.
Hyman SE, Malenka RC, Nestler EJ . Neural mechanisms of addiction: the role of reward-related learning and memory. Annu Rev Neurosci 2006; 29: 565–598.
Renthal W, Nestler EJ . Epigenetic mechanisms in drug addiction. Trends Mol Med 2008; 14: 341–350.
Luscher C, Malenka RC . Drug-evoked synaptic plasticity in addiction: from molecular changes to circuit remodeling. Neuron 2011; 69: 650–663.
Kalivas PW . How do we determine which drug-induced neuroplastic changes are important? Nat Neurosci 2005; 8: 1440–1441.
Deroche-Gamonet V, Belin D, Piazza PV . Evidence for addiction-like behavior in the rat. Science 2004; 305: 1014–1017.
Belin D, Balado E, Piazza PV, Deroche-Gamonet V . Pattern of intake and drug craving predict the development of cocaine addiction-like behavior in rats. Biol Psychiatry 2009; 65: 863–868.
Belin D, Berson N, Balado E, Piazza PV, Deroche-Gamonet V . High-novelty-preference rats are predisposed to compulsive cocaine self-administration. Neuropsychopharmacology 2011; 36: 569–579.
Kasanetz F, Deroche-Gamonet V, Berson N, Balado E, Lafourcade M, Manzoni O et al. Transition to addiction is associated with a persistent impairment in synaptic plasticity. Science 2010; 328: 1709–1712.
Kauer JA, Malenka RC . Synaptic plasticity and addiction. Nat Rev Neurosci 2007; 8: 844–858.
Everitt BJ, Robbins TW . Neural systems of reinforcement for drug addiction: from actions to habits to compulsion. Nat Neurosci 2005; 8: 1481–1489.
Goldstein RZ, Craig AD, Bechara A, Garavan H, Childress AR, Paulus MP et al. The neurocircuitry of impaired insight in drug addiction. Trends Cogn Sci 2009; 13: 372–380.
Kalivas PW, Volkow N, Seamans J . Unmanageable motivation in addiction: a pathology in prefrontal-accumbens glutamate transmission. Neuron 2005; 45: 647–650.
Sun W, Rebec GV . Repeated cocaine self-administration alters processing of cocaine-related information in rat prefrontal cortex. J Neurosci 2006; 26: 8004–8008.
Volkow ND, Fowler JS, Wang GJ, Goldstein RZ . Role of dopamine, the frontal cortex and memory circuits in drug addiction: insight from imaging studies. Neurobiol Learn Mem 2002; 78: 610–624.
Robinson TE, Gorny G, Mitton E, Kolb B . Cocaine self-administration alters the morphology of dendrites and dendritic spines in the nucleus accumbens and neocortex. Synapse 2001; 39: 257–266.
Peters J, Kalivas PW, Quirk GJ . Extinction circuits for fear and addiction overlap in prefrontal cortex. Learn Mem 2009; 16: 279–288.
Lasseter HC, Xie X, Ramirez DR, Fuchs RA . Prefrontal cortical regulation of drug seeking in animal models of drug relapse. Curr Top Behav Neurosci 2010; 3: 101–117.
Capriles N, Rodaros D, Sorge RE, Stewart J . A role for the prefrontal cortex in stress- and cocaine-induced reinstatement of cocaine seeking in rats. Psychopharmacology (Berl) 2003; 168: 66–74.
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th edn, revised edn. American Psychiatric Press: Washington, DC, 2000.
Kasanetz F, Manzoni OJ . Maturation of excitatory synaptic transmission of the rat nucleus accumbens from juvenile to adult. J Neurophysiol 2009; 101: 2516–2527.
Lafourcade M, Elezgarai I, Mato S, Bakiri Y, Grandes P, Manzoni OJ . Molecular components and functions of the endocannabinoid system in mouse prefrontal cortex. PLoS One 2007; 2: e709.
Revest JM, Kaouane N, Mondin M, Le Roux A, Rouge-Pont F, Vallee M et al. The enhancement of stress-related memory by glucocorticoids depends on synapsin-Ia/Ib. Mol Psychiatry 2010; 15: 1125, 1140–1151.
Revest JM, Di Blasi F, Kitchener P, Rouge-Pont F, Desmedt A, Turiault M et al. The MAPK pathway and Egr-1 mediate stress-related behavioral effects of glucocorticoids. Nat Neurosci 2005; 8: 664–672.
Marrs WR, Blankman JL, Horne EA, Thomazeau A, Lin YH, Coy J et al. The serine hydrolase ABHD6 controls the accumulation and efficacy of 2-AG at cannabinoid receptors. Nat Neurosci 2010; 13: 951–957.
Maldonado R, Valverde O, Berrendero F . Involvement of the endocannabinoid system in drug addiction. Trends Neurosci 2006; 29: 225–232.
