Abstract
Cerebral hyperperfusion, anemia and hypertension are common in patients with end-stage renal disease (ESRD). Young ESRD adults might afford a better hemodynamic tolerance; however, their cerebral vascular disorders are often overlooked. This phase-contrast MRI study investigated relationships between cerebral blood flow (CBF), anemia and hypertension in young adults undergoing hemodialysis (HD). Blood flows, velocities and cross-sectional areas of bilateral internal carotid arteries and vertebral arteries were quantified on phase maps in 33 patients and 27 healthy controls. Cerebral oxygen delivery (COD) and vascular resistance were (CVR) were computed based on CBF, hemoglobin and mean arterial pressure (MAP). We found strong correlations among hemoglobin, MAP and CBF. Hemoglobin rather than MAP was directly related to CBF. COD was negatively related to MAP, while CVR was positively related to hemoglobin. The cross-sectional areas of arteries were increased which were directly associated with hemoglobin rather than MAP. HD patients were of elevated CBF, decreased COD and unchanged CVR. Although elevated CBF compensated anemia-induced hypoxia, COD of these patients was still lower. Anemia directly contributed to elevated CBF and hypertension affected CBF through anemia. Unaffected CVR of young patients probably indicated that they could maintain basic functions of cerebral circulation under multiple risk factors.
Similar content being viewed by others
Introduction
Altered cerebral autoregulation is common in uremic hypertension1. By local vasomotor adjustments in cerebral vascular resistance (CVR), cerebral autoregulation mechanism keeps CBF relatively constant to ensure tight coupling between oxygen supply and brain oxygen demand in healthy subjects2. In end-stage renal disease (ESRD) patients, elevated cerebral blood flow (CBF) had been repeatedly reported3,4,5,6. Anemia-induced hypoxia caused low cerebral oxygen delivery (COD) which was thought to play a key role in cerebral hyperperfusion in ESRD patients5. To make up low oxygen supply, CBFs of ESRD patients were increased to maintain basic oxidative metabolisms4,5. Hypertension may also contribute to cerebral circulation disorders in ESRD patients. Coinciding with hypertension, low arterial wall distensibility and increased CVR have been reported in ESRD patients7,8. It seems that anemia and hypertension are coupled risk factors of cerebral hyperperfusion in ESRD patients. However, the relationships among them are still unclear.
The average age of patients with ESRD in China is much younger than United State and Japan9. About 90% ESRD patients undergoing dialysis received hemodialysis (HD)9. And hence, there is a large population of young ESRD adults undergoing HD. Young HD patients might afford a better hemodynamic tolerance than elder ones. However, their cerebral circulation disorders are often overlooked because of their age. In this study, we aim to expore the relationships among anemia, hypertension and cerebral hyperperfusion in young HD patients. Also, COD and CVR were quantified to identify oxygen supply and vascular reaction in these patients, respectively. The relationships between changes in cerebral circulation and their risk factors and the patterns of CBF, COD and CVR may improve our knowledge about impaired cerebral autoregulation in HD patients.
Results
Clinical and laboratory data
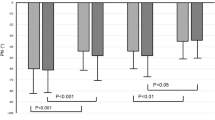
The clinical and laboratory data of the subjects in this study are shown in Table 1. The age and gender were not significantly different between HD patients and healthy controls (both P > 0.05). Our patients were suffered from serious hypertension and their systolic and diastolic blood pressures were significantly higher than those of healthy subjects (both P < 0.001). Anemia was commonly found in HD patients who had significantly lower hemoglobin compared with healthy subjects (P < 0.001).
Changes in tCBF, CVR and COD
Table 2 illustrates the total CBF (tCBF), CVR and COD of HD and healthy groups. HD patients had significantly higher tCBF compared with healthy subjects (P < 0.001, Bonferroni corrected). Although increased tCBF can help deliver more oxygen to brain, HD patients had significantly lower total COD compared with healthy subjects (P < 0.001, Bonferroni corrected) because of their severe anemia. There was no difference in CVR between HD patients and healthy controls (P = 0.79; Bonferroni corrected).
