Abstract
Heterogeneous nuclear ribonucleoprotein K (hnRNP K) mediates antiapoptotic activity in part by inducing downstream antiapoptotic genes. To systematically identify hnRNP K targets in nasopharyngeal carcinoma (NPC), affymetrix chips were used to identify genes that were both overexpressed in primary NPC and downregulated by hnRNP K knockdown in NPC-TW02 cells. The resulting gene set included the antiapoptotic gene, FLIP, which was selected for further study. In cells treated with hnRNP K siRNA, TRAIL-induced apoptosis was enhanced and the FLIP protein level was reduced. Promoter, DNA pull-down and chromatin-immunoprecipitation assays revealed that hnRNP K directly interacts with the poly(C) element on the FLIP promoter, resulting in transcriptional activation. Through iTRAQ-mass spectrometric identification of proteins differentially associated with the poly(C) element or its mutant, nucleolin was determined to be a cofactor of hnRNP K for FLIP activation. Furthermore, FLIP was highly expressed in tumor cells, and this high-level expression was significantly correlated with high-level hnRNP K expression (P=0.002) and poor overall survival (P=0.015) as examined in 67 NPC tissues. A multivariate analysis confirmed that FLIP was an independent prognostic factor for NPC. Taken together, these findings indicate that FLIP expression is transcriptionally regulated by hnRNP K and nucleolin, and may be a potential prognostic and therapeutic marker for NPC.
Similar content being viewed by others
Main
Heterogeneous nuclear ribonucleoprotein K (hnRNP K) belongs to the hnRNP family of proteins. The members of this family interact directly with DNA and RNA through their K-homology domains and regulate gene expression at multiple levels, including transcription, RNA splicing, RNA stability and translation.1, 2 The expression of hnRNP K has been shown to be aberrantly increased in numerous cancers,3, 4, 5, 6 and we recently reported that high-level hnRNP K expression is correlated with poorer overall survival (OS) and decreased metastasis-free survival among nasopharyngeal carcinoma (NPC) patients.5 Our findings were consistent with those from clinical correlation studies in oral squamous cell carcinoma4 and prostate cancer.3
HnRNP K is a nucleocytoplasmic shuttling protein that is primarily located in the nucleus for transcriptional regulation.1 However, cytoplasmic accumulation of hnRNP K through ERK-mediated phosphorylation of hnRNP K serines-284 and -353 has been reported in cervical carcinoma HeLa,7 chronic myelogenous leukemia8 and NPC9 cells. The tumorigenic activity of hnRNP K appears to be conferred through its ability to increase proliferation,10 antiapoptotic effects,9 clonogenic potential8 and metastasis.11 These functions may be due, at least in part, to the ability of hnRNP K to upregulate the c-myc,8, 12 thymidine phosphorylase (TP)9 and eIF4E10 genes through transcriptional or post-transcriptional regulation. However, the full spectrum of targets regulated by hnRNP K has not as yet been systematically examined.
The acquisition of resistance to apoptosis is considered a hallmark of cancer, and impairment of apoptotic pathways is one of the underlying mechanisms of chemoresistance in cancer cells. The TRAIL ligand is known to be involved in apoptotic activity; TRAIL interacts with the DR4 and DR5 death receptors, thereby regulating the extrinsic apoptotic pathway through sequential recruitment of the Fas-associated death domain and inactive procaspase 8 (FLICE) to form the death-inducing signaling complex (DISC).13 At the DISC, activation of procaspase 8 triggers the activation of effector caspases 3 and -7, resulting in apoptosis. TRAIL has been shown to induce apoptosis in a broad range of tumor cells and in numerous preclinical trials, without evidence of systemic toxicity.13 TRAIL triggers apoptosis in cancer cells regardless of the p53 status, making it a particularly attractive agent for cancer cells that have become resistant to chemo- or radiotherapy through inactivation of p53.13 The combined use of TRAIL plus traditional chemotherapeutic agents or radiotherapy has been shown to enhance cancer-killing efficacies and restore the TRAIL sensitivity in previously TRAIL-resistant cells. Phase I and II clinical trials with rhTRAIL and agonist antibodies are ongoing.13 One important inhibitor of TRAIL-induced apoptosis is the FLICE-inhibitory protein (FLIP; CFLAR), which is predominantly expressed as long (FLIPL) and short (FLIPS) splice forms, and functions to inhibit caspase 8.14 Both isoforms of FLIP are recruited to the DISC, where they prevent the full activation of caspase 8 and inhibit apoptosis.14 FLIP has been found to be overexpressed in various cancers,15, 16 and FLIP overexpression has been shown to determine tumoral resistance to treatments with death ligands such as FasL and TRAIL.14 Conversely, downregulation of FLIP sensitizes previously resistant tumor cells to these ligands.14
NPC is relatively rare in Caucasians, but the disease is relatively common in the Southeastern region of China and in Taiwan. NPC is sensitive to radiation therapy, and more advanced cases may be treated with a combination of radio- and chemotherapy. Although the survival rate of NPC is ∼92% at 1 year and ∼50% at 5 years, 20–25% of patients display distant metastases after treatment.17 Thus, it is essential that we develop more sensitive and effective therapeutic strategies against NPC. In this study, we identified FLIP as a target gene of hnRNP K in NPC cells, and found that FLIP expression is regulated by the hnRNP K/nucleolin complex through direct interaction with a poly(C) sequence in the FLIP promoter. Finally, we found that FLIP overexpression was significantly correlated with hnRNP K overexpression and poor OS in NPC patients.
Results
Affymetrix analysis of hnRNP K targets
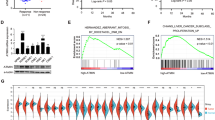
HnRNP K-mediated upregulation of TP can prevent hypoxia-induced apoptosis9 and has been associated with poor prognosis in NPC patients,5 suggesting that hnRNP K may function as an upstream regulator protein of cancer. To identify additional genes positively regulated by hnRNP K, we performed systematic microarray expression profiling using the Affymetrix HG U133 plus 2.0 chip (Affymetrix, Santa Clara, CA, USA) on samples representing NPC tumor tissues, adjacent normal tissues, control NPC-TW02 cells and hnRNP K-knockdown NPC-TWO2 cells. As shown in Figure 1a, 1945 genes were reduced in NPC-TW02 cells transiently transfected with hnRNP K siRNA versus those transfected with the negative control siRNA (cutoff value, 1.5-fold), whereas 5466 genes were elevated 1.5-fold or more in NPC tissues versus adjacent normal tissues. Comparison of these data sets revealed that 363 genes were elevated in NPC tissues and downregulated in hnRNP K-knockdown cells; these were considered potential targets for hnRNP K (Figure 1a). Ontological analysis of these 363 genes revealed that 20 and 26 of the genes reportedly contribute to cancer development-related antiapoptosis and migration, respectively (Supplementary Tables S1 and S2). Consistent with our previous findings,5, 9 TP was overexpressed in NPC tissues and downregulated by hnRNP K knockdown in NPC cells (Figure 1a).