De Vries TJ, Schoffelmeer AN . Cannabinoid CB1 receptors control conditioned drug seeking. Trends Pharmacol Sci 2005; 26: 420–426.
De Vries TJ, Shaham Y, Homberg JR, Crombag H, Schuurman K, Dieben J et al. A cannabinoid mechanism in relapse to cocaine seeking. Nat Med 2001; 7: 1151–1154.
Orio L, Edwards S, George O, Parsons LH, Koob GF . A role for the endocannabinoid system in the increased motivation for cocaine in extended-access conditions. J Neurosci 2009; 29: 4846–4857.
Soria G, Mendizabal V, Tourino C, Robledo P, Ledent C, Parmentier M et al. Lack of CB1 cannabinoid receptor impairs cocaine self-administration. Neuropsychopharmacology 2005; 30: 1670–1680.
Fourgeaud L, Mato S, Bouchet D, Hemar A, Worley PF, Manzoni OJ . A single in vivo exposure to cocaine abolishes endocannabinoid-mediated long-term depression in the nucleus accumbens. J Neurosci 2004; 24: 6939–6945.
Huang CC, Yang PC, Lin HJ, Hsu KS . Repeated cocaine administration impairs group II metabotropic glutamate receptor-mediated long-term depression in rat medial prefrontal cortex. J Neurosci 2007; 27: 2958–2968.
Robbe D, Alonso G, Chaumont S, Bockaert J, Manzoni OJ . Role of p/q-Ca2+ channels in metabotropic glutamate receptor 2/3-dependent presynaptic long-term depression at nucleus accumbens synapses. J Neurosci 2002; 22: 4346–4356.
Moussawi K, Kalivas PW . Group II metabotropic glutamate receptors (mGlu2/3) in drug addiction. Eur J Pharmacol 2010; 639: 115–122.
Thomas MJ, Beurrier C, Bonci A, Malenka RC . Long-term depression in the nucleus accumbens: a neural correlate of behavioral sensitization to cocaine. Nat Neurosci 2001; 4: 1217–1223.
Dumont EC, Mark GP, Mader S, Williams JT . Self-administration enhances excitatory synaptic transmission in the bed nucleus of the stria terminalis. Nat Neurosci 2005; 8: 413–414.
Ungless MA, Whistler JL, Malenka RC, Bonci A . Single cocaine exposure in vivo induces long-term potentiation in dopamine neurons. Nature 2001; 411: 583–587.
Vanderschuren LJ, Everitt BJ . Drug seeking becomes compulsive after prolonged cocaine self-administration. Science 2004; 305: 1017–1019.
Adewale AS, Platt DM, Spealman RD . Pharmacological stimulation of group ii metabotropic glutamate receptors reduces cocaine self-administration and cocaine-induced reinstatement of drug seeking in squirrel monkeys. J Pharmacol Exp Ther 2006; 318: 922–931.
Baptista MA, Martin-Fardon R, Weiss F . Preferential effects of the metabotropic glutamate 2/3 receptor agonist LY379268 on conditioned reinstatement versus primary reinforcement: comparison between cocaine and a potent conventional reinforcer. J Neurosci 2004; 24: 4723–4727.
Peters J, Kalivas PW . The group II metabotropic glutamate receptor agonist, LY379268, inhibits both cocaine- and food-seeking behavior in rats. Psychopharmacology (Berl) 2006; 186: 143–149.
Morishima Y, Miyakawa T, Furuyashiki T, Tanaka Y, Mizuma H, Nakanishi S . Enhanced cocaine responsiveness and impaired motor coordination in metabotropic glutamate receptor subtype 2 knockout mice. Proc Natl Acad Sci USA 2005; 102: 4170–4175.
Spanagel R . Are metabotropic glutamate receptors promising targets for the treatment of alcoholism? Biol Psychiatry 2010; 67: 798–799.
Moussawi K, Pacchioni A, Moran M, Olive MF, Gass JT, Lavin A et al. N-Acetylcysteine reverses cocaine-induced metaplasticity. Nat Neurosci 2009; 12: 182–189.
Moussawi K, Zhou W, Shen H, Reichel CM, See RE, Carr DB et al. Reversing cocaine-induced synaptic potentiation provides enduring protection from relapse. Proc Natl Acad Sci USA 2010; 108: 385–390.
Bowers MS, McFarland K, Lake RW, Peterson YK, Lapish CC, Gregory ML et al. Activator of G protein signaling 3: a gatekeeper of cocaine sensitization and drug seeking. Neuron 2004; 42: 269–281.
Xi ZX, Ramamoorthy S, Baker DA, Shen H, Samuvel DJ, Kalivas PW . Modulation of group II metabotropic glutamate receptor signaling by chronic cocaine. J Pharmacol Exp Ther 2002; 303: 608–615.
