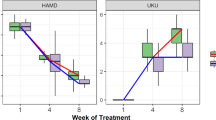
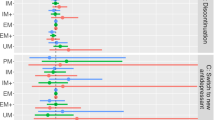
Abstract
The inactivation and clearance of the tricyclic antidepressant imipramine is dependent on CYP2D6 activity. First, CYP2C19 converts imipramine into the active metabolite desipramine, which is then inactivated by CYP2D6. This retrospective single center study aimed to prove whether CYP2C19 and ample CYP2D6 genotyping (taking into consideration four null alleles and three decreased-activity alleles) could be used to predict imipramine and desipramine plasma concentrations in depressed patients, and whether genotype-based drug dose recommendations might assist in the early management of imipramine pharmacotherapy. In 181 subjects with major depressive disorder, drug doses were recorded, imipramine and desipramine plasma concentrations were monitored and CYP2C19 (*2) and CYP2D6 genotype (*3, *4, *5, *6, *9, *10, *41 and gene duplication) were obtained, yielding graded allele-specific CYP2D6 patient groups. Desipramine and imipramine+desipramine plasma concentration per drug dose unit, imipramine dose at steady state, and imipramine dose requirement significantly depended on CYP2D6 genotype (Kruskal–Wallis test, P<0.0001). Mean (±s.d.) drug dose requirements were 131 (±109), 155 (±70), 217 (±95), 245 (±125), 326 (±213), and 509 (±292) mg imipramine/day in carriers of 0, 0.5, 1, 1.5, 2, and >2 active CYP2D6 genes, respectively. Our protocol for CYP2D6 genotyping will thus importantly aid in the prediction of imipramine metabolism, allowing for the use of an adjusted starting dose and faster achievement of predefined imipramine+desipramine plasma levels in all genetic patient subgroups. Therefore, therapeutic efficacy and efficiency may be improved, the number of adverse drug reactions decreased, and hospital stay reduced.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Slattery DA, Hudson AL, Nutt DJ . Invited review: the evolution of antidepressant mechanisms. Fundam Clin Pharmacol 2004; 18: 1–21.
Brøsen K . Some aspects of genetic polymorphism in the biotransformation of antidepressants. Therapie 2004; 59: 5–12.
Steimer W, Zöpf K, Von Amelunxen S, Pfeiffer H, Bachofer J, Popp J et al. Allele-specific change of concentration and functional gene dose for the prediction of steady-state serum concentrations of amitriptyline and nortriptyline in CYP2C19 and CYP2D6 extensive and intermediate metabolizers. Clin Chem 2004; 50: 1623–1633.
Gram LF . Metabolism of tricyclic antidepressants: a review. Dan Med Bull 1974; 21: 218–231.
Preskorn SH, Dorey RC, Jerkovich GS . Therapeutic drug monitoring of tricyclic antidepressants. Clin Chem 1988; 34: 822–828.
Meyer UA . Pharmacogenetics and adverse drug reactions. Lancet 2000; 356: 1667–1671.
Perry PJ, Zeilmann C, Arndt S . Tricyclic antidepressant concentrations in plasma: an estimate of their sensitivity and specificity as a predictor of response. J Clin Psychopharmacol 1994; 14: 230–240.
Madsen H, Rasmussen BB, Brøsen K . Imipramine demethylation in vivo: impact of CYP1A2, CYP2C19, and CYP3A4. Clin Pharmacol Ther 1997; 61: 319–324.
Linder MW, Prough RA, Valdes Jr R . Pharmacogenetics: a laboratory tool for optimizing therapeutic efficiency. Clin Chem 1997; 43: 254–266.
Perry PJ, Pfohl BM, Holstad SG . The relationship between antidepressant response and tricyclic antidepressant plasma concentrations. A retrospective analysis of the literature using logistic regression analysis. Clin Pharmacokinet 1987; 13: 381–392.