FLIP, MMP13, ITGB6, Rad9B and Rap2A are potential targets of hnRNP K in NPC cells. (a) Systematic identification of genes upregulated by hnRNP K. The open circles represent genes whose expressions were reduced by at least 1.5-fold in hnRNP K-knockdown NPC-TW02 cells versus controls and increased by at least 1.5-fold in nine NPC tissues compared with pooled adjacent normal tissues, as assessed using an Affymetrix HG U133 plus 2.0 microarray set. The gray ellipses represent the genes that were both reduced by hnRNP K knockdown and increased in NPC tissues. The bottom panel shows the fold changes of the nine genes shown in the gray ellipses. (b) Changes in mRNA expression were assessed in cells treated with hnRNP K siRNA (K) or control siRNA (C). Forty-eight hours after siRNA transfection, cells were harvested for RNA preparation. Relative transcript amounts were measured by quantitative RT-PCR, and the results were normalized with regard to GAPDH expression. All data are presented as the mean±S.D. from at least three experiments. *P<0.05
To validate our results, we used quantitative RT-PCR to examine the mRNA expression levels of the apoptosis-related genes FLIP,14 DcR3,18 Rad9B19 and IL7R,20 and the migration-associated genes RANTES,21 MMP13,22 ITGB623 and Rap2A24 (Figure 1b). The mRNA levels of FLIP, MMP13, ITGB6, Rad9B and Rap2A were significantly reduced (to 0.2-, 0.4-, 0.4-, 0.3- and 0.8-fold, respectively) in hnRNP K-knockdown cells compared with control siRNA-transfected NPC-TW02 cells. As FLIP gene expression appeared to be reduced the most, we further investigated the regulation and antiapoptotic role of hnRNP K-mediated FLIP.
HnRNP K-mediated FLIP expression contributes to resistance against TRAIL-induced apoptosis in NPC cells
FLIP is an important inhibitor of TRAIL-induced apoptosis, and FLIP overexpression has been correlated with poor prognosis in colorectal cancer15 and bladder urothelial carcinoma.16 TRAIL is a ligand of the DR4 and DR5 death receptors, and these interactions can induce apoptosis in cancer cells.13 However, the role of hnRNP K in TRAIL-induced apoptosis has not as yet been established. To gain insight into the potential role of hnRNP K in TRAIL-induced apoptosis, we tested FLIP expression in hnRNP K-knockdown and control cells in the presence and absence of TRAIL in three NPC cell lines, NPC-TW01, -TW02 and -TW04. As shown in Figure 2a, the level of FLIP protein was reduced significantly in hnRNP K siRNA-treated NPC cells compared with cells treated with control siRNA regardless of TRAIL treatment. As typical apoptotic morphologies and cellular shrinkage were observed in TRAIL-treated hnRNP K-knockdown NPC-TW02 cells (Figure 2b, left panel), we further measured the effect of hnRNP K knockdown on TRAIL-induced apoptosis using Annexin V staining. As shown in Figure 2b (the bottom part of the left panel), TRAIL dose-dependently induced apoptosis in both hnRNP K-knockdown and control NPC-TW02 cells, but TRAIL-induced apoptosis was higher in hnRNP K-knockdown cells versus controls (34.2 versus 15.2% in 100 ng/ml-treated cells; P<0.05). Similar results were found in both NPC-TW01 and -TW04 cells (Figure 2b, right panel).
Prevention of TRAIL-induced apoptosis by hnRNP K through FLIP. (a) Effect of hnRNP K knockdown on FLIP expression in three NPC cell lines, NPC-TW01, -TW02 and -TW04. NPC cells transfected with hnRNP K (K) or control (C) siRNA were treated with or without 100 ng/ml TRAIL for 48 h. The levels of FLIP, hnRNP K and tubulin (loading control) protein were determined by western blotting. (b) Enhancement of TRAIL-induced apoptosis by hnRNP K knockdown. Morphology of NPC-TW02 cells transfected with hnRNP K or control siRNA, followed by treatment with or without 100 ng/ml TRAIL for 48 h. The bottom and right panels show the percentage of apoptotic cells in three NPC cell lines, NPC-TW01, -TW02 and -TW04, respectively, as assessed using Annexin V staining. All data are presented as the mean±S.D. from at least three experiments. *P<0.05. (c) Effect of ectopic FLIP on TRAIL-induced apoptosis in hnRNP K-knockdown cells. NPC-TW02 cells transfected with hnRNP K (K) or control (C) siRNA were transfected with constructs encoding FLAG-tagged FLIP or empty vector, and further treated with or without 100 ng/ml TRAIL for 48 h. The levels of FLIP, hnRNP K and tubulin (loading control) were determined by western blotting. All data are presented as the mean±S.D. from at least three experiments. *P<0.01. (d) The expression levels of the TRAIL receptors, DR4 and DR5, in hnRNP K-knockdown cells. NPC-TW02 cells transfected with hnRNP K (K) or control (C) siRNA were incubated for 48 h. The levels of DR4, DR5, hnRNP K and actin (loading control) were determined by western blotting
We next examined whether FLIP is involved in the enhancement of TRAIL-induced apoptosis in hnRNP K-knockdown cells. As shown in Figure 2c, TRAIL treatment significantly increased the proportion of apoptotic cells from 17.9% in control cells to 34.1% in hnRNP K-knockdown cells. However, the apoptotic effect of TRAIL was obviously abolished in cells transfected with a FLAG-tagged FLIP expression vector, with the percentage of apoptotic cells decreasing from 34.1 to 21.6%. As TRAIL has been shown to mediate apoptosis by binding to DR4 and DR5, we examined the expression of these death receptors in hnRNP K-knockdown and control NPC-TW02 cells. We found that the knockdown of hnRNP K did not affect the expression of DR4 or DR5 (Figure 2d). Taken together, our findings reveal that hnRNP K knockdown decreases the expression of FLIP but not DR4 and DR5, and enhances the sensitivity of NPC cells to TRAIL-induced apoptosis.
HnRNP K regulates the FLIP promoter directly through its poly(C) element
We next sought to determine the mechanism(s) underlying hnRNP K knockdown-mediated FLIP downregulation. Analysis of the FLIP promoter sequence revealed four potential hnRNP K-binding poly(C) or C-C-C-C elements, R1 to R4 (Figure 3a).1 To distinguish between transcriptional activation and post-transcriptional regulation, we examined the effect of hnRNP K knockdown on FLIP promoter activity and mRNA stability. We transfected a FLIP promoter reporter construct (−1179 to +281) into hnRNP K-knockdown NPC-TW02 cells and examined reporter activity 24 h later. HnRNP K knockdown significantly inhibited FLIP promoter activity by 54.3% compared with that in control cells (P<0.001) (Figure 3b). In contrast, the half-life of the FLIP mRNA was similar in control (8.4±1.8 h) and hnRNP K-knockdown (7.8±1.5 h) cells, as determined by the actinomycin D blockade of de novo RNA synthesis followed by the quantitative RT-PCR of FLIP mRNA levels at 1, 2, 4 and 8 h post-treatment (Figure 3c). These results suggest that the hnRNP K-mediated changes in FLIP gene expression are due to promoter inhibition, not mRNA destabilization.
