Abstract
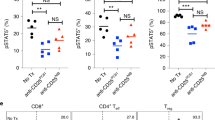
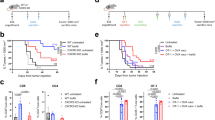
Immunomodulatory drugs (IMiDs) are effective therapeutic agents with direct inhibitory effects on malignant B- and plasma-cells and immunomodulatory effects on the T-cell activation. This dual function of IMiDs makes them appealing candidates for combination with a cancer vaccine. We investigated the immune stimulatory effects of lenalidomide, administrated to mice in doses, which provided comparable pharmacokinetics to human patients, on the potency of a novel fusion DNA lymphoma vaccine. The combination was curative in the majority of mice with 8d pre-established syngeneic A20 lymphomas compared with vaccine or lenalidomide alone and induced immune memory. In vivo depletion experiments established the requirement for effector CD8+ and CD4+ T cells in protective immunity. Unexpectedly, lenalidomide alone was also associated with reduced numbers of systemic myeloid-derived suppressor cell (MDSC) and regulatory T cell (Treg) in tumor-bearing but not naïve mice, an effect that was independent of simple tumor burden reduction. These results confirm and extend results from other models describing the effect of lenalidomide on enhancing T-cell immunity, highlight the potency of this effect, and provide a rationale for clinical application. Independently, a novel mechanism of action reversing tumor-induced immune suppression by MDSC is suggested.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Witzig TE, Wiernik PH, Moore T, Reeder C, Cole C, Justice G et al. Lenalidomide oral monotherapy produces durable responses in relapsed or refractory indolent non-hodgkin's lymphoma. J Clin Oncol 2009; 27: 5404–5409.
Witzig TE, Vose JM, Zinzani PL, Reeder CB, Buckstein R, Polikoff JA et al. An international phase II trial of single-agent lenalidomide for relapsed or refractory aggressive B-cell non-Hodgkin's lymphoma. Ann Oncol 2011; 22: 1622–1627.
Fowler N, McLaughlin P, Hagemeister FB, Kwak LW, Fanale M, Neelapu S et al. A Biologic combination of lenalidomide and rituximab for front-line therapy of indolent b-cell non-hodgkin’s lymphoma. Blood 2009; 114, abstract 1714.
Nowakowski GS, LaPlant B, Habermann TM, Rivera CE, Macon WR, Inwards DJ et al. Lenalidomide can be safely combined with R-CHOP (R2CHOP) in the initial chemotherapy for aggressive B-cell lymphomas: phase I study. Leukemia 2011; 25: 1877–1881.
Verhelle D, Corral LG, Wong K, Mueller JH, Moutouh-de Parseval L, Jensen-Pergakes K et al. Lenalidomide and CC-4047 inhibit the proliferation of malignant B cells while expanding normal CD34+ progenitor cells. Cancer Res 2007; 67: 746–755.
Hideshima T, Chauhan D, Shima Y, Raje N, Davies FE, Tai YT et al. Thalidomide and its analogs overcome drug resistance of human multiple myeloma cells to conventional therapy. Blood 2000; 96: 2943–2950.
Xu YB, Li JW, Ferguson GD, Mercurio F, Khambatta G, Morrison L et al. Immunomodulatory drugs reorganize cytoskeleton by modulating Rho GTPases. Blood 2009; 114: 338–345.
Lopez-Girona A, Heintel D, Zhang LH, Mendy D, Gaidarova S, Brady H et al. Lenalidomide downregulates the cell survival factor, interferon regulatory factor-4, providing a potential mechanistic link for predicting response. Br J Haematol 2011; 154: 325–336.
Li SR, Pal R, Monaghan SA, Schafer P, Ouyang HJ, Mapara M et al. IMiD immunomodulatory compounds block C/EBP beta translation through eIF4E down-regulation resulting in inhibition of MM. Blood 2011; 117: 5157–5165.
Dredge K, Marriott JB, Macdonald CD, Man HW, Chen R, Muller GW et al. Novel thalidomide analogues display anti-angiogenic activity independently of immunomodulatory effects. Br J Cancer 2002; 87: 1166–1172.
