Abstract
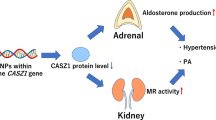
11β-Hydroxysteroid dehydrogenase type 2 (11β-HSD2) catalyzes the inactivation of cortisol (F) to cortisone (E) in aldosterone target tissues, thereby protects mineralocorticoid receptor from F. Failure of 11β-HSD2 function is the basis of apparent mineralocorticoid excess, and its mild disturbances are suggested to lead to hypertension. The aim of the study was to analyze the 11β-HSD2 activity in hypertensives and healthy volunteers. Glucocorticoids (GCs) profile was estimated to verify whether the disorders of GCs balance may be involved in essential hypertension etiology. Exons and short introns of HSD11B2 were sequenced to evaluate existing mutations and their potential implications in the disease. The identified polymorphisms were assessed in case-control study to determine their relevance to hypertension. No significant differences in values of plasma F/E and UFF/UFE (urinary free F to free E) were observed between hypertensives and controls. The value of (THF+allo-THF)/(THE+allo-THE) (urinary tetrahydro-metabolites of F to tetrahydro-metabolites of E) in hypertensives was higher than in normotensives. Logistic regression demonstrated that the increase of one unit of (THF+allo-THF)/(THE+allo-THE) value increases the risk of hypertension over 11-fold. Genotyping indicated no hypertension related mutations in the coding region and short introns of HSD11B2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Litchfield WR, Hunt SC, Jeunemaitre X, Fisher NDL, Hopkins PN, Corvol RR et al. Increased urinary free cortisol: a potential intermediate phenotype of essential hypertension. Hypertension 1998; 31: 569–574.
Whitworth JA, Williamson PM, Mangos G, Kelly JJ . Cardiovascular consequences of cortisol excess. Vasc Health Risk Manag 2005; 1: 291–299.
White PC, Mune T, Rogerson FM, Kayes KM, Agarwal AK . Molecular analysis of 11β-hydroxysteroid dehydrogenase and its role in the syndrome of apparent mineralocorticoid excess. Steroids 1997; 62: 83–88.
Romer TE, Litwin M, Małunowicz E . Apparent mineralocorticoid excess. Pol J Endocrinol 2004; 4: 463–470.
Draper N, Stewart PM . 11β-hydroxysteroid dehydrogenase and the pre-receptor regulation of corticosteroid hormone action. J Endocrinol 2005; 186: 251–271.
National Center for Biotechnology Information. www.ncbi.nlm.nih.gov Access date 28 January 2010.
Ensembl Genome Browser. www.ensembl.org Access date 28 January 2010.
Agarwal AK, Rogerson FM, Mune T, White PC . Gene structure and chromosomal localization of the human HSD11K gene encoding the kidney (type2) isozyme of 11β-hydroxysteroid dehydrogenase. Genomics 1995; 29: 195–199.
Tomlinson JW, Stewart PM . Cortisol metabolism and the role of 11β-hydroxysteroid dehydrogenase. Best Pract Res Clin Endocrinol Metab 2001; 15: 61–78.
Ferrari P . The role of 11β-hydroxysteroid dehydrogenase type 2 in human hypertension. Biochim Biophys Acta 2010; 1802: 1178–1187.
Quinkler M, Stewart PM . Hypertension and the cortisol-cortisone shuttle. J Clin Endocrinol Metab 2003; 88: 2384–2392.
Melander O, Orho-Melander M, Bengtsson K, Lindblad U, Råstam L, Groop L et al. Association between a variant in the 11β-hydroxysteroid dehydrogenase type 2 gene and primary hypertension. J Hum Hypertens 2000; 14: 819–823.
Hammer F, Stewart PM . Cortisol metabolism in hypertension. Best Pract Res Clin Endocrinol Metab 2006; 20: 337–353.
Alikhani-Koopaei R, Fouladkou F, Frey FJ, Frey BM . Epigenetic regulation of 11β-hydroxysteroid dehydrogenase type 2 expression. J Clin Invest 2004; 114: 1146–1157.
Morineau G, Marc JM, Boudi A, Galons H, Gourmelen M, Corvol P et al. Genetic, biochemical and clinical studies of patients with A328V or R213C mutations in 11βHSD2 causing apparent mineralocorticoid excess. Hypertension 1999; 34: 435–441.
Główka FK, Kosicka K, Karaźniewicz-Łada M . HPLC method for determination of fluorescence derivatives of cortisol, cortisone and their tetrahydro- and allo-tetrahydro-metabolites in biological fluids. J Chromatogr B 2010; 878: 283–289.
Carvajal CA, Romero DG, Mosso LM, Gonzàlez AA, Campino C, Montero J et al. Biochemical and genetic characterization of 11β-hydroxysteroid dehydrogenase type 2 in low-renin essential hypertensives. J Hypertens 2005; 23: 71–77.
Krall P, Carvajal C, Ortiz E, Muñoz C, Garrido JL, Mosso L et al. Urinary free cortisol is not a biochemical marker of hypertension. Am J Hypertens 2007; 20: 459–465.
Whitworth JA, Schyvens CG, Zhang Y, Andrews MC, Mangos GJ, Kelly JJ . The nitric oxide system in glucocorticoid-induced hypertension. J Hypertens 2002; 20: 1035–1043.