The poly(C) element in the FLIP promoter is directly bound by hnRNP K. (a) Schematic of the FLIP promoter (−1179 to +281) containing four poly(C) sequences (C, −667 to −664, −600 to −597, −280 to −277 and −26 to −22). The numbers 1–4 indicate sequences R1–R4, as used for the DNA pull-down and ChIP assays. The arrow indicates the transcription start site. (b) Inhibition of FLIP promoter activity by hnRNP K knockdown. The pGL3-basic vector (pGL3, promoterless luciferase vector) and a FLIP promoter construct (−1179, encompassing nucleotides −1179 to +281) were transfected into NPC-TW02 cells 48 h after the cells had been transfected with control siRNA (C) or hnRNP K siRNA (K). Firefly and Renilla luciferase activities were determined 24 h later. The levels of hnRNP K were determined by western blotting; actin was measured as a loading control. *P<0.01 (c) The half-life of FLIP mRNA in hnRNP K-knockdown cells. The levels of FLIP mRNA in NPC-TW02 cells transfected with control siRNA (C) or hnRNP K siRNA (K) for 48 h were measured following treatment with actinomycin D for 1, 2, 4 and 8 h. (d) Binding of hnRNP K to a poly(C) element of the FLIP promoter in vitro. Probes encompassing the region shown in panel a were generated by PCR amplification using 5′ biotin-conjugated primers and templates from wild-type (for R1, R2, R3 and R4) and mutant (for R4mut.) FLIP promoters. DNA pull-down assays were performed with nuclear extracts isolated from NPC-TW02 cells. The hnRNP K levels of the immunoprecipitates and 2% inputs were determined by western blotting. (e) HnRNP K binds to the FLIP promoter sequence in vivo. Chromatin immunoprecipitation was performed using nuclear extracts from NPC-TW02 cells and an antibody against hnRNP K, followed by quantitative PCR of the sequence within the FLIP promoter region shown in panel c. Mouse IgG immunoprecipitation was performed as a negative control. (f) The poly(C) sequence is a responsive element for hnRNP K-mediated promoter activity. NPC-TW02 cells were pretreated with control siRNA (C) and hnRNP K siRNA (K) for 48 h and then transfected with pGL3-basic (pGL3), or FLIP promoter constructs −1179 (−1179 to +281), −37 (−37 to +281) or −37R4mut (−37 to +281) containing the mutant poly(C) sequence. Firefly and Renilla luciferase activities were determined at 24 h post-transfection. *P<0.01
To verify the ability of hnRNP K to bind to the predicted poly(C) elements, we performed DNA pull-down assays with R1, -2, -3 and -4 DNA-specific probes (Figure 3a). Our results showed that hnRNP K specifically bound to probe R4, but not to the other three probes (Figure 3d). Mutation of the poly(C) element in R4 (R4mut) abolished this binding, indicating that the interaction between hnRNP K and R4 is specific (Figure 3d). To further support our contention that hnRNP K can interact with the endogenous FLIP promoter, we performed a chromatin-immunoprecipitation analysis. As shown in Figure 3e, hnRNP K specifically immunoprecipitated with the R4 region of the FLIP promoter, but not with R1, R2 or R3.
To better delineate the function of the poly(C) element (−26 to −22) in R4 for FLIP promoter activity, we generated FLIP mini-promoter-reporter constructs (−37 to +281) with and without a mutation in the poly(C) element (−37R4mut), and transfected these constructs into hnRNP K-knockdown and control NPC-TW02 cells. As shown in Figure 3f, the wild-type promoter activity was significantly reduced (by 64.5%) in hnRNP K-knockdown versus control cells. In contrast, the poly(C) mutant promoter showed substantially reduced basal activity (to 48.6%) compared with the wild-type promoter, but was resistant to hnRNP K knockdown. Taken together, these results indicate that the poly(C) element (−26 to −22) is critical for hnRNP K-mediated FLIP promoter activity.
Identification of FLIP promoter poly(C) element-binding proteins
To identify other proteins involved in FLIP promoter regulation, proteins capable of binding to the mutant and wild-type poly(C) element (−26 to −22) were profiled using iTRAQ mass spectrometry (LTQ-Orbitrap, Thermo Electron, Bremen, Germany). Compared with the proteins that bound to the wild-type probe, 367 proteins showed 1.3-fold or larger reductions in the amount of DNA–protein complexes pulled down by the poly(C)-mutated R4 probe. The results of proteins that were previously known as hnRNP K-associated proteins or the factors predicted for having binding sites on FLIP promoter are shown in Table 1. Three of the differentially interacting proteins – nucleolin,2 hnRNP F25 and YB-126 – were previously reported to associate with hnRNP K. Two others, AP-2α and p300, were predicted to bind to R4 of the FLIP promoter based on a publicly available website for predicting transcription factor-binding elements (http://www.cbil.upenn.edu/cgi-bin/tess/tess). YY1 were also predicted to bind to elements within R4, but did not show differential binding with the wild-type versus mutated probes.
To further examine these candidate proteins, we used western blotting to analyze their protein levels in pulled-down DNA–protein complexes, and found that the bindings of AP-2α and nucleolin to probe R4 were markedly reduced to 0.2- and 0.5-fold, respectively, by the poly(C) mutation (Figure 4a). We then tested whether AP-2α and nucleolin formed a protein complex with hnRNP K. As shown in Figure 4b, nucleolin and hnRNP K, but not AP-2α, were present in immunoprecipitates separately generated using hnRNP K- and nucleolin-specific antibodies. When we used an anti-AP-2α antibody, neither nucleolin nor hnRNP K were detected in the immunoprecipitates. These results suggest that hnRNP K formed a protein complex with nucleolin, but not with AP-2α. These results collectively suggest that nucleolin and AP-2α may bind to the FLIP promoter in an hnRNP K-dependent and -independent manner, respectively.