Correlation results
There were strong relationships between tCBF, mean arterial pressure (MAP) and hemoglobin (Pearson correlation; all P < 0.001; Bonferroni corrected; Fig. 1A–C). Decreased COD was correlated with elevated MAP (Pearson correction; r = −0.368, P = 0.019; Bonferroni corrected; Fig. 1D). CVR was positively correlated with hemoglobin (Pearson correlation; r = 0.353, P = 0.028; Bonferroni corrected; Fig. 1E). Factoring out the contribution of MAP, CBF was negatively correlated with hemoglobin (Partial correlation; r = −0.743, P < 0.001; Bonferroni corrected; Fig. 2A). However, CBF was not related to MAP after controlling hemoglobin (Partial correlation; r = −0.0827, P > 1; Bonferroni corrected; Fig. 2B). Significant partial correlation was still observed between MAP and hemoglobin while controlling CBF (Partial correlation; r = −0.501, P < 0.001; Bonferroni corrected; Fig. 2C).
The Pearson cross correlations among cerebral brain flow, oxygen delivery, vascular resistance, hemoglobin and mean arterial pressure of young end-stage renal disease adults undergoing hemodialysis.
(A) Cerebral blood flow and mean arterial pressure; (B) Cerebral blood flow and hemoglobin; (C) Mean arterial pressure and hemoglobin; (D) Cerebral oxygen delivery and mean arterial pressure; (E) Cerebral vascular resistance and hemoglobin. Note: Correlation p values were Bonferroni corrected.
The partial correlations among cerebral blood flow, hemoglobin and mean arterial pressure.
(A) Cerebral blood flow and hemoglobin controlling mean arterial pressure; (B) Cerebral blood flow and mean arterial pressure controlling hemoglobin; (C) Hemoglobin and mean arterial pressure controlling cerebral blood flow. Note: Correlation p values were Bonferroni corrected.
Table 3 shows the blood flows, velocities and cross-sectional areas of four feeding arterials of HD and control groups. The blood flows of bilateral internal carotid arteries (ICAs) and right vertebral artery (VA) significantly increased in HD patients compared with controls (All P < 0.01, FDR corrected). The average flow velocity of HD patients significantly increased in bilateral VAs (Both P < 0.01, FDR corrected), but not in bilateral ICAs (Both P > 0.6, FDR corrected). The maximum, minimum and average cross-sectional areas of bilateral ICAs were significantly larger in HD patients compared with controls (All P < 0.05, FDR corrected), whereas the maximum, minimum and average cross-sectional areas of bilateral VAs were not different between two groups (All P > 0.4, FDR corrected). The total cross-sectional area of the four feeding arterials of HD patients was significantly greater than that of healthy subjects (P = 0.009, FDR corrected).
Pearson cross correlations showed that both hemoglobin and MAP were related with blood flows, velocities and cross-sectional areas of feeding arterials (Table 4). Factoring out the contribution of MAP, hemoglobin was still negatively correlated with blood flows and average velocities of four arterials (Partial correlations; all P < 0.05; FDR corrected; Table 4), with peak velocity of bilateral VAs (Partial correlations; all P < 0.01; FDR corrected; Table 4) and with the cross-sectional area of left ICA. But, MAP was not correlated with any of hemodynamic measurements of four feeding arterials after controlling the contribution of hemoglobin (Partial correlations; all P > 0.05; FDR corrected; Table 4).
The serum calcium levels of our patients (2.21 ± 0.16 mmol/L) were nearly normal (normal range: 2.02–2.60 mmol/L), whereas their phosphorus levels (2.04 ± 0.47 mmol/L) were higher than normal level (normal range: 0.81–1.65 mmol/L). There were significant correlations in HD patients between calcium and tCBF or hemoglobin (Pearson correlations; r = −0.44, P = 0.01 and r = 0.45, P = 0.009, respectively; uncorrected). No correlation was found among patients between their MRI measurements and their hemodialysis duration, phosphorus, or calcium*phosphorus (Pearson correlations; all P > 0.05; uncorrected).
Discussion
This study found that there were strong correlations among elevated MAP, decreased hemoglobin and elevated CBF in young ESRD HD adults. Hemoglobin level was negatively correlated with CBF, whereas MAP was not related to CBF after controlling hemoglobin. Moreover, we found decreased COD and unchanged CVR of HD group compared with controls. Increased CBFs can be attributed to dilated bilateral ICAs and increased flow velocity of bilateral VAs. The relationships among anemia, hypertension and CBF and the patterns of changes in CBF, COD and CVR provide us a better understanding about the abnormal autoregulation mechanism in young ESRD HD adults.
