Abstract
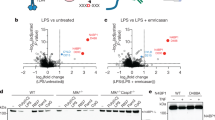
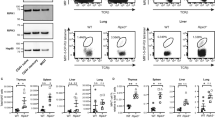
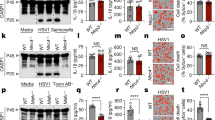
The immune system consists of two evolutionarily different but closely related responses, innate immunity and adaptive immunity. Each of these responses has characteristic receptors—Toll-like receptors (TLRs) for innate immunity and antigen-specific receptors for adaptive immunity. Here we show that the caspase recruitment domain (CARD)-containing serine/threonine kinase Rip2 (also known as RICK, CARDIAK, CCK and Ripk2)1,2,3,4 transduces signals from receptors of both immune responses. Rip2 was recruited to TLR2 signalling complexes after ligand stimulation. Moreover, cytokine production in Rip2-deficient cells was reduced on stimulation of TLRs with lipopolysaccharide, peptidoglycan and double-stranded RNA, but not with bacterial DNA, indicating that Rip2 is downstream of TLR2/3/4 but not TLR9. Rip2-deficient cells were also hyporesponsive to signalling through interleukin (IL)-1 and IL-18 receptors, and deficient for signalling through Nod proteins—molecules also implicated in the innate immune response. Furthermore, Rip2-deficient T cells showed severely reduced NF-κB activation, IL-2 production and proliferation on T-cell-receptor (TCR) engagement, and impaired differentiation to T-helper subtype 1 (TH1) cells, indicating that Rip2 is required for optimal TCR signalling and T-cell differentiation. Rip2 is therefore a signal transducer and integrator of signals for both the innate and adaptive immune systems.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Inohara, N., del Peso, L., Koseki, T., Chen, S. & Nunez, G. RICK, a novel protein kinase containing a caspase recruitment domain, interacts with CLARP and regulates CD95-mediated apoptosis. J. Biol. Chem. 273, 12296–12300 (1998).
McCarthy, J. V., Ni, J. & Dixit, V. M. RIP2 is a novel NF-κB-activating and cell death-inducing kinase. J. Biol. Chem. 273, 16968–16975 (1998).
Thome, M. et al. Identification of CARDIAK, a RIP-like kinase that associates with caspase-1. Curr. Biol. 8, 885–888 (1998).
Medzhitov, R. & Janeway, C. Jr. Innate immune recognition: mechanisms and pathways. Immunol. Rev. 173, 89–97 (2000).
Kelliher, M. A. et al. The death domain kinase RIP mediates the TNF-induced NF-κB signal. Immunity 8, 297–303 (1998).
Poltorak, A. et al. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science 282, 2085–2088 (1998).
Takeuchi, O. et al. Differential roles of TLR2 and TLR4 in recognition of gram-negative and gram-positive bacterial cell wall components. Immunity 11, 443–451 (1999).
Hemmi, H. et al. A Toll-like receptor recognizes bacterial DNA. Nature 408, 740–745 (2000).
Alexopoulou, L., Holt, A. C., Medzhitov, R. & Flavell, R. A. Recognition of double-stranded RNA and activation of NF-κB by Toll-like receptor 3. Nature 413, 732–738 (2001).
Demuth, A., Goebel, W., Beuscher, H. U. & Kuhn, M. Differential regulation of cytokine and cytokine receptor mRNA expression upon infection of bone marrow-derived macrophages with Listeria monocytogenes. Infect. Immun. 64, 3475–3483 (1996).
Humke, E. W., Shriver, S. K., Starovasnik, M. A., Fairbrother, W. J. & Dixit, V. M. ICEBERG: a novel inhibitor of interleukin-1β generation. Cell 103, 99–111 (2000).
Medzhitov, R. et al. MyD88 is an adaptor protein in the hToll/IL-1 receptor family signaling pathways. Mol. Cell 2, 253–258 (1998).
Kawai, T., Adachi, O., Ogawa, T., Takeda, K. & Akira, S. Unresponsiveness of MyD88-deficient mice to endotoxin. Immunity 11, 115–122 (1999).
Bertin, J. et al. Human CARD4 protein is a novel CED-4/Apaf-1 cell death family member that activates NF-κB. J. Biol. Chem. 274, 12955–12958 (1999).
Inohara, N. et al. Nod1, an Apaf-1-like activator of caspase-9 and nuclear factor-κB. J. Biol. Chem. 274, 14560–14567 (1999).