Lentzsch S, Rogers MS, LeBlanc R, Birsner AE, Shah JH, Treston AM et al. S-3-amino-phthalimido-glutarimide inhibits angiogenesis and growth of B-cell neoplasias in mice. Cancer Res 2002; 62: 2300–2305.
Zhu YX, Braggio E, Shi CX, Bruins LA, Schmidt JE, Van Wier S et al. Cereblon expression is required for the antimyeloma activity of lenalidomide and pomalidomide. Blood 2011; 118: 4771–4779.
Sampaio EP, Sarno EN, Galilly R, Cohn ZA, Kaplan G . Thalidomide selectively inhibits tumor-necrosis-factor-alpha production by stimulated human monocytes. J Exp Med 1991; 173: 699–703.
Davies FE, Raje N, Hideshima T, Lentzsch S, Young G, Tai YT et al. Thalidomide and immunomodulatory derivatives augment natural killer cell cytotoxicity in multiple myeloma. Blood 2001; 98: 210–216.
Gorgun G, Calabrese E, Soydan E, Hideshima T, Perrone G, Bandi M et al. Immunomodulatory effects of lenalidomide and pomalidomide on interaction of tumor and bone marrow accessory cells in multiple myeloma. Blood 2010; 116: 3227–3237.
LeBlanc R, Hideshima T, Catley LP, Shringarpure R, Burger R, Mitsiades N et al. Immunomodulatory drug costimulates T cells via the B7-CD28 pathway. Blood 2004; 103: 1787–1790.
Luptakova K, Rosenblatt J, Glotzbecker B, Mills H, Stroopinsky D, Kufe T et al. Lenalidomide enhances anti-myeloma cellular immunity. Cancer Immunol Immunother 2013; 62: 39–49.
Ramsay AG, Clear AJ, Kelly G, Fatah R, Matthews J, Macdougall F et al. Follicular lymphoma cells induce T-cell immunologic synapse dysfunction that can be repaired with lenalidomide: implications for the tumor microenvironment and immunotherapy. Blood 2009; 114: 4713–4720.
Cheever MA, Higano CS . PROVENGE (Sipuleucel-T) in prostate cancer: the first FDA-approved therapeutic cancer vaccine. Clin Cancer Res 2011; 17: 3520–3526.
Schwartzentruber DJ, Lawson DH, Richards JM, Conry RM, Miller DM, Treisman J et al. gp100 peptide vaccine and interleukin-2 in patients with advanced melanoma. N Engl J Med 2011; 364: 2119–2127.
Schuster SJ, Neelapu SS, Gause BL, Janik JE, Muggia FM, Gockerman JP et al. Vaccination with patient-specific tumor-derived antigen in first remission improves disease-free survival in follicular lymphoma. J Clin Oncol 2011; 29: 2787–2794.
Biragyn A, Ruffini PA, Leifer CA, Klyushnenkova E, Shakhov A, Chertov O et al. Toll-like receptor 4-dependent activation of dendritic cells by beta-defensin 2. Science 2002; 298: 1025–1029.
Biragyn A, Tani K, Grimm MC, Weeks S, Kwak LW . Genetic fusion of chemokines to a self tumor antigen induces protective, T-cell dependent antitumor immunity. Nat Biotechnol 1999; 17: 253–258.
Biragyn A, Ruffini PA, Coscia M, Harvey LK, Neelapu SS, Baskar S et al. Chemokine receptor-mediated delivery directs self-tumor antigen efficiently into the class II processing pathway in vitro and induces protective immunity in vivo. Blood 2004; 104: 1961–1969.
Biragyn A, Surenhu M, Yang D, Ruffini PA, Haines BA, Klyushnenkova E et al. Mediators of innate immunity that target immature, but not mature, dendritic cells induce antitumor immunity when genetically fused with nonimmunogenic tumor antigens. J Immunol 2001; 167: 6644–6653.
Qin H, Cha SC, Neelapu SS, Lou Y, Wei J, Liu YJ et al. Vaccine site inflammation potentiates idiotype DNA vaccine-induced therapeutic T cell-, and not B cell-, dependent antilymphoma immunity. Blood 2009; 114: 4142–4149.
