Abstract
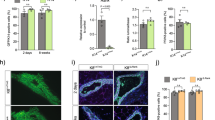
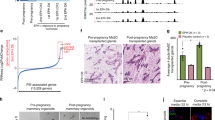
A parity-induced mammary population, marked by β-galactosidase expression conditionally activated through cre-lox recombinase originates in WAP-Cre/Rosa-lox-STOP-lox-LacZ (WAP-Cre/Rosa-LacZ) female mice during pregnancy, lactation and involution. During subsequent pregnancies, these parity-induced mammary epithelial cells (PI-MEC) proliferated to produce new secretory acini composed of secretory luminal cells and myoepithelium. In serial transplantation assays, PI-MEC were able to self-renew over several transplant generations and to contribute significantly to the resulting mammary outgrowths. In limiting dilution transplantation, they proliferated to produce both luminal and myoepithelial cells, comprised both lobule-limited and duct-limited epithelial outgrowths, and differentiated into all the cellular subtypes recognized in murine mammary epithelium. TGF-β1 expression from the whey acidic protein promoter (WAP) in triply transgenic females did not prevent the appearance of PI-MEC after pregnancy despite the absence of full lactation or their ability to proliferate and produce progeny with diverse cellular fates in situ upon subsequent pregnancies. However, in transplants from triple transgenic parous females, the WAP-TGF-β1-positive PI-MEC did not contribute to the newly recapitulated mammary outgrowths, suggesting that they were incapable of expansive cellular proliferation (self-renewal). This result is consistent with our earlier publication that WAP-TGF-β1 expression in mammary epithelium induces premature stem cell senescence in mammary transplants and decreases mammary cancer risk in mouse mammary tumor virus (MMTV)-infected females even after multiple pregnancies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Boulanger CA and Smith GH . (2001). Oncogene, 20, 2264–2272.
Callahan R and Smith GH . (2000). Oncogene, 19, 992–1001.
Daniel C, DeOme K, Young L, Blair P and Faulkin L . (1968). Proc. Natl. Acad. Sci. USA, 61, 53–60.
DeOme KB, Fauklin LJ, Bern HA and Blair PB . (1959). J. Natl. Cancer Inst., 78, 751–757.
DeOme KB, Miyamoto MJ, Osborn RC, Guzman RC and Lum K . (1978). Cancer Res., 38, 2103–2111.
Henry MD, Triplett AA, Oh KB, Smith GH and Wagner K-U . (2004). Oncogene, 23, 6980–6985.
Jhappan C, Geiser AG, Kordon EC, Bagheri D, Hennighausen L, Roberts AB, Smith GH and Merlino G . (1993). EMBO J., 12, 1835–1845.
Kordon EC, McKnight RA, Jhappan C, Hennighausen L, Merlino G and Smith GH . (1995). Dev. Biol., 168, 47–61.
Kordon EC and Smith GH . (1998). Development, 125, 1921–1930.
Lin H . (2002). Nat. Rev. Genet., 3, 931–939.
Robinson GW, McKnight RA, Smith GH and Hennighausen L . (1995). Development, 121, 2079–2090.
Robinson GW, Smith GH, Gallahan D, Zimmer A, Furth PA and Hennighausen L . (1996). Dev. Dynam., 206, 159–168.
Sivaraman L, Conneely OM, Medina D and O’Malley BW . (2001). Proc. Natl. Acad. Sci. USA, 98, 12379–12384.
Sivaraman L, Stephens LC, Markaverich BM, Clark JA, Krnacik S, Conneely OM, O’Malley BW and Medina D . (1998). Carcinogenesis, 19, 1573–1581.
Smith G, Gallahan D, Zweibel J, Freeman S, Bassin R and Callahan R . (1991). J. Virol., 65, 6365–6370.
Smith GH . (1996). Breast Cancer Res. Treat., 39, 21–31.
Wagner K-U, Boulanger CA, Henry MD, Sagagias M, Hennighausen L and Smith GH . (2002). Development, 129, 1377–1386.
Wagner KU, Wall RJ, St-Onge L, Gruss P, Wynshaw-Boris A, Garrett L, Li M, Furth PA and Hennighausen L . (1997). Nucleic. Acids Res., 25, 4323–4330.
Williams JM and Daniel CW . (1983). Dev. Biol., 97, 274–290.
Acknowledgements
We acknowledge the outstanding contributions of Amy James in the care and breeding of the experimental animals and the stimulating discussions with Dr David Mack regarding the interpretation of our experimental data. We also thank Drs Robert Callahan, David S Salomon and Barbara K Vonderhaar for critical reading of the manuscript. KUW is supported by Susan G Karen Breast Cancer Foundation (BCTRO402956).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boulanger, C., Wagner, KU. & Smith, G. Parity-induced mouse mammary epithelial cells are pluripotent, self-renewing and sensitive to TGF-β1 expression. Oncogene 24, 552–560 (2005). https://doi.org/10.1038/sj.onc.1208185
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208185
Keywords
This article is cited by
-
p63 suppresses the ability of pregnancy-identified mammary epithelial cells (PIMECs) to drive HER2-positive breast cancer
Cell Death & Disease (2021)
-
Hormones induce the formation of luminal-derived basal cells in the mammary gland
Cell Research (2019)
-
Does the Mouse Mammary Gland Arise from Unipotent or Multipotent Mammary Stem/Progenitor Cells?
Journal of Mammary Gland Biology and Neoplasia (2018)
-
Amphiregulin regulates proliferation and migration of HER2-positive breast cancer cells
Cellular Oncology (2018)
-
Tissue specific microenvironments: a key tool for tissue engineering and regenerative medicine
Journal of Biological Engineering (2017)