Elevated CBF was found in ESRD HD patients, which directly attributed to anemia rather than hypertension. Increased CBF in CKD patients has been repeatedly reported by multimodality imaging studies, such as 133Xe inhalation technique3, 15O positron emission tomography4,5, arterial spin labeling MRI6 and Doppler ultrasonography10. Our findings based on pcMRI were consistent with previous studies3,4,5,6,10, supporting that pcMRI technique could provide reliable results. Specially, the measured blood velocity and blood flow were not sensitive to T1 or T2 of the spins because pcMRI utilizes the phase of an image to encode the velocity of moving spins. Anemia in ESRD patients was mainly caused by the declined erythropoietin production11,12. Low hemoglobin tended to reduce both blood viscosity and oxygen supply. However, the change in viscosity per se did not result in the hypoxia-induced increase in CBF13. Thus, brain tissue hypoxia caused by anemia could be the most important factor of elevated CBF in ESRD patients5. Similar with anemia, hypertension is also common in ESRD patients. Coinciding with renal dysfunction, the renin–angiotensin–aldosterone-system can be activated, which has been implicated in pathogenesis of hypertension14. Although MAP was correlated with CBF, it was not related to CBF after controlling hemoglobin, supporting that hypertension was not a direct risk factor of elevated cerebral hyperperfusion. Thus, the correlation between MAP and CBF could be caused by the strong correlation between hemoglobin and CBF.
Low COD played an important role in cerebral vascular changes in HD patients. Low oxygen saturation15 and low oxygen supply5 of ESRD patients had been reported. In our study, COD was lower in HD patients because of their severe anemia. Increased CBF would deliver more arterial blood to the brain, however, such a compensational procedure was not sufficient to make up hypoxia in ESRD patients4. Hypoxia could reduce the amount of ATP which causes ATP-gated ion channels in smooth muscle cells to open and hyperpolarize16. Hyperpolarization could reduce contractile ability, which could induce arteries to dilate. New et al.1 reported that vessel diameter was significantly greater in uremic Wistar-Kyoto rats compared with normotensive controls. In this study, we observed a significant increase of total cross-sectional arterial area and negative correlation between hemoglobin level and arterial area, indicating that anemia-induced hypoxia might cause cerebral vascular changes to deliver more blood to compensate the oxygen limitation. Also, we observed negative correlation between COD and MAP, supporting that HD patients with severe hypoxia might suffer from more serious hypertension.
Unchanged CVR of HD patients could be an outcome of anemia and vasodilation. Heyman et al. reported increased CVR in patients with uremia8. Our finding was not consistent with theirs, possibly because our patients were much younger and had more severe renal disease compared with their patients. CVR was reported to relate with degree of anemia17. We also observed such a positive correlation between CVR and hemoglobin in anemic patients. Low hemoglobin level might be associated with low blood viscosity18. Besides, we detected vasodilation in bilateral ICAs. According to the law of Poiseuille, unchanged CVR in HD patients can be explained by the decreased viscosity and increased vessel diameter. Young HD patients in our study had unchanged CVRs and free of severe cerebrovascular disease, supporting that young HD adults might maintain basic functional of cerebral circulation system under multiple risks of vascular disease.
The mechanisms of elevated blood flow were different between ICA and VA. Bilateral VAs increased flow velocities and kept arterial areas unchanged to support elevated blood flow. Our findings about elevated velocities of VAs were consistent with one previous study based on Doppler sonography10. However, the cross-sectional areas of VAs were not significantly changed in our study, which was not consistent with theirs10. Our patients were much younger and had much higher blood pressure than theirs. It seemed that the cross-sectional areas of VAs were restricted by the cervical vertebrae or by the transverse foramen, which can limit the further dilatation of VAs. Elevated velocities of bilateral VAs were associated with increased MAP, indicating that blood flows of VAs were increased through elevated velocities. Being different from VAs, bilateral ICAs increased blood flow through arterial dilatation rather than increased velocities. Both maximal and minimal cross-sectional areas of bilateral ICAs were significantly increased, leading to a significantly increased total cross-sectional area of feeding arterials.
Young ESRD HD adults were of high risks of cerebrovascular disease. Abnormalities in calcium and phosphorus metabolism played an important factor of causing vascular calcification19. To improve bone metabolism as well as to prevent progression of vascular calcification, the calcium containing phosphate binders were generally provided to HD patients in our hospital. Coinciding with high serum phosphorus concentrations and a high calcium–phosphorus ion product in serum, vascular calcification was common and progressive in young adults with ESRD undergoing dialysis20. Our patients were of abnormal high phosphorus and normal calcium levels, indicating that they would have much higher risk of vascular calcification. The positive relation between serum calcium level and CBF were found in our patients. The dialysate calcium concentration for HD patients is generally adjusted to optimize calcium and phosphate balance. The coupling between calcium and CBF could be caused by the adjustment dialysate calcium concentration. Further study is needed to find the mechanism of this coupling.