Validation of the FLIP promoter poly(C) element-binding proteins identified by iTRAQ. (a) Binding of AP-2α and nucleolin to the FLIP promoter poly(C) element. Proteins enriched from NPC-TW02 nuclear extracts by DNA pull-down with wild-type (WT) and poly(C)-mutated (MU) R4 probes were analyzed by western blotting. YY1 was detected as an internal control. The level of MU relative to WT is indicated. * Below the level of detection. (b) HnRNP K and nucleolin interacting with each other, but not with AP-2α. HnRNP K, nucleolin and AP-2α, were immunoprecipitated from NPC-TW02 nuclear extracts and detected by western blotting using specific antibodies. (c) Inhibition of FLIP promoter activity by nucleolin and AP-2α knockdown. The pGL3-basic vector and FLIP promoter constructs (encompassing nucleotides −1179 or −37 to +281) were transfected into NPC-TW02 cells 48 h after the cells had been transfected with control siRNA or siRNA against hnRNP K, nucleolin or AP-2α. Firefly and Renilla luciferase activities were determined at 24 h post-transfection. The levels of FLIP, hnRNP K, nucleolin, AP-2α and actin (loading control) protein were determined by western blotting. (d) Effect of nucleolin and AP-2α knockdown on FLIP expression. Forty-eight hours after siRNA transfection, the levels of FLIP proteins and mRNA were determined by western blotting and quantitative RT-PCR, respectively. The amounts of FLIP protein and mRNA were normalized with respect to the levels of actin and GAPDH, respectively, and the resulting values from the control siRNA treatment were set as 1.0. *P<0.01. (e) Effect of nucleolin and AP-2α knockdown on TRAIL-induced apoptosis. The percentage of apoptotic cells of NPC-TW02 cells transfected with hnRNP K, nucleolin, AP-2α or control siRNA, followed by treatment with or without 100 ng/ml TRAIL for 48 h, as assessed using Annexin V staining. All data are presented as the mean±S.D. from at least three experiments. *P<0.05
Nucleolin and AP-2α are responsible for FLIP gene regulation
Next, we investigated whether hnRNP K, nucleolin and AP-2α have direct effects on FLIP promoter activity, hnRNP K-, nucleolin- and AP-2α-knockdown. NPC-TW02 cells were separately transfected with reporter constructs corresponding to the FLIP mini promoter, and reporter activity was examined after 24 h (Figure 4c). Our results showed that hnRNP K, nucleolin and AP-2α knockdown dramatically inhibited FLIP promoter activity by 62.1 (P<0.01), 73.8 (P<0.01) and 51.1% (P<0.01), respectively. To further test whether FLIP expression could be physiologically regulated by these three proteins, FLIP protein and mRNA levels were examined in NPC-TW02 cells transfected with siRNA directed against hnRNP K, nucleolin and AP-2α. As shown in Figure 4d, FLIP mRNA levels were significantly reduced to 0.36-, 0.61- and 0.78-fold in cells transfected with siRNA against hnRNP K, nucleolin and AP-2α, respectively, compared with cells transfected with the control siRNA. Similarly, FLIP protein levels were significantly reduced to 0.1-, 0.3- and 0.8-fold in cells transfected with siRNA against hnRNP K, nucleolin and AP-2α, respectively (Figure 4d). We further assessed the impact of nucleolin and AP-2α knockdown on TRAIL-induced apoptosis in NPC-TW02 cells. As shown in Figure 4e, nucleolin knockdown significantly enhanced TRAIL-induced apoptosis (28.4%) compared with the cells transfected with control siRNA (15.8%). On the other hand, AP-2α knockdown had lesser effect (21.2%) compared with hnRNP K or nucleolin knockdown. Our results suggest that hnRNP K, nucleolin and AP-2α may be involved in the transcriptional activation of the FLIP promoter. hnRNP K and nucleolin knockdown significantly downregulate FLIP expression and subsequently enhance TRAIL-induced apoptosis.
Elevated FLIP correlates with hnRNP K in NPC tissues
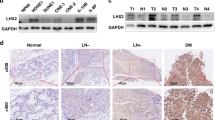
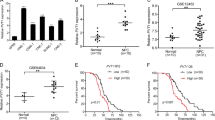
No previous study has examined FLIP expression in NPC tissues. Here, we examined 67 NPC biopsies by immunohistochemical staining with an anti-FLIP antibody. As shown in Figures 5a and b, FLIP was highly expressed in tumor cells, whereas only relatively weak staining was seen in the adjacent normal nasopharyngeal epithelium. Forty of the tumor specimens (60%; Figure 5g) showed highly intense FLIP staining and were designated ‘FLIP high’ (Figure 5c), whereas the remaining 27 specimens (40%) showed weaker staining intensities (Figure 5e) and were designated ‘FLIP low.’ Representative specimens are shown at lower magnification ( × 200) in Supplementary Figure S1.
Correlation of FLIP with hnRNP K and overall survival in NPC patients. (a) Immunohistochemical staining of FLIP in NPC specimens. Representative images of NPC specimens containing tumor (T) and adjacent nontumor (N) cells stained with a specific anti-FLIP antibody are shown at × 100 magnification. The T and N areas are shown at × 400 magnification in (b), in the upper and lower panels, respectively. Consecutive NPC tissue sections were stained using anti-hnRNP K and anti-FLIP antibodies, and were evaluated immunohistochemically. (c) and (d) Tumor cells with high-level FLIP and hnRNP K expression are shown. (e) and (f) Tumor cells with relatively weak expression of FLIP and hnRNP K are shown at × 400 magnification. Scale bar, 100 μm. (g) Correlation of FLIP and hnRNP K expression. * Statistically significant by χ2-test. (h) Kaplan–Meier survival analysis of OS as a function of elevated FLIP expression in NPC patients. FLIP high, n=40 and FLIP low, n=27
To examine the physiological role of hnRNP K in FLIP expression in primary NPC samples, we further analyzed the correlation between the levels of FLIP expression and the levels of hnRNP K expression in NPC tissues. Consecutive tissue slides of the same set of specimens were subjected to immunohistochemistry (IHC) using an anti-hnRNP K antibody. As shown in Figure 5, 30 specimens (45%) showed high levels of hnRNP K (Figure 5d), whereas 37 specimens (55%) showed low levels of hnRNP K (Figure 5f). Statistical analysis using the Pearson χ2-test showed that high-level hnRNP K was positively correlated with high-level FLIP expression (Figure 5g; P=0.002). These results strongly suggest that FLIP expression is positively regulated by hnRNP K in NPC tumor cells.
Association of FLIP with OS
To evaluate the prognostic significance of FLIP under current therapeutic protocols, we subjected the same retrospective cohort of 67 NPC patients to a clinical outcome assessment study. No significant correlations were found between high-level FLIP expression and clinicopathological features, including age, gender, tumor stage, node stage, AJCC stage and chemotherapy (Supplementary Table S3). A Kaplan–Meier survival analysis was then carried out to determine the OS for patients as a function of FLIP expression. As shown in Figure 5h, there was a significant difference in OS between patients with high and low levels of FLIP expression (P=0.015). We next conducted a multivariate analysis of FLIP with age, gender, tumor stage, node stage and chemotherapy, and found that high FLIP expression (P=0.005) was an independent prognostic predictor of poor OS (Table 2).
Discussion
Overexpression of hnRNP K is found in various cancers and has been correlated with poor prognosis.3, 4, 5, 6 In this study, we showed a new function for hnRNP K in FLIP regulation and resistance to TRAIL-induced apoptosis. The poly(C) sequence at −26 to −22 bp upstream of the FLIP transcription start site is bound by the hnRNP K/nucleolin complex, which is responsible for transcriptional activation. Moreover, induction of FLIP by hnRNP K increases the resistance of NPC cells to TRAIL-induced apoptosis. Importantly, high-level FLIP expression was found to be correlated with poor prognosis and increased expression of hnRNP K in NPC patients. Our findings collectively show that hnRNP K and nucleolin form a complex on the FLIP promoter, thereby inducing FLIP expression through transcriptional activation. This provides a mechanistic explanation for the correlation between FLIP and hnRNP K in NPC. In addition, FLIP may serve as an independent prognostic and therapeutic marker for NPC.