Chen BT, Bowers MS, Martin M, Hopf FW, Guillory AM, Carelli RM et al. Cocaine but not natural reward self-administration nor passive cocaine infusion produces persistent LTP in the VTA. Neuron 2008; 59: 288–297.
Martin M, Chen BT, Hopf FW, Bowers MS, Bonci A . Cocaine self-administration selectively abolishes LTD in the core of the nucleus accumbens. Nat Neurosci 2006; 9: 868–869.
Mu P, Moyer JT, Ishikawa M, Zhang Y, Panksepp J, Sorg BA et al. Exposure to cocaine dynamically regulates the intrinsic membrane excitability of nucleus accumbens neurons. J Neurosci 2010; 30: 3689–3699.
McCutcheon JE, Wang X, Tseng KY, Wolf ME, Marinelli M . Calcium-permeable AMPA receptors are present in nucleus accumbens synapses after prolonged withdrawal from cocaine self-administration but not experimenter-administered cocaine. J Neurosci 2011; 31: 5737–5743.
Huang CC, Lin HJ, Hsu KS . Repeated cocaine administration promotes long-term potentiation induction in rat medial prefrontal cortex. Cereb Cortex 2007; 17: 1877–1888.
Dong Y, Nasif FJ, Tsui JJ, Ju WY, Cooper DC, Hu XT et al. Cocaine-induced plasticity of intrinsic membrane properties in prefrontal cortex pyramidal neurons: adaptations in potassium currents. J Neurosci 2005; 25: 936–940.
Heifets BD, Castillo PE . Endocannabinoid signaling and long-term synaptic plasticity. Annu Rev Physiol 2009; 71: 283–306.
Grueter BA, Gosnell HB, Olsen CM, Schramm-Sapyta NL, Nekrasova T, Landreth GE et al. Extracellular-signal regulated kinase 1-dependent metabotropic glutamate receptor 5-induced long-term depression in the bed nucleus of the stria terminalis is disrupted by cocaine administration. J Neurosci 2006; 26: 3210–3219.
Swanson CJ, Baker DA, Carson D, Worley PF, Kalivas PW . Repeated cocaine administration attenuates group I metabotropic glutamate receptor-mediated glutamate release and behavioral activation: a potential role for Homer. J Neurosci 2001; 21: 9043–9052.
Stuber GD, Klanker M, de Ridder B, Bowers MS, Joosten RN, Feenstra MG et al. Reward-predictive cues enhance excitatory synaptic strength onto midbrain dopamine neurons. Science 2008; 321: 1690–1692.
Yin HH, Mulcare SP, Hilario MR, Clouse E, Holloway T, Davis MI et al. Dynamic reorganization of striatal circuits during the acquisition and consolidation of a skill. Nat Neurosci 2009; 12: 333–341.
Mameli M, Halbout B, Creton C, Engblom D, Parkitna JR, Spanagel R et al. Cocaine-evoked synaptic plasticity: persistence in the VTA triggers adaptations in the NAc. Nat Neurosci 2009; 12: 1036–1041.
Acknowledgements
This work was supported by INSERM, Région Aquitaine, ANR grant (2005; P-VP and OM), EU-STREP-PheCOMP grant (FP6; P-VP), MILDT/INCa/INSERM grant (2008; VD-G and OM) and AXA Research Fund (FK). We thank A Le Roux and V Roullot-Lacarriére for their technical assistance with the western blot experiments.
Author contributions: FK and ML performed the electrophysiology experiments, conducted the data analyses and contributed to the design of the experiments. VD-G designed and supervised the behavioral experiments, and conducted the data analyses. J-MR designed and conducted the immunoblotting analyses. NB, EB, J-FF and PR conducted the behavioral experiments. P-VP and OJM supervised the entire project. FK, VD-G, P-VP and OJM wrote the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website
Rights and permissions
About this article
Cite this article
Kasanetz, F., Lafourcade, M., Deroche-Gamonet, V. et al. Prefrontal synaptic markers of cocaine addiction-like behavior in rats. Mol Psychiatry 18, 729–737 (2013). https://doi.org/10.1038/mp.2012.59
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2012.59
Keywords
This article is cited by
-
Psilocybin-induced default mode network hypoconnectivity is blunted in alcohol-dependent rats
Translational Psychiatry (2023)
-
Pathways to the persistence of drug use despite its adverse consequences
Molecular Psychiatry (2023)
-
Increased responsiveness to punishment of cocaine self-administration after experience with high punishment
Neuropsychopharmacology (2022)
-
PAI-1 protein is a key molecular effector in the transition from normal to PTSD-like fear memory
Molecular Psychiatry (2021)
-
Drug addiction: from bench to bedside
Translational Psychiatry (2021)