We acknowledge that there were several limitations in this study. First, only young ESRD HD adults were included in this study. Different dialysis modalities for ESRD patients could affect their cerebral circulation system in different ways. In future studies, patients treated by peritoneal dialysis, non-dialysis patients with ESRD as well as patients undergoing renal transplantation should be included to verify our findings. Second, individual intracranial pressure (ICP) was not available in this study. ICP was generally much lower than MAP and was ignored in the definition of CVR17. In our study, no medical record showed that young ESRD HD patients had significantly increased ICP. All our subjects were free of encephalopathy and brain injury. And hence, the ignored ICP term in the CVR definition would not significantly affect the conclusion about CVR. Third, the arterial oxygen saturation was assumed to be 98% in our study. The arterial oxygen saturation was not affected in HD patients4. Thus, our assumption about the arterial oxygen saturation would not significantly affect our findings.
Materials and Methods
Subjects
This prospective study was approved by the Jinling hospital Medical Research Ethics Committee and all experimental protocols were performed in accordance with relevant guidelines and regulations. All participants gave written consent forms before MRI scans. Thirty-three young ESRD adults undergoing HD (23 males, 10 females, age from 18 to 35 years, mean age 26.6 ± 5.4 years) were recruited in this study. Patient including criteria were: young adults ageing from 18 to 35 years old, no clinical symptoms of encephalopathy and no any MRI contraindications. Twenty-seven age- and gender- matched healthy subjects (18 males, 9 females, age from 21 to 35 years, mean age 25.0 ± 2.6 years) were recruited from local community. All healthy subjects had no diseases affecting brain functions. No drug abuse history was reported. Abdominal ultrasound scans revealed no abnormal findings for all healthy subjects.
Brachial artery blood pressure of each subject was measured by an automatic sphygmomanometer (Omron HEM 1000, Omron electronics LLC, Japan). The measurements were performed at rest before MRI examination and the average value of three consecutive measurements over ten minutes was computed. All subjects took routine blood test right after MRI examination. HD patients took additional blood biochemistry tests, including serum creatinine level, calcium and phosphorus concentrations.
Imaging acquisition
All the MRI examinations were performed on a clinical 3T whole-body MR scanner (Siemens TIM Trio, Siemens Medical Solutions, Erlangen, Germany). High resolution T1-weighted images were acquired by the 3D MPRAGE sequence for the localization of pcMRI and for individual CBF quantification. The cardiac-triggered pcMRI scans were positioned perpendicular to the feeding arteries. The imaging parameters of pcMRI were as follows: field of view = 200 * 200 * 5 mm3, based resolution = 256, single slice, TR = 38.15 ms, TE = 3.47 ms, flip angle = 25 deg, segments = 3, velocity encoding = 120 cm/s, bandwidth = 543 Hz/Px. All subjects were instructed to stay still and not think about anything with eyes closed during MRI scans.
Quantification of tCBF, CVR and COD
The tCBF is measured by the pcMRI technique applied at the four feeding arteries (i.e. bilateral ICAs and bilateral VAs at the bottom of the brain. In this paper, the blood flow (BF, in unit ml/s) of each artery is quantified on the phase image of pcMRI images by the Siemens’s product software Argus. Firstly, both magnitude and phase series are loaded into Argus Viewer. Secondly, the contour of each artery is drawn on the first magnitude image (Fig. 3A), which will be also created on the phase image (Fig. 3B). Thirdly, the four contours of the feeding arterials are auto adjusted and then propagated within slice. The first image is discarded to reduce anthropogenic influence. Finally, the results about velocities, flows and cross-sectional areas of arteries are reported in the summary table of Argus.
The magnitude and phase images of phase contrast MRI.
(A) The magnitude image; (B) The phase image. Notes: Four regions of interests were drawn on the magnitude image. R1/R2 = right/left internal carotid artery; R3/R4 = right/left vertebral artery. The circles in R1-R4 illustrate the voxels with maximal velocities.