Qin X-P, Xie H-G, Wang W, He N, Huang S-L, Xu Z-H et al. Effect of the gene dosage of CYP2C19 on diazepam metabolism in Chinese subjects. Clin Pharmacol Ther 1999; 66: 642–646.
Zanger UM, Fischer J, Raimundo S, Stüven T, Evert BO, Schwab M et al. Comprehensive analysis of the genetic factors determining expression and function of hepatic CYP2D6. Pharmacogenetics 2001; 11: 573–585.
Griese E-U, Zanger UM, Brudermanns U, Gaedigk A, Mikus G, Mörike K et al. Assessment of the predictive power of genotypes for the in-vivo catalytic function of CYP2D6 in a German population. Pharmacogenetics 1998; 8: 15–26.
Bergmann TK, Bathum L, Brosen K . Duplication of CYP2D6 predicts high clearance of desipramine but high clearance does not predict duplication of CYP2D6. Eur J Clin Pharmacol 2001; 57: 123–127.
Gaedigk A, Gotschall RR, Forbes NS, Simon SD, Kearns GL, Leeder JS . Optimization of cytochrome P4502D6 (CYP2D6) phenotype assignment using a genotyping algorithm based on allele frequency data. Pharmacogenetics 1999; 9: 669–682.
Steijns LSW, Van Der Weide J . Ultrarapid drug metabolism: PCR-based detection of CYP2D6 gene duplication. Clin Chem 1998; 44: 914–917.
Zanger UM, Raimundo S, Eichelbaum M . Cytochrome P450 2D6: overview and update on pharmacology, genetics, biochemistry. Naunyn Schmiedebergs Arch Pharmacol 2004; 369: 23–37.
Rau T, Wohlleben G, Wuttke H, Thuerauf N, Lunkenheimer J, Lanczik M et al. CYP2D6 genotype: impact on adverse effects and nonresponse during treatment with antidepressants—a pilot study. Clin Pharmacol Ther 2004; 75: 386–393.
De Leon J, Susce MT, Pan R-M, Fairchild M, Koch WH, Wedlund PJ . The CYP2D6 poor metabolizer phenotype may be associated with risperidone adverse drug reactions and discontinuation. J Clin Psychiatry 2005; 66: 15–27.
Mulder H, Wilmink FW, Beumer TL, Tamminga WJ, Jedema JN, Egberts ACG . The association between cytochrome P450 2D6 genotype and prescription patterns of antipsychotic and antidepressant drugs in hospitalized psychiatric patients: a retrospective follow-up study. J Clin Psychopharmacol 2005; 25: 188–191.
Furman KD, Grimm DR, Mueller T, Holley-Shanks RR, Bertz RJ, Williams LA et al. Impact of CYP2D6 intermediate metabolizer alleles on single-dose desipramine pharmacokinetics. Pharmacogenetics 2004; 14: 279–284.
Raimundo S, Fischer J, Eichelbaum M, Griese E-U, Schwab M, Zanger UM . Elucidation of the genetic basis of the common ‘intermediate metabolizer’ phenotype for drug oxidation by CYP2D6. Pharmacogenetics 2000; 10: 577–581.
Steimer W, Potter JM . Pharmacogenetic screening and therapeutic drugs. Clin Chim Acta 2002; 315: 137–155.
De Morais SM, Wilkinson GR, Blaisdell J, Nakamura K, Meyer UA, Goldstein JA . The major genetic defect responsible for the polymorphism of S-mephenytoin metabolism in humans. J Biol Chem 1994; 269: 15419–15422.
http://www.imm.ki.se/CYPalleles/. Home page of the Human Cytochrome P450 (CYP) Allele Nomenclature Committee. Accessed November 2006.
Hamilton M . A rating scale for depression. J Neurol Neurosurg Psychiatry 1960; 23: 56–62.