Ogura, Y. et al. Nod2, a Nod1/Apaf-1 family member that is restricted to monocytes and activates NF-κB. J. Biol. Chem. 276, 4812–4818 (2001).
Inohara, N., Ogura, Y., Chen, F. F., Muto, A. & Nunez, G. Human nod1 confers responsiveness to bacterial lipopolysaccharides. J. Biol. Chem. 276, 2551–2554 (2001).
Ogura, Y. et al. A frameshift mutation in NOD2 associated with susceptibility to Crohn's disease. Nature 411, 603–606 (2001).
Girardin, S. E. et al. CARD4/Nod1 mediates NF-κB and JNK activation by invasive Shigella flexneri. EMBO Rep. 2, 736–742 (2001).
Growney, J. D. & Dietrich, W. F. High-resolution genetic and physical map of the Lgn1 interval in C57BL/6J implicates Naip2 or Naip5 in Legionella pneumophila pathogenesis. Genome Res. 10, 1158–1171 (2000).
Hugot, J. P. et al. Association of NOD2 leucine-rich repeat variants with susceptibility to Crohn's disease. Nature 411, 599–603 (2001).
Miceli-Richard, C. et al. CARD15 mutations in Blau syndrome. Nature Genet. 29, 19–20 (2001).
Yang, J., Zhu, H., Murphy, T. L., Ouyang, W. & Murphy, K. M. IL-18-stimulated GADD45-β required in cytokine-induced, but not TCR-induced, IFN-γ production. Nature Immunol. 2, 157–164 (2001).
Leitenberg, D. & Bottomly, K. Regulation of naive T cell differentiation by varying the potency of TCR signal transduction. Semin. Immunol. 11, 283–292 (1999).
Ruland, J. et al. Bcl10 is a positive regulator of antigen receptor-induced activation of NF-κB and neural tube closure. Cell 104, 33–42 (2001).
Sun, Z. et al. PKC-Θ is required for TCR-induced NF-κB activation in mature but not immature T lymphocytes. Nature 404, 402–407 (2000).
Aronica, M. A. et al. Preferential role for NF-κ B/Rel signaling in the type 1 but not type 2 T cell-dependent immune response in vivo. J. Immunol. 163, 5116–5124 (1999).
Celada, A., Gray, P. W., Rinderknecht, E. & Schreiber, R. D. Evidence for a γ-interferon receptor that regulates macrophage tumoricidal activity. J. Exp. Med. 160, 55–74 (1984).
Kobayashi, K., Hatano, M., Otaki, M., Ogasawara, T. & Tokuhisa, T. Expression of a murine homologue of the inhibitor of apoptosis protein is related to cell proliferation. Proc. Natl Acad. Sci. USA 96, 1457–1462 (1999).
Acknowledgements
We thank C. L. Stewart and M. Schnare for providing reagents; L. Evangelisti for technical assistance; and F. Manzo and G. Chenell for manuscript preparation. This work was supported by grants from the National Institutes of Health (G.N., J.G., C.A.J., R.M. and R.A.F) and from the Howard Hughes Medical Institute (C.A.J., R.M. and R.A.F.). K.K. is a recipient of a Postdoctoral Fellowship of the Cancer Research Institute; L.D.H. was supported by a fellowship from the Cancer Research Fund of the Damon Runyon-Walter Winchel Foundation; and R.A.F. and C.A.J are Investigators and R.M. is an Assistant Investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Kobayashi, K., Inohara, N., Hernandez, L. et al. RICK/Rip2/CARDIAK mediates signalling for receptors of the innate and adaptive immune systems. Nature 416, 194–199 (2002). https://doi.org/10.1038/416194a
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/416194a
This article is cited by
-
OTUD1 ameliorates cerebral ischemic injury through inhibiting inflammation by disrupting K63-linked deubiquitination of RIP2
Journal of Neuroinflammation (2023)
-
Microbial ligand-independent regulation of lymphopoiesis by NOD1
Nature Immunology (2023)
-
Nod1-dependent NF-kB activation initiates hematopoietic stem cell specification in response to small Rho GTPases
Nature Communications (2023)
-
The role of diosgenin in crohn’s disease
Clinical Phytoscience (2022)
-
A pancancer analysis of the carcinogenic role of receptor-interacting serine/threonine protein kinase-2 (RIPK2) in human tumours
BMC Medical Genomics (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.