The tCBF (in unit  ) is calculated by summing the blood flows of the four feeding arteries via:
) is calculated by summing the blood flows of the four feeding arteries via:

where  ,
,  ,
,  and
and  are the blood flow velocities of left ICA, right ICA, left VA and right VA, respectively.
are the blood flow velocities of left ICA, right ICA, left VA and right VA, respectively.  in Eq. (1) represents the total weight of a given brain and is used to adjust tCBF. Suppose that the density of the brain tissue is 1.06 g/ml, the brain weight can be calibrated by
in Eq. (1) represents the total weight of a given brain and is used to adjust tCBF. Suppose that the density of the brain tissue is 1.06 g/ml, the brain weight can be calibrated by

where  is the total volume of gray and white matter which are obtained by partitioning the 3D T1 weighted image based on SPM8 (Statistical Parametric Mapping, http://www.fil.ion.ucl.ac.uk/spm/).
is the total volume of gray and white matter which are obtained by partitioning the 3D T1 weighted image based on SPM8 (Statistical Parametric Mapping, http://www.fil.ion.ucl.ac.uk/spm/).
CVR can be calculated as follows:

where  represents mean arterial pressure.
represents mean arterial pressure.  is calculated by
is calculated by

where  and
and  are diastolic and systolic pressure, respectively.
are diastolic and systolic pressure, respectively.
Generally, the amount contributed by dissolved oxygen in plasma is very small. In this paper, the oxygen dissolved in plasma is omitted. Suppose that 1.0 g of hemoglobin binds 1.39 ml oxygen21, COD can be computed by:

where  represents the arterial oxygen saturation. Because
represents the arterial oxygen saturation. Because  is known to be relatively stable, it is assumed to be 98%.
is known to be relatively stable, it is assumed to be 98%.
Statistical analysis
Statistical analyses were performed by the software IBM SPSS statistics (version 22). The tCBF, COD and CVR were analyzed with an ANCOVA model to detect hemodynamic differences between HD patients and controls. Age and gender were included in ANCOVA as nuisance covariates. Correlations between aforementioned global measurements and MAP or hemoglobin were computed based on Pearson cross correlation. Partial correlation was performed among tCBF, MAP and hemoglobin to control the interactions among them. To control the multiple competitions among tCBF, COD and CVR, a Bonferroni-corrected P less than 0.05 was considered as significant.
One-way ANCOVA was performed to compare pcMRI measurements between two groups. Correlations between pcMRI measurements and MAP or hemoglobin were calculated based on Pearson cross correlation and partial correlation. The false discovery rate (FDR) was applied to correct the multiple comparisons among multiple pcMRI measurements. An FDR-corrected P value less than 0.05 was considered as significant.
Correlations were also performed within HD patients to explore the relationships between the MRI measurements and HD duration, calcium, phosphorus, or the product of calcium and phosphorus. The significance level was set at a p value less than 0.05 without correction.
Additional Information
How to cite this article: Zheng, G. et al. Anemia rather than hypertension contributes to cerebral hyperperfusion in young adults undergoing hemodialysis: A phase contrast MRI study. Sci. Rep. 6, 22346; doi: 10.1038/srep22346 (2016).
References
New, D. I., Chesser, A. M., Thuraisingham, R. C. & Yaqoob, M. M. Cerebral artery responses to pressure and flow in uremic hypertensive and spontaneously hypertensive rats. Am. J. Physiol. Heart Circ. Physiol. 284, H1212–1216 (2003).
Spilt, A. et al. Reproducibility of total cerebral blood flow measurements using phase contrast magnetic resonance imaging. J. Magn. Reson. Imaging. 16, 1–5 (2002).
Mathew, R. J., Rabin, P., Stone, W. J. & Wilson W. H. Regional cerebral blood flow in dialysis encephalopathy and primary degenerative dementia. Kidney Int. 28, 64–68 (1985).
Hirakata, H. et al. CBF and oxygen metabolism in hemodialysis patients: effects of anemia correction with recombinant human EPO. Am. J. Physiol. 262, F737–743 (1992).
Kuwabara Y. et al. Cerebral blood flow and vasodilatory capacity in anemia secondary to chronic renal failure. Kidney Int. 61, 564–569 (2002).
Papoiu, A. D. et al. Voxel-based morphometry and arterial spin labeling fMRI reveal neuropathic and neuroplastic features of brain processing of itch in end-stage-renal-disease. J. Neurophysiol. 112, 1729–1738 (2014).
Groothoff, J. W. et al. Increased arterial stiffness in young adults with end-stage renal disease since childhood. J. Am. Soc. Nephrol. 13, 2953–2961 (2002).
Heyman, A., Patterson, J. L. & Jones, R. W. Cerebral circulation and metabolism in uremia. Circulation. 3, 558–563 (1951).