Whitworth JA, Mangos GJ, Kelly JJ . Cushing, cortisol, and cardiovascular disease. Hypertension 2000; 36: 912–916.
Baum M, Moe OW . Glucocorticoid-mediated hypertension: does the vascular smooth muscle hold all the answers? J Am Soc Nephrol 2008; 19: 1251–1253.
Mariniello B, Ronconi V, Sardu C, Pagliericcio A, Galletti F, Strazzullo P et al. Analysis of the 11β-hydroxysteroid dehydrogenase type 2 gene (HSD11B2) in human essential hypertension. Am J Hypertens 2005; 18: 1091–1098.
Agarwal AK, Giacchetti G, Lavery G, Nikkila H, Palermo M, Ricketts M et al. CA-repeat polymorphism in intron 1 of HSD11B2: effects on gene expression and salt sensitivity. Hypertension 2000; 36: 187–194.
Sandeep TC, Walker BR . Pathophysiology of modulation of local glucocorticoid levels by 11β-hydroxysteroid dehydrogenases. TRENDS Endocrinol Metab 2011; 10: 446–453.
Stewart PM, Boulton A, Kumar S, Clark PMS . Shackleton CHL. Cortisol metabolism in human obesity: impaired cortisone→cortisol conversion in subjects with central adiposity. J Clin Endocrinol Metab 1999; 84: 1022–1027.
Rogoff D, Smolenicka Z, Bergadá I, Vallejo G, Barontini M, Heinrich JJ et al. The codon 213 of the 11β-hydroxysteroid dehydrogenase type 2 gene is a hot spot for mutations in apparent mineralocorticoid excess. J Clin Endocrinol Metab 1998; 83: 4391–4393.
Wilson RC, Harbison MD, Krozowski ZS, Funder JW, Shackleton CHL, Hanauske-Abel HM et al. Several homozygous mutations in the gene for 11β-hydroxysteroid dehydrogenase type 2 in patients with apparent mineralocorticoid excess. J Clin Endocrinol Metab 1995; 80: 3145–3150.
Odermat A, Dick B, Arnold P, Zaehner T, Plueschke V, Deregibus MN et al. A mutation in the cofactor-binding domain of 11β-hydroxysteroid dehydrogenase type 2 associated with mineralocorticoid hypertension. J Clin Endocrinol Metab 2001; 86: 1247–1252.
Lavery GG, Ronconi V, Draper N, Rabbitt EH, Lyons V, Chapman KE et al. Late-onset apparent mineralocorticoid excess caused by novel compound heterozygous mutations in the HSD11B2 gene. Hypertension 2003; 42: 123–129.
Zaehner T, Plueshke V, Frey BM, Frey FJ, Ferrari P . Structural analysis of the 11β-hydroxysteroid dehydrogenase type 2 gene in end-stage renal disease. Kidney Int 2000; 58: 1413–1419.
Brand E, Kato N, Chatelain N, Krozowski ZS, Jeunemaitre X, Corvol P et al. Structural analysis and evaluation of the 11β-hydroxysteroid dehydrogenase type 2 (11β-HSD2) gene in human essential hypertension. J Hypertens 1998; 16: 1627–1633.
Smolenicka Z, Bach E, Schaer S, Liechti-Gallati S, Frey BM, Frey FJ et al. A new polymorphic restriction site in the human 11β-hydroxysteroid dehydrogenase type 2 gene. J Clin Endocrinol Metab 1998; 83: 1814–1817.
Baserga M, Kaur R, Hale MA, Bares A, Yu X, Callaway CW et al. Fetal growth restriction alters transcription factor binding and epigenetic mechanisms of renal 11β-hydroxysteroid dehydrogenase type 2 in a sex-specific manner. Am J Physiol Regul Integr Comp Physiol 2010; 299: R334–R342.
Friso S, Pizzolo F, Choi S, Guarini P, Castagna A, Ravagnani V et al. Epigenetic control of 11β-hydroxysteroid dehydrogenase 2 gene promotor is related to human hypertension. Atherosclerosis 2008; 199: 323–327.
Acknowledgements
This work was supported by Polish Ministry of Science and Higher Education (grant number N-405-01632/1498). The authors would like to thank Professor Marek Niedziela from The Department of Pediatric Endocrinology and Rheumathology at Poznan University of Medical Sciences and Professor Elżbieta Kaczmarek from The Department of Bioinformatics and Computational Biology at Poznan University of Medical Sciences for their professional support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Kosicka, K., Cymerys, M., Majchrzak-Celińska, A. et al. 11β-Hydroxysteroid dehydrogenase type 2 in hypertension: comparison of phenotype and genotype analysis. J Hum Hypertens 27, 510–515 (2013). https://doi.org/10.1038/jhh.2012.67
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jhh.2012.67
Keywords
This article is cited by
-
Clinical, biochemical, and miRNA profile of subjects with positive screening of primary aldosteronism and nonclassic apparent mineralocorticoid excess
Endocrine (2022)
-
Apparent mineralocorticoid excess caused by novel compound heterozygous mutations in HSD11B2 and characterized by early-onset hypertension and hypokalemia
Endocrine (2020)
-
Mimicry and well known genetic friends: molecular diagnosis in an Iranian cohort of suspected Bartter syndrome and proposition of an algorithm for clinical differential diagnosis
Orphanet Journal of Rare Diseases (2019)