HnRNP K is overexpressed in various cancers, and this overexpression has been correlated with poor prognosis3, 4, 5, 6 suggesting that hnRNP K contributes to tumor progression and malignancy. Recent work has shown that hnRNP K regulates antiapoptosis and cell migration, two biological functions that are critically related to cancer development.9, 11 In this study, we identified 46 genes involved in apoptosis and migration as being overexpressed in NPC and upregulated by hnRNP K. Our differentially expressed gene set included TP9 and ITGB6,27 along with 44 other genes that had not been previously reported as hnRNP K targets. Among these differentially expressed genes, FLIP,15, 16 FGF2,28 TP,5 SULF229 and MMP1330 are known to be overexpressed in various cancers and have been associated with poor prognosis. Thus, our findings suggest that several genes downstream of hnRNP K are overexpressed in cancer and may promote tumor development.
FLIP is an antiapoptotic molecule critical to TRAIL-induced apoptosis.14 Consistent with this antiapoptotic function of FLIP, we found that hnRNP K-mediated FLIP expression is necessary and sufficient to maintain resistance to TRAIL-induced apoptosis. Although FLIP is known to be regulated at the translational and post-translational levels14, 31 we herein show that hnRNP K can directly bind to the poly(C) element of the FLIP promoter, transcriptionally activating FLIP and consequently preventing apoptosis. Although transcription of the FLIP gene is also regulated by NF-κB,32 c-Myc,33 nuclear factor of activated T cells,34 androgen receptor response element35 and c-Fos,36 we found that hnRNP K is indispensable for the transcriptional activation of FLIP. Previous studies have shown that hnRNP K can regulate the expression of TP9 and gastrin2 post-transcriptionally. However, we did not find evidence that hnRNP K affects the stability of FLIP mRNA (Figure 3c), although FLIP mRNA could be regulated by IRF8 through mRNA stabilization.37 Instead, similar to the hnRNP K-mediated induction of c-myc, Src and eIF4E gene expression through binding of poly(C) elements in their gene promoters,1 we herein show that the poly(C) element of the FLIP promoter is important for its recognition and activation by both hnRNP K and nucleolin.
NF-κB has a major role in the transcriptional activation of multiple antiapoptotic genes during cancer progression. The prototypical NF-κB complex is a p65/p50 heterodimer that is important for NF-κB-mediated antiapoptotic effects.38 In the absence of appropriate stimuli, NF-κB is sequestered in the cytoplasm by IκBα protein. On stimulation by factors such as TNFα, IκBα protein is degraded by proteasome, leading to NF-κB nuclear translocation and transcriptional activation.39 Although hnRNP K knockdown-enhanced TRAIL-induced apoptosis is correlated with the loss of FLIP expression, we assess the impact of hnRNP K knockdown on NF-κB activation. As shown in Supplementary Figure S2a and S2b, TNFα stimulation induced nuclear translocation of p65 and p50, as well as downregulation of IκBα, which were not affected by hnRNP K knockdown. In addition, TNFα treatment had no effect on the survival of NPC-TW02 cells regardless of hnRNP K knockdown (Supplementary Figure S2c). These results together suggest that hnRNP K knockdown-enhanced TRAIL-induced apoptosis resulted from the loss of FLIP expression, rather than from the alteration of NF-κB activation.
Nucleolin is a multifunctional protein involved in ribosome biogenesis,40 transcriptional regulation40 and mRNA stability.2 Nucleolin is an abundant protein, but its overexpression has been correlated with poor prognosis in various cancers.40 Currently, a guanine-rich aptamer of nucleolin, AS1411, is in phase II clinical trials as an anticancer drug for acute myeloid leukemia and renal cell carcinoma.40 Recent work has shown that nucleolin can bind to a guanine-rich DNA sequence upstream of the c-myc promoter (nt −142 to −115) and that it can function as a transcriptional repressor in MCF10A cells.41 Other studies have revealed that a cytosine-rich DNA sequence corresponding to the same region of the c-myc promoter can also be bound by hnRNP K, leading to transcriptional activation in HeLa cells.12 Both nucleolin and hnRNP K favor the binding of single-strand DNA over double-strand (ds)DNA.41, 42 We suggest that these previous findings seem to indicate that hnRNP K and nucleolin may recognize the same promoter site, where they appear to function as cofactors for transcriptional regulation. Consistent with this hypothesis, nucleolin and hnRNP K can form a cytoplasmic protein complex that acts to stabilize gastrin mRNA.2 Here, we show for the first time that hnRNP K and nucleolin can function as cofactors for promoter activation.
TRAIL-based systems, many of which are currently undergoing preclinical trials, are thought to have high therapeutic potential because TRAIL induces apoptosis in a broad range of tumor cells, spares normal cells, and does not appear to be associated with systemic toxicity.13 However, various cancer cells can resist TRAIL-induced apoptosis by overexpressing FLIP. It is hoped that the development of agents capable of inhibiting FLIP expression could sensitize cancer cells resistant to TRAIL, thereby recovering the efficacy of TRAIL-based cancer therapies in resistant tumors.14 This study, which provides insight into the induction of FLIP by hnRNP K and nucleolin, may aid in the development of new agents capable of inhibiting FLIP expression. In particular, we suggest that the nucleolin-targeting aptamer, AS1411, could prove useful for sensitizing cells to TRAIL-induced apoptosis through inhibition of FLIP expression.
In conclusion, we show herein that hnRNP K and nucleolin exert an antiapoptotic function by inducing the antiapoptotic protein, FLIP, through binding to the poly(C) element of the FLIP promoter and inducing transcriptional activation. FLIP is overexpressed in NPC and is associated with poor prognosis. We believe that high-level expression of hnRNP K, nucleolin and FLIP could be prognostic for TRAIL resistance in NPC patients. Finally, we propose that hnRNP K, nucleolin and FLIP should all be considered potential targets for developing new anticancer agents.
Materials and Methods
Quantitative RT-PCR
RNA samples from NPC tissues and NPC-TW02 cells were isolated using the TRIzol reagent (Invitrogen, Carlsbad, CA, USA). Reverse transcription of RNA (1 μg) was performed using ImProm-II (Promega, Madison, WI, USA) and oligo(dT)15 primers (Promega). The primers used to amplify the cDNA corresponding to FLIP, RANTES, IL7R, MMP13, ITGB6, DcR3, Rad9B, TP, Rap2A and GAPDH are presented in Supplementary Table S4. Quantitative RT-PCR was performed on a Light-Cycler (Roche Diagnostics), according to the manufacturer’s instructions, using the FastStart DNA Master SYBR Green I reagent (Roche Diagnostics, Mannheim, Germany). The gene expression results were normalized with regard to the expression of GADPH. For mRNA half-life assessment, actinomycin D (5 μg/ml) was added 48 h after transfection with control or hnRNP K-targeting siRNA (see below), and RNA was prepared at the indicated times.