Liu, Z. H. Nephrology in China. Nat. Rev. Nephrol. 9, 523–528 (2013).
Haktanir, A. et al. Doppler sonographic evaluation of cerebral blood flow in anemia resulting from chronic renal failure. J. Ultrasound Med. 24, 947–952 (2005).
Fisher, J. W. Mechanism of the anemia of chronic renal failure. Nephron. 25, 106–111 (1980).
Bonomini, M. & Sirolli, V. Uremic toxicity and anemia. J. Nephrol. 16, 21–28 (2003).
Waschke, K. F. et al. Lack of dependence of cerebral blood flow on blood viscosity after blood exchange with a Newtonian O2 carrier. J. Cereb. Blood Flow Metab. 14, 871–876 (1994).
Frohlich, E. D. Renin–angiotensin system inhibition improves coronary flow reserve in hypertension. J. Cardiovasc. Pharmacol. 37, S35–9 (2001).
Prohovnik, I. et al. Cerebrovascular effects of hemodialysis in chronic kidney disease. J. Cereb. Blood Flow Metab. 27, 1861–1869 (2007).
Choi, Y. K. & Kim, K. W. Blood-neural barrier: its diversity and coordinated cell-to-cell communication. BMB Rep. 41, 345–352 (2008).
Scheinberg, P. Cerebral blood flow and metabolism in pernicious anemia. Blood. 6, 213–227 (1951).
Wood, J. H., Simeone, F. A., Kron, R. E. & Snyder, L. L. Experimental hypervolemic hemodilution: physiological correlations of cortical blood flow, cardiac output and intracranial pressure with fresh blood viscosity and plasma volume. Neurosurgery. 14, 709–723 (1984).
Mitsnefes, M. M. et al. Cardiac and vascular adaptation in pediatric patients with chronic kidney disease: role of calcium-phosphorus metabolism. J. Am. Soc. Nephrol. 16, 2796–2803 (2005).
Goodman, W. G. et al. Coronary-artery calcification in young adults with end-stage renal disease who are undergoing dialysis. N. Engl. J. Med. 342, 1478–1483 (2000).
Ibaraki, M. et al. Interindividual variations of cerebral blood flow, oxygen delivery and metabolism in relation to hemoglobin concentration measured by positron emission tomography in humans. J. Cereb. Blood Flow Metab. 30, 1296–1305 (2010).
Acknowledgements
This work was supported by grants from National Natural Science Foundation of China (grant No. 81471644 and 81101039 to G.Z., grant No. 81570681 to J.W., grants No. 81322020, 81230032 and 81171313 to L.J.Z.), the Program for New Century Excellent Talents in the University (NCET-12-0260 to L.J.Z.) and the China Postdoctoral Science Foundation (grants No. 2014T71014 and 2015M570436 to G.Z.).
Author information
Authors and Affiliations
Contributions
G.Z. was involved in literature review, data collection, analysis of MRI data and writing of the manuscript. J.W. contributed to the clinical data collection. W.Y. contributed to the laboratory data collection. X.L. contributed to the measurement of blood pressures. Z.Z. contributed to recruitment of ESRD patients and healthy controls. H.C. contributed to the MRI data collection. X.K. contributed to the MRI data collection. S.L. was involved in the MRI data collection. X.J. was involved in MRI data analysis. Y.L. was involved in the analysis of MRI data. Z.Z. contributed to the design of the pcMRI study. L.J.Z. contributed in the experimental design and revision of the manuscript. G.M.L. was involved in the experimental design and in the writing process. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Zheng, G., Wen, J., Yu, W. et al. Anemia rather than hypertension contributes to cerebral hyperperfusion in young adults undergoing hemodialysis: A phase contrast MRI study. Sci Rep 6, 22346 (2016). https://doi.org/10.1038/srep22346
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep22346
This article is cited by
-
Association between white matter hyperintensities and altered cerebral blood flow in maintenance hemodialysis patients: a longitudinal study
BMC Nephrology (2024)
-
Blunted cerebral hemodynamic responses to incremental exercise in patients with end-stage renal disease
International Urology and Nephrology (2024)
-
Relations of hippocampal subfields atrophy patterns with memory and biochemical changes in end stage renal disease
Scientific Reports (2023)
-
Altered resting-state functional networks in patients with hemodialysis: a graph-theoretical based study
Brain Imaging and Behavior (2021)
-
Increased cerebral blood flow is correlated with neurocognitive impairment in long-term hemodialysis patients: an arterial spin labeling MRI study
Brain Imaging and Behavior (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.