Grasmäder K, Verwohlt PL, Rietschel M, Dragicevic A, Müller M, Hiemke C et al. Impact of polymorphisms of cytochrome-P450 isoenzymes 2C9, 2C19 and 2D6 on plasma concentrations and clinical effects of antidepressants in a naturalistic clinical setting. Eur J Clin Pharmacol 2004; 60: 329–336.
Informatorium Medicamentorum. Koninklijke Nederlandse Maatschappij ter bevordering der Pharmacie: The Hague, The Netherlands 2006.
Parfitt K (ed). Martindale: The Complete Drug Reference. Pharmaceutical Press: London, UK, 1999.
Goldstein JA, Blaisdell J . Genetic tests which identify the principal defects in CYP2C19 responsible for the polymorphism in mephenytoin metabolism. Methods Enzymol 1996; 272: 210–218.
Armstrong M, Idle JR, Daly AK . A polymorphic CfoI site in exon 6 of the human cytochrome P450 CYP2D6 gene detected by the polymerase chain reaction. Hum Genet 1993; 91: 616–617.
Topic E, Stefanovic M, Nikolic V, Zoricic I, Ivanisevic A-M, Zuntar I . Detection of CYP2D6*3 and 2D6*4 allelic variants by PCR-restriction fragment length polymorphism. Clin Chem Lab Med 1998; 36: 655–658.
Van der Weide J, Steijns LSW . Impaired drug metabolism: detection of the nonfunctional CYP2D6*6 allele by an allele-specific oligonucleotide hybridisation method. Ned Tijdschr Klin Chem 1998; 23: 245–248.
Gaedigk A, Ryder DL, Bradford LD, Leeder JS . CYP2D6 poor metabolizer status can be ruled out by a single genotyping assay for the -1584G promoter polymorphism. Clin Chem 2003; 49: 1008–1011.
Raimundo S, Toscano C, Klein K, Fischer J, Griese E-U, Eichelbaum M et al. A novel intronic mutation, 2988G>A, with high predictivity for impaired function of cytochrome P450 2D6 in white subjects. Clin Pharmacol Ther 2004; 76: 128–138.
Steen VM, Andreassen OA, Daly AK, Tefre T, Børresen AL, Idle JR et al. Detection of the poor metabolizer-associated CYP2D6(D) gene deletion allele by long-PCR technology. Pharmacogenetics 1995; 5: 215–223.
Løvlie R, Daly AK, Molven A, Idle JR, Steen VM . Ultrarapid metabolizers of debrisoquine: characterization and PCR-based detection of alleles with duplication of the CYP2D6 gene. FEBS Lett 1996; 392: 30–34.
Brøsen K, Klysner R, Gram LF, Otton SV, Bech P, Bertilsson L . Steady-state concentrations of imipramine and its metabolites in relation to the sparteine/debrisoquine polymorphism. Eur J Clin Pharmacol 1986; 30: 679–684.
Tyndale R, Aoyama T, Broly F, Matsunaga T, Inaba T, Kalow W et al. Identification of a new variant CYP2D6 allele lacking the codon encoding Lys-281: possible association with the poor metabolizer phenotype. Pharmacogenetics 1991; 1: 26–32.
Broly F, Meyer UA . Debrisoquine oxidation polymorphism: phenotypic consequences of a 3-base-pair deletion in exon 5 of the CYP2D6 gene. Pharmacogenetics 1993; 3: 123–130.
Johansson I, Oscarson M, Yue Q-Y, Bertilsson L, Sjöqvist F, Ingelman-Sundberg M . Genetic analysis of the Chinese cytochrome P4502D locus: characterization of variant CYP2D6 genes present in subjects with diminished capacity for debrisoquine hydroxylation. Mol Pharmacol 1994; 46: 452–459.
Kootstra-Ros JE, Van Weelden MJM, Hinrichs JWJ, De Smet PAGM, Van der Weide J . Therapeutic drug monitoring of antidepressants and cytochrome P450 genotyping in general practice. J Clin Pharmacol 2006; 46: 1320–1327.