RNA interference
SMARTpool reagents, including four 21-bp RNA duplexes targeting hnRNP K, AP-2α and nucleolin, respectively, were purchased from Dharmacon (Lafayette, CO, USA), and the negative control siRNA was synthesized by Research Biolabs Ayer Rajah Industrial Estate (Research Biolabs, Singapore, Singapore). The oligonucleotide sequences are presented in Supplementary Table S5. NPC cells were transfected with 50 nmol/l dsRNA duplexes and 50 μg dsRNA transfection reagents (TransIT-TKO; Mirus Bio Corporation, Madison, WI, USA) according to the manufacturer’s protocol. At 6 h post-transfection, the siRNA-containing medium was replaced with fresh complete medium. After a further 48 h of culture, cells were harvested, and cell extracts were prepared and subjected to western blotting to confirm target gene knockdown.
DNA pull-down
Probes corresponding to the four potential binding elements (R1-R4) were generated by PCR using appropriate biotinylated primers and the FLIP promoter construct (−1179 to +281) as a template. The specificity and requirement of the poly(C) nucleotides within the four probes for hnRNP K binding were further determined using a poly(C) mutant probe that was PCR-generated from a template that had been mutated by site-directed mutagenesis. The biotinylated probes were conjugated with M-280 Streptavidin Dynabeads (Invitrogen) in binding buffer (10 mM Tris-HCl [pH 7.5], 50 mM KCl, 1 mM MgCl2, 1 mM EDTA (pH 8.0), 1 mM Na3VO4, 5 mM DTT, 5% glycerol and 0.3% NP-40) for 40 min at room temperature. NPC-TW02 cells were extracted using the Compartmental Protein Extraction reagent (Millipore, Billerica, MA, USA), and nuclear fractions (50 μg) were incubated with unconjugated Dynabeads (Invitrogen) in the presence of 25 μg/ml poly (dI:dC) for 20 min at room temperature. The unbound fraction was incubated with 250 μg of Dynabeads bound to 50 pmol of immobilized wild-type or mutant FLIP promoter for 1 h at room temperature. The Dynabead-bound complexes were collected using a Dynal MPC-S magnetic particle concentrator (Dynal, Lake Success, NY, USA) and washed with binding buffer. The DNA-bound proteins were eluted in SDS sample buffer and assayed by western blotting.
Chromatin immunoprecipitation
NPC-TW02 cells were crosslinked by treatment with 1% formaldehyde for 10 min at room temperature, and the reactions were quenched with glycine (0.125 M) for 10 min at room temperature. Cell pellets were incubated with RSB buffer (10 mM Tris-HCl (pH 7.4), 10 mM NaCl, 3 mM MgCl2 and protease inhibitors) for 10 min at 4 °C, an equal volume of RSB buffer containing 0.4% NP-40 was added, and samples were incubated at 4 °C for an additional 10 min. The samples were then centrifuged at 500 g for 5 min at 4 °C, the supernatants were removed, and the nuclear pellets were washed with RSB buffer and SNSB buffer (10 mM Tris-HCl (pH 7.4), 1 M NaCl, 1 mM EDTA, 0.1% NP-40 and protease inhibitors). The nuclei were incubated with RIPA buffer (50 mM Tris-HCl (pH 8.0), 150 mM NaCl, 5 mM EDTA, 0.5% deoxycholate, 0.1% SDS, 1% NP-40 and protease inhibitors). The resulting nuclear fractions were sonicated on ice and diluted 10-fold in WCE buffer (20 mM HEPES, 1 mM EDTA, 1 mM EGTA, 0.2 M NaCl, 0.5% Triton X-100, 10% glycerol and protease inhibitors). The diluted samples were centrifuged at 13 000 r.p.m. for 15 min at 4 °C. Approximately 500 μg of the resulting chromatin solution was used for immunoprecipitation. Samples were precleared with 30 μl of 50/50 salmon sperm DNA/protein A agarose slurry (Millipore, cat. no. 16–157C) for 1 h at 4 °C. The precleared samples were incubated overnight at 4 °C with 5 μg of an anti-hnRNP K antibody (Invitrogen). Mouse IgG1 was used as a control antibody. The immunocomplexes were collected with 30 μl of 50/50 salmon sperm DNA/protein A agarose slurry for 1 h at 4 °C. The beads were washed with a 9 : 1 WCE/RIPA buffer, washed with Tris-EDTA, and then resuspended in 100 μl Tris-EDTA. An equal volume of 2 × PK buffer (200 mM NaCl, 1% SDS and 600 μg/ml proteinase K) was added to the immunoprecipitates and the input samples, and all samples were incubated for 3 h at 56 °C. The samples were then de-crosslinked by overnight incubation at 65 °C. DNA fragments were purified using a QIAquick PCR purification kit (Qiagen, Hilden, Germany), and quantitative PCR was performed using the appropriate primers against the four potential hnRNP K-binding elements.
Patients, clinical staging, treatment and assessment of clinical outcome
The retrospective cohort comprised 67 NPC patients who had been admitted to Chang Gung Memorial Hospital (CGMH; Lin-Kou) from 1990 to 1999. Clinical stage was defined according to the 2002 cancer staging system revised by the American Joint Committee on Cancer. Histological typing was done according to the World Health Organization (WHO) classification criteria, as previously described.5 This study was reviewed and approved by the institutional review board and ethics committee of CGMH. Informed consent was obtained from all patients. All enrolled patients had been treated with definitive radiotherapy (cumulative dose of external beam radiotherapy >R64.8 Gy). Among them, 16 patients had received additional chemotherapy from the Department of Medical Oncology at CGMH. Patients diagnosed with distant metastatic disease at presentation (M1 stage) and/or those who had undergone previous treatment at another institute were excluded from this study. For all enrolled patients, pathology reports were obtained from pathologic databases and medical records, and reviewed for confirmation of the NPC diagnosis (reviewed by C Hsueh). Information on stage, treatment and follow-up were collected from hospital tumor registries and medical files, as was limited information on family history. Patient characteristics and clinical features are summarized in Supplementary Table S3. The median age at diagnosis was 46 years (range, 25.5–76.9), and the male-to-female ratio was ∼3.8 : 1. The clinicopathological features were comparable between patient subgroups classified according to FLIP status. The primary end point was OS, which was calculated from the date of diagnosis to the date of death or the last follow-up.