Armstrong AC, Dermime S, Allinson CG, Bhattacharyya T, Mulryan K, Gonzalez KR et al. Immunization with a recombinant adenovirus encoding a lymphoma idiotype: induction of tumor-protective immunity and identification of an idiotype-specific T cell epitope. J Immunol 2002; 168: 3983–3991.
Iida S, Chou T, Okamoto S, Nagai H, Hatake K, Murakami H et al. Lenalidomide plus dexamethasone treatment in Japanese patients with relapsed/refractory multiple myeloma. Int J Hematol 2010; 92: 118–126.
Qin H, Cha SC, Neelapu SS, Liu C, Wang YH, Wei J et al. Generation of an immune microenvironment as a novel mechanism for myotoxins to potentiate genetic vaccines. Vaccine 2010; 28: 7970–7978.
Brody JD, Ai WZ, Czerwinski DK, Torchia JA, Levy M, Advani RH et al. In situ vaccination with a TLR9 agonist induces systemic lymphoma regression: a phase I/II study. J Clin Oncol 2010; 28: 4324–4332.
Dredge K, Marriott JB, Todryk SM, Muller GW, Chen R, Stirling DI et al. Protective antitumor immunity induced by a costimulatory thalidomide analog in conjunction with whole tumor cell vaccination is mediated by increased Th1-type immunity. J Immunol 2002; 168: 4914–4919.
Corral LG, Haslett PA, Muller GW, Chen R, Wong LM, Ocampo CJ et al. Differential cytokine modulation and T cell activation by two distinct classes of thalidomide analogues that are potent inhibitors of TNF-alpha. J Immunol 1999; 163: 380–386.
Noonan K, Rudraraju L, Ferguson A, Emerling A, Pasetti MF, Huff CA et al. Lenalidomide-induced immunomodulation in multiple myeloma: impact on vaccines and antitumor responses. Clin Cancer Res 2012; 18: 1426–1434.
Galustian C, Meyer B, Labarthe MC, Dredge K, Klaschka D, Henry J et al. The anti-cancer agents lenalidomide and pomalidomide inhibit the proliferation and function of T regulatory cells. Cancer Immunol Immunother 2009; 58: 1033–1045.
Zhu D, Corral LG, Fleming YW, Stein B . Immunomodulatory drugs Revlimid (lenalidomide) and CC-4047 induce apoptosis of both hematological and solid tumor cells through NK cell activation. Cancer Immunol Immunother 2008; 57: 1849–1859.
Ito T, Ando H, Suzuki T, Ogura T, Hotta K, Imamura Y et al. Identification of a primary target of thalidomide teratogenicity. Science 2010; 327: 1345–1350.
Acknowledgements
This work was supported by a contract from Celgene Corporation and by grants from the Leukemia and Lymphoma Society (TRP 6003-07 and SCOR 7262-08), Cancer Prevention and Research Institute of Texas (RP 100457) and Department of Defense (W81XWH-07) to LWK.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Larry W Kwak has been funded by Celgene. Jian Chen, Sekhar Surapaneni, Scott Bateman are employed by Celgene. The remaining authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Sakamaki, I., Kwak, L., Cha, Sc. et al. Lenalidomide enhances the protective effect of a therapeutic vaccine and reverses immune suppression in mice bearing established lymphomas. Leukemia 28, 329–337 (2014). https://doi.org/10.1038/leu.2013.177
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2013.177
Keywords
This article is cited by
-
What happens to regulatory T cells in multiple myeloma
Cell Death Discovery (2023)
-
Application of Engineered Dendritic Cell Vaccines in Cancer Immunotherapy: Challenges and Opportunities
Current Treatment Options in Oncology (2023)
-
Myeloid-derived suppressor cells in hematologic malignancies: two sides of the same coin
Experimental Hematology & Oncology (2022)
-
Cancer combination therapies by angiogenesis inhibitors; a comprehensive review
Cell Communication and Signaling (2022)
-
Advances and perspectives of dendritic cell-based active immunotherapies in follicular lymphoma
Cancer Immunology, Immunotherapy (2020)