Product Information: Tofranil, Imipramine. Geigy Pharmaceuticals: Summit, NJ, 1995.
Kropp S, Lichtinghagen R, Winterstein K, Schlimme J, Schneider U . Cytochrome P-450 2D6 and 2C19 polymorphisms and length of hospitalization in psychiatry. Clin Lab 2006; 52: 237–240.
Marez D, Legrand M, Sabbagh N, Lo Guidice J-M, Spire C, Lafitte J-J et al. Polymorphism of the cytochrome P450 CYP2D6 gene in a European population: characterization of 48 mutations and 53 alleles, their frequencies and evolution. Pharmacogenetics 1997; 7: 193–202.
Tamminga WJ, Wemer J, Oosterhuis B, De Zeeuw RA, De Leij LFMH, Jonkman JHG . The prevalence of CYP2D6 and CYP2C19 genotypes in a population of healthy Dutch volunteers. Eur J Clin Pharmacol 2001; 57: 717–722.
De Leon J, Barnhill J, Rogers T, Boyle J, Chou W-H, Wedlund PJ . Pilot study of the cytochrome P450-2D6 genotype in a psychiatric state hospital. Am J Psychiatry 1998; 155: 1278–1280.
Steimer W, Müller B, Leucht S, Kissling W . Pharmacogenetics: genotyping of cytochromes P450 2D6 and 2C19 in psychiatric patients treated with tricyclic antidepressants (TCA) or neuroleptics (Abstract). Ther Drug Monit 1999; 21: 474.
Shimoda K, Someya T, Yokono A, Morita S, Hirokane G, Takahashi S et al. Impact of CYP2C19 and CYP2D6 genotypes on metabolism of amitriptyline in Japanese psychiatric patients. J Clin Psychopharmacol 2002; 22: 371–378.
Koyama E, Tanaka T, Chiba K, Kawakatsu S, Morinobu S, Totsuka S et al. Steady-state plasma concentrations of imipramine and desipramine in relation to S-mephenytoin 4′-hydroxylation status in Japanese depressive patients. J Clin Psychopharmacol 1996; 16: 286–293.
Amitai Y, Frischer H . Excess fatality from desipramine and dosage recommendations. Ther Drug Monit 2004; 26: 468–473.
Kirchheiner J, Nickchen K, Bauer M, Wong M-L, Licinio J, Roots I et al. Pharmacogenetics of antidepressants and antipsychotics: the contribution of allelic variations to the phenotype of drug response. Mol Psychiatry 2004; 9: 442–473.
Acknowledgements
Part of this work was supported by an internal grant (Erasmus MC Revolving Fund, Top-Down, MEC 194.305/2000/168B).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schenk, P., van Fessem, M., Verploegh-Van Rij, S. et al. Association of graded allele-specific changes in CYP2D6 function with imipramine dose requirement in a large group of depressed patients. Mol Psychiatry 13, 597–605 (2008). https://doi.org/10.1038/sj.mp.4002057
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.mp.4002057
Keywords
This article is cited by
-
An economic model of the cost-utility of pre-emptive genetic testing to support pharmacotherapy in patients with major depression in primary care
The Pharmacogenomics Journal (2019)
-
A combined high CYP2D6-CYP2C19 metabolic capacity is associated with the severity of suicide attempt as measured by objective circumstances
The Pharmacogenomics Journal (2015)
-
Response to CYP2D6 substrate antidepressants is predicted by a CYP2D6 composite phenotype based on genotype and comedications with CYP2D6 inhibitors
Journal of Neural Transmission (2015)
-
Influence of CYP2D6 and CYP2C19 gene variants on antidepressant response in obsessive-compulsive disorder
The Pharmacogenomics Journal (2014)
-
The Coriell personalized medicine collaborative pharmacogenomics appraisal, evidence scoring and interpretation system
Genome Medicine (2013)