Immunohistochemical staining
Immunohistochemical analyses were performed using an automatic IHC-staining device, according to the manufacturer’s instructions (Bond-max Automated Immunostainer; Vision Biosystems, Melbourne, Australia). Tissue sections were retrieved using Bond Epitope Retrieval Solution 1 (Vision BioSystems) and stained with antibodies against hnRNP K (mouse monoclonal antibody, 1 : 300 dilution; Santa Cruz Biotechnology, Santa Cruz, CA, USA) and FLIP (rabbit polyclonal antibody, 1 : 100 dilution; Santa Cruz Biotechnology). A polymer detection system (Bond Polymer Refine; Vision BioSystems) was used to reduce nonspecific staining. Tissue sections were treated with liquid DAB reagent using 3′-diaminobenzidine tetrahydrochloride as the chromogen and hematoxylin as the counterstaining reagent. For analysis of hnRNP K expression, specimens in which >50% of the tumor cells displayed strong staining were defined as having a ‘high-level’ hnRNP K expression, and those where ⩽50% of tumor cells stained strongly were defined as having ‘low-level’ hnRNP K expression. For analysis of FLIP expression, specimens in which >50% of tumor cells displayed positive staining were defined as having ‘high-level’ FLIP expression, and those in which ⩽50% tumor cells displayed positive staining were defined as having ‘low-level’ FLIP expression. FLIP- and hnRNP K-positive tumor cells in representative microscopic fields were scored independently by two experienced pathologists.
Statistical analysis
All statistical analyses were performed using the SPSS 13.0 statistical software package (SPSS Inc., Chicago, IL, USA). The data were analyzed using Student’s t-test. Differences were considered significant at a level of P<0.05.
Abbreviations
- hnRNP K:
-
heterogeneous nuclear ribonucleoprotein K
- NPC:
-
nasopharyngeal carcinoma
- FLICE:
-
inactive procaspase 8
- DISC:
-
death-inducing signaling complex
References
Choi HS, Hwang CK, Song KY, Law PY, Wei LN, Loh HH . Poly(C)-binding proteins as transcriptional regulators of gene expression. Biochem Biophys Res Commun 2009; 380: 431–436.
Lee PT, Liao PC, Chang WC, Tseng JT . Epidermal growth factor increases the interaction between nucleolin and heterogeneous nuclear ribonucleoprotein K/poly(C) binding protein 1 complex to regulate the gastrin mRNA turnover. Mol Biol Cell 2007; 18: 5004–5013.
Barboro P, Repaci E, Rubagotti A, Salvi S, Boccardo S, Spina B et al. Heterogeneous nuclear ribonucleoprotein K: altered pattern of expression associated with diagnosis and prognosis of prostate cancer. Br J Cancer 2009; 100: 1608–1616.
Matta A, Tripathi SC, DeSouza LV, Grigull J, Kaur J, Chauhan SS et al. Heterogeneous ribonucleoprotein K is a marker of oral leukoplakia and correlates with poor prognosis of squamous cell carcinoma. Int J Cancer 2009; 125: 1398–1406.
Chen LC, Hsueh C, Tsang NM, Liang Y, Chang KP, Hao SP et al. Heterogeneous ribonucleoprotein k and thymidine phosphorylase are independent prognostic and therapeutic markers for nasopharyngeal carcinoma. Clin Cancer Res 2008; 14: 3807–3813.
Carpenter B, McKay M, Dundas SR, Lawrie LC, Telfer C, Murray GI . Heterogeneous nuclear ribonucleoprotein K is over expressed, aberrantly localised and is associated with poor prognosis in colorectal cancer. Br J Cancer 2006; 95: 921–927.
Habelhah H, Shah K, Huang L, Ostareck-Lederer A, Burlingame AL, Shokat KM et al. ERK phosphorylation drives cytoplasmic accumulation of hnRNP-K and inhibition of mRNA translation. Nat Cell Biol 2001; 3: 325–330.
Notari M, Neviani P, Santhanam R, Blaser BW, Chang JS, Galietta A et al. A MAPK/HNRPK pathway controls BCR/ABL oncogenic potential by regulating MYC mRNA translation. Blood 2006; 107: 2507–2516.
Chen LC, Liu HP, Li HP, Hsueh C, Yu JS, Liang CL et al. Thymidine phosphorylase mRNA stability and protein levels are increased through ERK-mediated cytoplasmic accumulation of hnRNP K in nasopharyngeal carcinoma cells. Oncogene 2009; 28: 1904–1915.
Lynch M, Chen L, Ravitz MJ, Mehtani S, Korenblat K, Pazin MJ et al. hnRNP K binds a core polypyrimidine element in the eukaryotic translation initiation factor 4E (eIF4E) promoter, and its regulation of eIF4E contributes to neoplastic transformation. Mol Cell Biol 2005; 25: 6436–6453.
Inoue A, Sawata SY, Taira K, Wadhwa R . Loss-of-function screening by randomized intracellular antibodies: identification of hnRNP-K as a potential target for metastasis. Proc Natl Acad Sci USA 2007; 104: 8983–8988.
Michelotti EF, Michelotti GA, Aronsohn AI, Levens D . Heterogeneous nuclear ribonucleoprotein K is a transcription factor. Mol Cell Biol 1996; 16: 2350–2360.
Wang S . The promise of cancer therapeutics targeting the TNF-related apoptosis-inducing ligand and TRAIL receptor pathway. Oncogene 2008; 27: 6207–6215.
Yang JK . FLIP as an anti-cancer therapeutic target. Yonsei Med J 2008; 49: 19–27.
Ullenhag GJ, Mukherjee A, Watson NF, Al-Attar AH, Scholefield JH, Durrant LG . Overexpression of FLIPL is an independent marker of poor prognosis in colorectal cancer patients. Clin Cancer Res 2007; 13: 5070–5075.
Korkolopoulou P, Goudopoulou A, Voutsinas G, Thomas-Tsagli E, Kapralos P, Patsouris E et al. c-FLIP expression in bladder urothelial carcinomas: its role in resistance to Fas-mediated apoptosis and clinicopathologic correlations. Urology 2004; 63: 1198–1204.
Lee AW, Poon YF, Foo W, Law SC, Cheung FK, Chan DK et al. Retrospective analysis of 5037 patients with nasopharyngeal carcinoma treated during 1976–1985: overall survival and patterns of failure. Int J Radiat Oncol Biol Phys 1992; 23: 261–270.
Pitti RM, Marsters SA, Lawrence DA, Roy M, Kischkel FC, Dowd P et al. Genomic amplification of a decoy receptor for Fas ligand in lung and colon cancer. Nature 1998; 396: 699–703.
Komatsu K, Miyashita T, Hang H, Hopkins KM, Zheng W, Cuddeback S et al. Human homologue of S. pombe Rad9 interacts with BCL-2/BCL-xL and promotes apoptosis. Nat Cell Biol 2000; 2: 1–6.
Namen AE, Lupton S, Hjerrild K, Wignall J, Mochizuki DY, Schmierer A et al. Stimulation of B-cell progenitors by cloned murine interleukin-7. Nature 1988; 333: 571–573.
Schall TJ, Bacon K, Toy KJ, Goeddel DV . Selective attraction of monocytes and T lymphocytes of the memory phenotype by cytokine RANTES. Nature 1990; 347: 669–671.
Stringa E, Knauper V, Murphy G, Gavrilovic J . Collagen degradation and platelet-derived growth factor stimulate the migration of vascular smooth muscle cells. J Cell Sci 2000; 113 (Part 11): 2055–2064.
Huang X, Wu J, Spong S, Sheppard D . The integrin alphavbeta6 is critical for keratinocyte migration on both its known ligand, fibronectin, and on vitronectin. J Cell Sci 1998; 111 (Part 15): 2189–2195.
McLeod SJ, Li AH, Lee RL, Burgess AE, Gold MR . The Rap GTPases regulate B cell migration toward the chemokine stromal cell-derived factor-1 (CXCL12): potential role for Rap2 in promoting B cell migration. J Immunol 2002; 169: 1365–1371.
Wei CC, Zhang SL, Chen YW, Guo DF, Ingelfinger JR, Bomsztyk K et al. Heterogeneous nuclear ribonucleoprotein K modulates angiotensinogen gene expression in kidney cells. J Biol Chem 2006; 281: 25344–25355.
Shnyreva M, Schullery DS, Suzuki H, Higaki Y, Bomsztyk K . Interaction of two multifunctional proteins. Heterogeneous nuclear ribonucleoprotein K and Y-box-binding protein. J Biol Chem 2000; 275: 15498–15503.
Eiring AM, Neviani P, Santhanam R, Oaks JJ, Chang JS, Notari M et al. Identification of novel posttranscriptional targets of the BCR/ABL oncoprotein by ribonomics: requirement of E2F3 for BCR/ABL leukemogenesis. Blood 2008; 111: 816–828.
Donnem T, Al-Shibli K, Al-Saad S, Busund LT, Bremnes RM . Prognostic impact of fibroblast growth factor 2 in non-small cell lung cancer: coexpression with VEGFR-3 and PDGF-B predicts poor survival. J Thorac Oncol 2009; 4: 578–585.
Lai JP, Sandhu DS, Yu C, Han T, Moser CD, Jackson KK et al. Sulfatase 2 up-regulates glypican 3, promotes fibroblast growth factor signaling, and decreases survival in hepatocellular carcinoma. Hepatology 2008; 47: 1211–1222.
Gu ZD, Li JY, Li M, Gu J, Shi XT, Ke Y et al. Matrix metalloproteinases expression correlates with survival in patients with esophageal squamous cell carcinoma. Am J Gastroenterol 2005; 100: 1835–1843.
Zhang HG, Wang J, Yang X, Hsu HC, Mountz JD . Regulation of apoptosis proteins in cancer cells by ubiquitin. Oncogene 2004; 23: 2009–2015.
Micheau O, Lens S, Gaide O, Alevizopoulos K, Tschopp J . NF-kappaB signals induce the expression of c-FLIP. Mol Cell Biol 2001; 21: 5299–5305.
Ricci MS, Jin Z, Dews M, Yu D, Thomas-Tikhonenko A, Dicker DT et al. Direct repression of FLIP expression by c-myc is a major determinant of TRAIL sensitivity. Mol Cell Biol 2004; 24: 8541–8555.
Zaichuk TA, Shroff EH, Emmanuel R, Filleur S, Nelius T, Volpert OV . Nuclear factor of activated T cells balances angiogenesis activation and inhibition. J Exp Med 2004; 199: 1513–1522.
Gao S, Lee P, Wang H, Gerald W, Adler M, Zhang L et al. The androgen receptor directly targets the cellular Fas/FasL-associated death domain protein-like inhibitory protein gene to promote the androgen-independent growth of prostate cancer cells. Mol Endocrinol 2005; 19: 1792–1802.
Zhang X, Zhang L, Yang H, Huang X, Otu H, Libermann TA et al. c-Fos as a proapoptotic agent in TRAIL-induced apoptosis in prostate cancer cells. Cancer Res 2007; 67: 9425–9434.
Yang D, Wang S, Brooks C, Dong Z, Schoenlein PV, Kumar V et al. IFN regulatory factor 8 sensitizes soft tissue sarcoma cells to death receptor-initiated apoptosis via repression of FLICE-like protein expression. Cancer Res 2009; 69: 1080–1088.
Karin M, Cao Y, Greten FR, Li ZW . NF-kappaB in cancer: from innocent bystander to major culprit. Nat Rev Cancer 2002; 2: 301–310.
Ghosh S, May MJ, Kopp EB . NF-kappa B and Rel proteins: evolutionarily conserved mediators of immune responses. Annu Rev Immunol 1998; 16: 225–260.
Bates PJ, Laber DA, Miller DM, Thomas SD, Trent JO . Discovery and development of the G-rich oligonucleotide AS1411 as a novel treatment for cancer. Exp Mol Pathol 2009; 86: 151–164.
Gonzalez V, Guo K, Hurley L, Sun D . Identification and characterization of nucleolin as a c-myc G-quadruplex-binding protein. J Biol Chem 2009; 284: 23622–23635.
Tomonaga T, Levens D . Heterogeneous nuclear ribonucleoprotein K is a DNA-binding transactivator. J Biol Chem 1995; 270: 4875–4881.
Acknowledgements
Grant support was provided by the Ministry of Education, Taiwan (to Chang Gung University), the National Science Council, Taiwan (Grants NSC 97–2320-B-182–001-MY3 and 98–3112-B-182–006 to Y-S. Chang; NSC 99–2321-B-182–005-MY2 to L-C. Chen) and the Chang Gung Memorial Hospital, Taiwan (Grant CMRPD160053 to Y-S. Chang). We thank the Pathology Core of the Chang Gung Molecular Medicine Research Center for technical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Edited by JP Medema
Supplementary Information accompanies the paper on Cell Death and Differentiation website
Supplementary information
Rights and permissions
About this article
Cite this article
Chen, LC., Chung, IC., Hsueh, C. et al. The antiapoptotic protein, FLIP, is regulated by heterogeneous nuclear ribonucleoprotein K and correlates with poor overall survival of nasopharyngeal carcinoma patients. Cell Death Differ 17, 1463–1473 (2010). https://doi.org/10.1038/cdd.2010.24
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cdd.2010.24
Keywords
This article is cited by
-
Inactivation of the tight junction gene CLDN11 by aberrant hypermethylation modulates tubulins polymerization and promotes cell migration in nasopharyngeal carcinoma
Journal of Experimental & Clinical Cancer Research (2018)
-
hnRNPK inhibits GSK3β Ser9 phosphorylation, thereby stabilizing c-FLIP and contributes to TRAIL resistance in H1299 lung adenocarcinoma cells
Scientific Reports (2016)
-
Cytoplasmic hnRNPK interacts with GSK3β and is essential for the osteoclast differentiation
Scientific Reports (2015)
-
Matrix metalloproteinase 12 is induced by heterogeneous nuclear ribonucleoprotein K and promotes migration and invasion in nasopharyngeal carcinoma
BMC Cancer (2014)
-
Uncovering a key to the process of metastasis in human cancers: a review of critical regulators of anoikis
Journal of Cancer Research and Clinical Oncology (2013)